ELECTRONIC RESEARCH STUDY APPLICATION (ERSA) INITIAL APPLICATION GUIDEBOOK. (Version date 03/03/2006)
|
|
|
- John Riley
- 5 years ago
- Views:
Transcription
1 ELECTRONIC RESEARCH STUDY APPLICATION (ERSA) INITIAL APPLICATION GUIDEBOOK (Version date 03/03/2006)
2 Table of Contents ERSA Home Page and Login 3 CRC Desktop 4 Creating a: New Study Application 5 Amendment / Revision 19 Reportable Events (Averse Events, Revised Investigators Brochure, DSMB Reports, Protocol Deviations) 24 Continuing Review 33 Screen Shots Appendix 2
3 ERSA HOME PAGE AND LOGIN The link to the ERSA home page can be found on the IRB intranet site or by typing in the address bar of Internet Explorer. Currently, ERSA is only available through the intranet. The folders to the left of the home page contain useful information, links, and forms templates for you use. Updates of these folders will be ongoing, so visit them often. To login to ERSA, click Login in the top right-hand corner of the screen. (1) The first time you login to ERSA with your assigned user name and password, you will be prompted to change your password. (2) 3
4 CRC DESKTOP From the CRC desktop, you will be able to: Create a new application for A new study Amendments / Revisions Continuing review Reporting AE s, changes in investigator brochures, and protocol deviations Check your inbox for any notifications of study approvals and additional information being requested by the IRB office and ancillary committees (e.g., biosafety, radiation safety, and laser safety). View studies for which you are listed as CRC or study staff and check the status of Study approval Adverse events Continuing Reviews Amendments Your desktop also provides a quick link to the consent forms found in the Forms and Templates folder of the ERSA home page. This allows you easy access to these forms without having to logoff. (3) 4
5 CREATING A NEW STUDY APPLICATION To create a new study, click on the New Study bar on the left-hand side of your desktop page. You will then be presented with the pages of the electronic application. Fields labeled with red text indicate required elements. At any time during the entry of the application, you may stop and come back to the application later. Just click Save in the tool bar between the Back and Continue buttons. Then click Exit. The Basic Study Information page must be completed in order to exit an application. To return to the application from the CRC Desktop: (1) click the Studies tab, (2) click on the title of the study, and (3) click the View Study button in the left margin. Use the Continue button to page through to where you left off. The study pages provide help text in a blue box to the right of the page. Please read the help text if you have any doubt about the information that is being requested. Incomplete information can lead to your application being returned by the IRB. This will slow the assignment to a panel for study review. After completing a page, click the Continue button located at the top or bottom of the page. If you need to go back to a previously completed page, use the Back button within the application. Using the back button of the browser toolbar could result in lost information. (4) 5
6 The Protocol Summary should contain information as found in an abstract. It should be written in lay language and contain information pertaining to the study s purpose, procedures, and methodologies. DO NOT copy and paste from the protocol. It is on this page that you attach the electronic copy of the protocol. If not available electronically, contact the IRB office for assistance. To attach the protocol, click the Add button to open the Submit a Document pop up box. (5) In the box, type Protocol in the Title box with the version date in parentheses. (6) 6
7 Then click the Browse button to locate the appropriate file, and click Open. (7) Then click OK in the Submit a Document pop up box. (8) 7
8 The boxes checked on the Type of Research page will determine which pages of the application will be presented to you. For example, if you select Drugs the Drug Information Page will appear once you click on Continue. (9) Other pages in the application are completed in the same manner. Read the Help text in the blue box for further guidance while completing your application. (10) 8
9 Study Specific Pages The following screens are presented based on your selections on the Type of Research page. Each study is different, therefore, you may not see every page each time you submit a new application. For drug and device studies, the Investigator Brochure, package inserts, and other documents (except the protocol and consent forms) are attached in the FDA and Sponsor Documents section of the drug or device page. Use the same process as used in attaching the protocol document. (11) (12) 9
10 (13) The Diagnostic Radiology / Radiation Safety page will generate an automatic notification to the Radiation Safety Committee. However, it does not constitute an application to this committee for review. The Radiation Safety Committee will contact you for further documentation or application materials as necessary. Continuation of Standard Application Pages When selecting your study population, please keep in mind that the information requested refers to the specific target population. In other words, unless you are specifically recruiting institutionalized individuals as part of your inclusion criteria, you would not check the box for institutionalized individuals. See Help text. (14) 10
11 (15) The Subject Identification and Subject Recruitment pages are self explanatory. See Help text for additional guidance. (16) 11
12 (17) Click Add to open the pop up box and select ethnicity from the drop down box, then type in the number representing the percentage (do not use the % symbol). Click OK. Continue until the total population equals 100. Select the appropriate response regarding request for waiver of informed consent. If a waiver of informed consent is requested, you must provide justification for the request. (18) 12
13 (19) The Informed Consent Process page captures information regarding the informed consent process as well as provides the location to attach your informed consent document(s). Use the same process as used in attaching the protocol document. Based on responses to the risk assessment questions, ERSA will determine eligibility for expedited review. (20) 13
14 (21) At least one of the nine categories must be selected to support expedited review consideration. For definitions of these categories, click the blue Help link found in the upper right corner above the blue help box. When all information is completed, click Finish. Upon clicking finish, you will be taken to the desktop page for the study you just entered. (22) 14
15 (23) Once you have completed your application and are ready to forward it to the PI for review and signature, click the Check Required Elements button to the left. Any incomplete information will be presented to you in red text. You will need to return to the indicated pages for completion. The application can then be forwarded to the PI by clicking the Notify PI button. The PI will automatically receive an in GroupWise, with a link to ERSA, to review and electronically sign the application. NOTE: Only the PI can submit a document to place it into the IRB review process workflow. 15
16 (24) Once the PI has reviewed, signed, and submitted the application, you will receive the following notification: If in the IRB staff review of the application it is found that changes are needed, the IRB staff member will make comments on the application page and return the entire application back to the PI & CRC with comments. You will receive the following notification: (25) 16
17 (26) Once the application is complete and has been reviewed by the IRB staff, the study is assigned to a panel. Once panel assignment and meeting date selection occurs, you will receive the following notification: (27) 17
18 The following is an example of an approval letter generated by ERSA and sent to the PI via GroupWise: (28) 18
19 AMENDMENTS / REVISIONS NOTE: Amendments cannot be submitted for studies that have not been previously reviewed and approved by the IRB. If a study is in process in the IRB office, the process must be completed before you can make any additional changes. In some instances, however, you can request that the study be removed from its current review process and returned for inclusion of the new information to be submitted. To submit a protocol revision, click on the Studies tab on your desktop and then the Approved tab. Find the study needing amendment and click on the study title. To the left of your screen, click on New Amendment. You will be presented with additional pages for completion based on the items checked in question 1.0. (29) 19
20 (30) For consent form changes, enter a brief description of the changes being made. Click the Add button to attach the consent form. If your study already has a consent form attached to it, a link to the form will display in this area, click [edit] next to the form title, and make changes directly in the form. For protocol changes, provide a brief description of the changes in the text box. Later in the amendment form, you will be given the opportunity to return to the main application to attach the revised version of the protocol. (31) 20
21 (32) For advertisement/recruitment material changes, provide a description of the changes in the text box and click Continue. You will be prompted later in the form to return to the main application to attach these materials. Study team members are automatically populated from the original application into the amendment page. Make changes as necessary, either adding or deleting team members. (33) 21
22 (34) The Instructions for Other Changes page provides a link to go to the forms of the main study application for further changes. Click on the blue text link to access the main application. As with the original submission of the application, once changes on a page have been made, click the Continue button on the top or bottom of the page. Once all changes are made, click Finish to return to the amendment application. (35) 22
23 You will then you be presented with the Summary of Changes and Next Steps. This page also provides a link back to the main application pages should you need to go back and make additional changes to the application. (36) Click Finish to complete the Amendment application. Only the PI can submit the amendment application to the IRB office. 23
24 REPORTABLE EVENTS To submit a reportable event, click on the Studies tab on your desktop and then the Approved tab. Find the study for which you are reporting the event and click on the study title. To the left of your screen, click on New Adverse Event. Select the type of event being reported and provide the date of the event and the date the event was reported to the PI. Click Continue. Screens will be presented according to the type of event you select. Internal Serious Adverse Event (37) (38) Select responses as appropriate and click Continue. 24
25 (39) Select the responsible entity(ies) for event monitoring and click Continue. Select the entities that have been notified of the event and attach any related reports (e.g., MedWatch, sponsor reports, DSMB reports) by clicking Add in 2.0. The document pop-up box will appear to attach documentation. (40) 25
26 (41) Indicate whether or not the reported event has changed the overall risk-benefit ratio and the rationale for your response. Click Continue. Complete the responses regarding inclusion of this information in the protocol and/or investigator brochure. Click Continue. (42) 26
27 (43) Complete the responses regarding inclusion of this information in the consent form and whether or not the consent form requires revision as a result of this event. Click Continue. Complete the responses regarding notification to study subjects and/or the need for reconsenting subjects currently enrolled. Click Continue. (44) 27
28 (45) Review the Summary of Event and Next Steps for additional instructions, and click Finish. External Serious Adverse Event Click the Add button in 1.0 to complete the event report. (46) 28
29 (47) Click Ok when complete. If another event is being reported, click add again and complete a second report. Continue this process for each event. To attach safety report documents, click Add in 2.0 and attach document and click Ok to attach the document. Repeat as needed to attach multiple documents. When all documents have been attached, click Continue. (48) 29
30 (49) Review the Summary of Event and Next Steps for additional instructions, and click Finish. Data Safety Monitoring Report (50) Complete the required fields and attach a copy of the report. Click Continue. 30
31 (51) Review the Summary of Event and Next Steps for additional instructions, and click Finish. Protocol Violation (Deviation) Complete questions 1.0 to 5.0 and click Continue. (52) 31
32 (53) Review the Summary of Event and Next Steps for additional instructions, and click Finish. 32
33 CONTINUING REVIEW (CR) NOTE: Continuing Review cannot be submitted for studies that outstanding revisions or reportable events in process with the IRB. If a study is in process in the IRB office, the process must be completed before you can make any additional changes. In some instances, however, you can request that the study be removed from its current review process and returned for inclusion of the new information to be submitted. To submit a continuing review, click on the Studies tab on your desktop and then the Approved tab. Find the study needing continuing review and click on the study title. To the left of your screen, click on New Continuing Review. The study staff information and the protocol summary on the first page will be automatically populated by ERSA. This information is taken from the most recent version of the application in the system. The expiration date is found on the study s approval letter and is entered by clicking on the calendar and selecting the appropriate date. Current protocol activity is a required field. Select the appropriate response, and click Continue. (54) 33
34 (55) Complete all fields as appropriate, referring to help text for guidance / clarification. Remember to scroll down to make sure all fields have been entered. The numbers entered are total numbers for the study, not numbers since the last review. Click Continue. Complete all fields as appropriate, referring to help text for guidance / clarification. Remember to scroll down to make sure all fields have been entered. The numbers entered are reportable events since the last continuing review (or initial review if this is the first CR). Click Continue. (56) 34
35 (57) If a protocol amendment has been implemented but not yet reported to the IRB, the related materials can be attached here for simultaneous review at the time of CR. New/amended protocol, consent documents, and other materials can be attached using the Add button to open the pop-up window. Be sure to give each document an appropriate title (e.g., protocol (with version date), consent form (differentiate if more than one), recruitment brochure, etc.) If any study related publications have resulted since the last review, please attach those documents if available. Click Continue. Review the Summary of Event and Next Steps for additional instructions, and click Finish. (58) 35
UVMClick IRB Study Submission Guide
 UVMClick IRB Study Submission Guide September 2018 Table of Contents How to Login 3 How to Create a New Study 4 Find More Information 5 How to Edit a Study 6 Check the Study for Errors 7 Submit the Study
UVMClick IRB Study Submission Guide September 2018 Table of Contents How to Login 3 How to Create a New Study 4 Find More Information 5 How to Edit a Study 6 Check the Study for Errors 7 Submit the Study
Mentor eirb Researcher User Manual
 Ascension Wisconsin IRB Mentor eirb Researcher User Manual Contents 1 Introduction 1.1 Purpose of this Manual 1.2 User Support 2 Mentor eirb & User Accounts 2.1 About Mentor 2.2 User Roles 2.3 Requesting
Ascension Wisconsin IRB Mentor eirb Researcher User Manual Contents 1 Introduction 1.1 Purpose of this Manual 1.2 User Support 2 Mentor eirb & User Accounts 2.1 About Mentor 2.2 User Roles 2.3 Requesting
STUDY ASSISTANT. Adding a New Study & Submitting to the Review Board. Version 10.03
 STUDY ASSISTANT Adding a New Study & Submitting to the Review Board Version 10.03 Contents Introduction... 3 Add a Study... 3 Selecting an Application... 3 1.0 General Information... 3 2.0 Add Department(s)...
STUDY ASSISTANT Adding a New Study & Submitting to the Review Board Version 10.03 Contents Introduction... 3 Add a Study... 3 Selecting an Application... 3 1.0 General Information... 3 2.0 Add Department(s)...
Introduction to webirb
 Introduction to webirb Training Course for Investigators and Study Staff V: 01.24.13 0 You will learn to 1. Navigate webirb 2. Create a new Study application 3. Respond to IRB Requests 4. Create an Amendment
Introduction to webirb Training Course for Investigators and Study Staff V: 01.24.13 0 You will learn to 1. Navigate webirb 2. Create a new Study application 3. Respond to IRB Requests 4. Create an Amendment
Click. IRB Study Submission Guide
 Click IRB Study Submission Guide October 2016 Table of Contents Logging In... 3 Creating a New Study... 4 Finding More Information... 5 Editing a Study... 5 Checking the Study for Errors... 6 Submitting
Click IRB Study Submission Guide October 2016 Table of Contents Logging In... 3 Creating a New Study... 4 Finding More Information... 5 Editing a Study... 5 Checking the Study for Errors... 6 Submitting
Kuali Research User Guide: Create an IRB Protocol
 Kuali Research User Guide: Create an IRB Protocol Version.0: November 06 Purpose: To create an IRB Protocol document to be used for tracking new Human Subjects Research requests. Trigger / Timing / Frequency:
Kuali Research User Guide: Create an IRB Protocol Version.0: November 06 Purpose: To create an IRB Protocol document to be used for tracking new Human Subjects Research requests. Trigger / Timing / Frequency:
WITH MODIFICATIONS ), S, PHONE NUMBERS OR OTHER FORMS, PLEASE VISIT THE IRB S WEBSITE AT:
 Modification Request Dominican College Institutional Review Board (IRB) IRB #: Date Received: IRB Approval Name & Signature: FOR MORE IRB INSTRUCTIONS, APPLICATIONS (including the RENEWAL FORM that should
Modification Request Dominican College Institutional Review Board (IRB) IRB #: Date Received: IRB Approval Name & Signature: FOR MORE IRB INSTRUCTIONS, APPLICATIONS (including the RENEWAL FORM that should
Reviewers Guide on Clinical Trials
 Reviewers Guide on Clinical Trials Office of Research Integrity & Compliance Version 2 Updated: June 26, 2017 This document is meant to help board members conduct reviews for Full Board: Clinical Trial
Reviewers Guide on Clinical Trials Office of Research Integrity & Compliance Version 2 Updated: June 26, 2017 This document is meant to help board members conduct reviews for Full Board: Clinical Trial
ARC IRB Chair s Manual
 ARC IRB Chair s Manual This guide serves to aid an IRB Chair in becoming familiar with the basic functions of the ARC system and how to review and approve an application. ARC IRB Chairs Manual Page 2 Table
ARC IRB Chair s Manual This guide serves to aid an IRB Chair in becoming familiar with the basic functions of the ARC system and how to review and approve an application. ARC IRB Chairs Manual Page 2 Table
IRBNet User Manual. University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB)
 University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB) IRBNet User Manual Office of Research Integrity and Education Mary Reed Building 222 INTRODUCTION The Office
University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB) IRBNet User Manual Office of Research Integrity and Education Mary Reed Building 222 INTRODUCTION The Office
Coeus Extension IRB Administrative Process Grant Exemption/Expedited Approval (Initial Protocol or Amendment)
 Coeus Extension IRB Administrative Process Grant Exemption/Expedited Approval (Initial Protocol or Amendment) Complete the following steps only when ALL Reviewers have indicated Grant Exemption AND there
Coeus Extension IRB Administrative Process Grant Exemption/Expedited Approval (Initial Protocol or Amendment) Complete the following steps only when ALL Reviewers have indicated Grant Exemption AND there
Core Staff Review Approve with Contingencies
 Core Staff Review Approve with Contingencies Important Information In the event a submission is Approved with Contingencies, extra steps are required to complete the approval process. The basic steps to
Core Staff Review Approve with Contingencies Important Information In the event a submission is Approved with Contingencies, extra steps are required to complete the approval process. The basic steps to
Nova Scotia Health Authority Research Ethics Board. Researcher s (PI) User Manual
 Nova Scotia Health Authority Research Ethics Board Researcher s (PI) User Manual The Researcher s Portal is available through the Login at the following URL: http://nsha-iwk.researchservicesoffice.com/romeo.researcher/login.aspx
Nova Scotia Health Authority Research Ethics Board Researcher s (PI) User Manual The Researcher s Portal is available through the Login at the following URL: http://nsha-iwk.researchservicesoffice.com/romeo.researcher/login.aspx
eirb Electronic Institutional Review Board User Guide For more information contact: Research Administration (803)
 eirb Electronic Institutional Review Board User Guide For more information contact: Research Administration (803) 434 4899 Developed August 2008 by Research Development University of South Carolina 1 Introduction
eirb Electronic Institutional Review Board User Guide For more information contact: Research Administration (803) 434 4899 Developed August 2008 by Research Development University of South Carolina 1 Introduction
eirb User Manual for Research Staff
 eirb User Manual for Research Staff Version 4 1 10.2016 Table of Contents eirb Access... 3 Log-in to eirb... 4 Overview of the eirb Submission and Review Process... 7 Create a New Application and Specify
eirb User Manual for Research Staff Version 4 1 10.2016 Table of Contents eirb Access... 3 Log-in to eirb... 4 Overview of the eirb Submission and Review Process... 7 Create a New Application and Specify
Version GENESIS HEALTH SYSTEM. Institutional Review Board (IRB) IRBNet User s Guide
 Version 1 7-1-2012 GENESIS HEALTH SYSTEM Institutional Review Board (IRB) IRBNet User s Guide G E N E S I S H E A L T H S Y S T E M I N S T I T U T I O N A L R E V I E W B O A R D IRBNet User s Guide Genesis
Version 1 7-1-2012 GENESIS HEALTH SYSTEM Institutional Review Board (IRB) IRBNet User s Guide G E N E S I S H E A L T H S Y S T E M I N S T I T U T I O N A L R E V I E W B O A R D IRBNet User s Guide Genesis
Contents. Researcher Guidance for Reportable Events & Notifying the IRB. Office of Research Integrity & Compliance Version 2 Updated: June 23, 2017
 Researcher Guidance for Reportable Events & Notifying the IRB Office of Research Integrity & Compliance Version 2 Updated: June 23, 2017 This document to help investigators determine what event(s) need
Researcher Guidance for Reportable Events & Notifying the IRB Office of Research Integrity & Compliance Version 2 Updated: June 23, 2017 This document to help investigators determine what event(s) need
Kuali Research User Guide: Create a Protocol Amendment, Renewal or Event
 Kuali Research User Guide: Create a Protocol Amendment, Renewal or Event Version.0: November 06 Purpose: To create an amendment, renewal or event on an existing IRB Protocol document. Trigger / Timing
Kuali Research User Guide: Create a Protocol Amendment, Renewal or Event Version.0: November 06 Purpose: To create an amendment, renewal or event on an existing IRB Protocol document. Trigger / Timing
Information Sheet for Scientific Approvers
 1 Information Sheet for Scientific Approvers This document will provide you with the basic instructions for your role as a scientific approver in OSIRIS. All listed approvers have been designated by their
1 Information Sheet for Scientific Approvers This document will provide you with the basic instructions for your role as a scientific approver in OSIRIS. All listed approvers have been designated by their
Module 6: Validating Decisions
 Table of Contents Module 6: Validating Decisions Module 6 Objective: Covers how Core Staff members process the decisions of the reviewers on each eresearch submission through a decision validation. Refer
Table of Contents Module 6: Validating Decisions Module 6 Objective: Covers how Core Staff members process the decisions of the reviewers on each eresearch submission through a decision validation. Refer
Human Subjects Development Guide
 Research Integrity & Compliance Institutional Review Board Human Subjects Development Guide ERA Version 15, Nov. 2016 Table of Contents Logging In... 3 Adding a Delegate... 4 Viewing Items as a Delegate...
Research Integrity & Compliance Institutional Review Board Human Subjects Development Guide ERA Version 15, Nov. 2016 Table of Contents Logging In... 3 Adding a Delegate... 4 Viewing Items as a Delegate...
Module 6: Validating Decisions
 Table of Contents Module 6: Validating Decisions Module 6 Objective: Covers how Core Staff members process the decisions of the reviewers on each eresearch submission through a decision validation. Refer
Table of Contents Module 6: Validating Decisions Module 6 Objective: Covers how Core Staff members process the decisions of the reviewers on each eresearch submission through a decision validation. Refer
BRANDMAN UNIVERSITY INSTITUTIONAL REVIEW BOARD Continuing Review Request/Closure Report
 BRANDMAN UNIVERSITY INSTITUTIONAL REVIEW BOARD Continuing Review Request/Closure Report Instructions: Please complete all sections of this form and submit with attached documents, forms, and/or explanations
BRANDMAN UNIVERSITY INSTITUTIONAL REVIEW BOARD Continuing Review Request/Closure Report Instructions: Please complete all sections of this form and submit with attached documents, forms, and/or explanations
DISCOVR-e USER MANUAL. Vanderbilt University Human Research Protection Program
 DISCOVR-e USER MANUAL Vanderbilt University Human Research Protection Program Table of Contents Introduction and Overview... 3 Log into the System... 4 Investigator Dashboard... 5 Submitting a New Study...
DISCOVR-e USER MANUAL Vanderbilt University Human Research Protection Program Table of Contents Introduction and Overview... 3 Log into the System... 4 Investigator Dashboard... 5 Submitting a New Study...
SUBMITTING A NEW IBC PROJECT APPLICATION (INITIAL)
 SUBMITTING A After you have established and activated your username and password, you can begin to create an IBC application in IRBNet. To start building a new IBC application, you must first CREATE A
SUBMITTING A After you have established and activated your username and password, you can begin to create an IBC application in IRBNet. To start building a new IBC application, you must first CREATE A
How Does the PI make Pre Review ICF Changes?
 How Does the PI make Pre Review ICF Changes? 1 You, as PI of a study, have received notification from the IRB that Changes [are] required by IRB Staff. These changes include a request for a revision of
How Does the PI make Pre Review ICF Changes? 1 You, as PI of a study, have received notification from the IRB that Changes [are] required by IRB Staff. These changes include a request for a revision of
IRB News : Addition of a new application type for submitting reliance agreements
 In alignment with the NIH mandate for single IRB review that goes into effect in January 2018, HSERA is being updated to include a new reliance agreement specific application. This abbreviated online form
In alignment with the NIH mandate for single IRB review that goes into effect in January 2018, HSERA is being updated to include a new reliance agreement specific application. This abbreviated online form
***** ***** June
 SLU eirb Investigator Guide Saint Louis University ***** eirb Investigator Submitter Guide ***** Institutional Review Board June 2011 http://eirb.slu.edu Institutional Review Board Saint Louis University
SLU eirb Investigator Guide Saint Louis University ***** eirb Investigator Submitter Guide ***** Institutional Review Board June 2011 http://eirb.slu.edu Institutional Review Board Saint Louis University
General Guidance for Maintaining a Regulatory Binder
 General Guidance for Maintaining a Regulatory Binder Study documentation should be well organized, providing a complete and thorough history from protocol development to study completion. Maintaining a
General Guidance for Maintaining a Regulatory Binder Study documentation should be well organized, providing a complete and thorough history from protocol development to study completion. Maintaining a
Protocol Management System (PMS) Investigator User Guide. Harris Health System Administrative Approval
 Protocol Management System (PMS) Investigator User Guide Harris Health System Administrative Approval November 2013 eprotocol - Investigator User Guide 2 Table of Contents 1. INTRODUCTION 3 2. STARTING
Protocol Management System (PMS) Investigator User Guide Harris Health System Administrative Approval November 2013 eprotocol - Investigator User Guide 2 Table of Contents 1. INTRODUCTION 3 2. STARTING
renew & Amend A Protocol training guide
 renew & Amend A Protocol training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 About this guide 4 login 5 Before You Begin Hyperlinks The Table
renew & Amend A Protocol training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 About this guide 4 login 5 Before You Begin Hyperlinks The Table
Last revised: September 30, e-protocol User Guide 1
 e-protocol User Guide Last revised: September 30, 2015 e-protocol User Guide 1 e-protocol is an electronic system for submitting and monitoring the status of Institutional Review Board (IRB) submissions.
e-protocol User Guide Last revised: September 30, 2015 e-protocol User Guide 1 e-protocol is an electronic system for submitting and monitoring the status of Institutional Review Board (IRB) submissions.
UNIVERSITY OF NORTH FLORIDA. Institutional Review Board (IRB) Guide to IRBNet:
 UNIVERSITY OF NORTH FLORIDA Institutional Review Board (IRB) Guide to IRBNet: Version 2 11-15-2013 Table of Contents i Introduction 2 IRBNet User Registration 2 IRBNet ID Number 2 Training and Credentials
UNIVERSITY OF NORTH FLORIDA Institutional Review Board (IRB) Guide to IRBNet: Version 2 11-15-2013 Table of Contents i Introduction 2 IRBNet User Registration 2 IRBNet ID Number 2 Training and Credentials
COEUS LITE IRB COEUS 4.5.1_P3 USER GUIDE
 COEUS LITE IRB COEUS 4.5.1_P3 USER GUIDE Version: August 24, 2016 1 TABLE OF CONTENTS A note about this guide... 4 Getting access to Coeus Lite... 4 Preparing to enter a protocol... 4 Getting Started:
COEUS LITE IRB COEUS 4.5.1_P3 USER GUIDE Version: August 24, 2016 1 TABLE OF CONTENTS A note about this guide... 4 Getting access to Coeus Lite... 4 Preparing to enter a protocol... 4 Getting Started:
irb.unm.edu
 IRBNet Submission Instructions The purpose of this document is to provide IRBNet submission instructions for UNM researchers using IRBNet. Please contact the Office of the IRB for assistance: irb.unm.edu
IRBNet Submission Instructions The purpose of this document is to provide IRBNet submission instructions for UNM researchers using IRBNet. Please contact the Office of the IRB for assistance: irb.unm.edu
OnCore Enterprise Research. Exercises: Subject Administration
 OnCore Enterprise Research Exercises: Subject Administration Clinical Research Coordinator June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Exercises: Subject Administration Contents OnCore
OnCore Enterprise Research Exercises: Subject Administration Clinical Research Coordinator June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Exercises: Subject Administration Contents OnCore
eprmc User Manual For Investigators and Research Staff
 eprmc User Manual For Investigators and Research Staff Table of Contents How to Get an Account... 3 Logging In... 3 Overview of the eprmc Database... 4 Searching for a Protocol... 5 Adding a New Protocol...
eprmc User Manual For Investigators and Research Staff Table of Contents How to Get an Account... 3 Logging In... 3 Overview of the eprmc Database... 4 Searching for a Protocol... 5 Adding a New Protocol...
How to Submit a New UIW IRB Application
 How to Submit a New UIW IRB Application Step Log In Visit https://uiw.forms.ethicalreviewmanager.com/ click on at the top right corner of the page. New Users: Click on New User Fill in the applicable information
How to Submit a New UIW IRB Application Step Log In Visit https://uiw.forms.ethicalreviewmanager.com/ click on at the top right corner of the page. New Users: Click on New User Fill in the applicable information
Drexel University. Page 1 of 48. Version November agf
 Drexel University Page 1 of 48 TABLE OF CONTENTS Getting Started. 3 Creating a New Protocol.. 4 General Information... 7 Organization Information... 9 Investigators/Study Personnel.. 10 Correspondents
Drexel University Page 1 of 48 TABLE OF CONTENTS Getting Started. 3 Creating a New Protocol.. 4 General Information... 7 Organization Information... 9 Investigators/Study Personnel.. 10 Correspondents
Review Response Submission Form
 Review Response Submission Form This form is used to reply to the IRB and address the issues/stipulations raised when the IRB conditionally approves the submission, defers it, returns it as Incomplete
Review Response Submission Form This form is used to reply to the IRB and address the issues/stipulations raised when the IRB conditionally approves the submission, defers it, returns it as Incomplete
Office of Human Research
 Office of Human Research JeffTrial OHR Training Document OHR Workflow and Data Entry Office of Human Research 8/16/2013 Ver. 1.0 Table of Contents OHR and JeffTrial... 2 JeffTrial Workflow... 2 Workflow
Office of Human Research JeffTrial OHR Training Document OHR Workflow and Data Entry Office of Human Research 8/16/2013 Ver. 1.0 Table of Contents OHR and JeffTrial... 2 JeffTrial Workflow... 2 Workflow
STREAMLYNE GUIDE FOR STUDENTS/PRINCIPAL INVESTIGATORS
 STREAMLYNE GUIDE FOR STUDENTS/PRINCIPAL INVESTIGATORS Rev: 01/2017 In This Document Logging In... 1 Creating a New Protocol... 2 Revising a Returned Protocol... 7 Submitting an Amendment or Renewal Application...
STREAMLYNE GUIDE FOR STUDENTS/PRINCIPAL INVESTIGATORS Rev: 01/2017 In This Document Logging In... 1 Creating a New Protocol... 2 Revising a Returned Protocol... 7 Submitting an Amendment or Renewal Application...
Drexel University. Version March Page 1 of 58 Version 01 March 2017 agf
 Drexel University Version March 2017 Page 1 of 58 TABLE OF CONTENTS Getting Started 3 Connecting through the VPN.. 4 Electronic Questionnaires and Applications...10 Welcome to eirb 11 Creating a New Protocol.13
Drexel University Version March 2017 Page 1 of 58 TABLE OF CONTENTS Getting Started 3 Connecting through the VPN.. 4 Electronic Questionnaires and Applications...10 Welcome to eirb 11 Creating a New Protocol.13
OnCore Enterprise Research. Exercises: Subject Administration
 OnCore Enterprise Research Exercises: Subject Administration Clinical Research Coordinator August 2018 P a g e 1 This page is intentionally blank. P a g e 2 Exercises: Subject Administration Contents OnCore
OnCore Enterprise Research Exercises: Subject Administration Clinical Research Coordinator August 2018 P a g e 1 This page is intentionally blank. P a g e 2 Exercises: Subject Administration Contents OnCore
retrieving study documents training guide
 retrieving study documents training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 about this document 4 retrieving study documents 5 current
retrieving study documents training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 about this document 4 retrieving study documents 5 current
University of the Sciences. IRBNet User s Guide
 University of the Sciences IRBNet User s Guide Instructions for On-line Submission of Research Protocols to the Research Committees - IRB, IACUC or IBC. www.irbnet.org If you have any questions regarding
University of the Sciences IRBNet User s Guide Instructions for On-line Submission of Research Protocols to the Research Committees - IRB, IACUC or IBC. www.irbnet.org If you have any questions regarding
Revised: February 28, Investigator/Coordinator Guide
 2014 Revised: February 28, 2014 Table of Contents Getting Started... 3 Navigational Tips... 4 Adding a New Application (Form 1)... 6 Routing Form... 11 Routing for Signatures... 15 Workflow Tracking and
2014 Revised: February 28, 2014 Table of Contents Getting Started... 3 Navigational Tips... 4 Adding a New Application (Form 1)... 6 Routing Form... 11 Routing for Signatures... 15 Workflow Tracking and
Drexel University. Version April Page 1 of 23. Version April agf
 Drexel University Page 1 of 23 TABLE OF CONTENTS Getting Started 3 IRB Protocol Committee Member Reviewer..4 Review IRB Protocol Submission..6 Enter Reviewer Comments... 7 Upload Reviewer Attachments......9
Drexel University Page 1 of 23 TABLE OF CONTENTS Getting Started 3 IRB Protocol Committee Member Reviewer..4 Review IRB Protocol Submission..6 Enter Reviewer Comments... 7 Upload Reviewer Attachments......9
IRB Committee Member Adverse Event (AE) Single Member Review
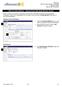 IRB Committee Member AE Single IRB Committee Member Adverse Event (AE) Single When an Adverse Event (AE) is reported by the study team, IRB Staff will assign it to the appropriate reviewer(s). Once an
IRB Committee Member AE Single IRB Committee Member Adverse Event (AE) Single When an Adverse Event (AE) is reported by the study team, IRB Staff will assign it to the appropriate reviewer(s). Once an
How to Create and Submit an Amendment in INSPIR II
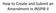 How to Create and Submit an Amendment in INSPIR II Login into INSPIR and from your Home page, click on My Studies to list all the studies that you have access to Locate the study and click on the Open
How to Create and Submit an Amendment in INSPIR II Login into INSPIR and from your Home page, click on My Studies to list all the studies that you have access to Locate the study and click on the Open
SUBMITTING AN IBC AMENDMENT
 The submission of an amendment/modification to an approved IBC protocol requires the creation of a subsequent PACKAGE in a project. After an IBC application is approved, the project may require modifications
The submission of an amendment/modification to an approved IBC protocol requires the creation of a subsequent PACKAGE in a project. After an IBC application is approved, the project may require modifications
Questions and Answers. Converting Existing Protocols into CHeRP IRB
 Questions and Answers Converting Existing Protocols into CHeRP IRB Questions: (hold ctrl and click on the question to jump directly to that answer) 1. Do I need to convert my existing protocol applications
Questions and Answers Converting Existing Protocols into CHeRP IRB Questions: (hold ctrl and click on the question to jump directly to that answer) 1. Do I need to convert my existing protocol applications
ELECTRONIC SITE DELEGATION LOG (esdl) USER MANUAL
 Version 1.0 24May16 ELECTRONIC SITE DELEGATION LOG (esdl) USER MANUAL - Table of Contents - 1. INTRODUCTION... 3 Background... 3 Purpose of the Site Delegation Log... 3 TNCC Contacts... 3 2. SYSTEM REQUIREMENTS...
Version 1.0 24May16 ELECTRONIC SITE DELEGATION LOG (esdl) USER MANUAL - Table of Contents - 1. INTRODUCTION... 3 Background... 3 Purpose of the Site Delegation Log... 3 TNCC Contacts... 3 2. SYSTEM REQUIREMENTS...
Training Manual for Researchers. How to Create an Online Human Ethics Application
 Training Manual for Researchers How to Create an Online Human Ethics Application What is in this document This manual is intended to provide general tips on using functionality specific to QUEST online
Training Manual for Researchers How to Create an Online Human Ethics Application What is in this document This manual is intended to provide general tips on using functionality specific to QUEST online
Office of Research Integrity / Office of Sponsored Projects IACUC PROTOCOL SUBMISSION GUIDE
 Office of Research Integrity / Office of Sponsored Projects IACUC PROTOCOL SUBMISSION GUIDE Coeus Lite 4.5.1.Px Document Version 3 Updated May 2016 Table of Contents ABOUT COEUS... 3 o About the Coeus
Office of Research Integrity / Office of Sponsored Projects IACUC PROTOCOL SUBMISSION GUIDE Coeus Lite 4.5.1.Px Document Version 3 Updated May 2016 Table of Contents ABOUT COEUS... 3 o About the Coeus
The most current protocol information should be entered. This includes incorporation of all past amendments and modifications.
 Georgetown University Institutional Review Board Frequently Asked Questions (FAQs) about eric 1. What is eric? eric stands for Electronic Research and Institutional Compliance. This is an on-line IRB application
Georgetown University Institutional Review Board Frequently Asked Questions (FAQs) about eric 1. What is eric? eric stands for Electronic Research and Institutional Compliance. This is an on-line IRB application
Online Reliance System FAQs
 Online Reliance System FAQs Table of Contents Online Reliance System FAQs... 1 When should the Online Reliance System be used?...3 Who can use the Online Reliance System?...3 Do I have to use the Online
Online Reliance System FAQs Table of Contents Online Reliance System FAQs... 1 When should the Online Reliance System be used?...3 Who can use the Online Reliance System?...3 Do I have to use the Online
How to Submit a New Application
 ehirb User Guide: How to Submit a New Application Last Update February 7, 2014 Intended Audience Purpose Principal Investigator/Researcher To provide the user with step- by- step instructions on how to
ehirb User Guide: How to Submit a New Application Last Update February 7, 2014 Intended Audience Purpose Principal Investigator/Researcher To provide the user with step- by- step instructions on how to
College Deans, Department Chairs & Supervising Faculty
 Revised: 8.26.09 Sam Houston State University Protection of Human Subjects Committee (PHSC/IRB) College Deans, Department Chairs & Supervising Faculty Sam Houston State University has developed a new online
Revised: 8.26.09 Sam Houston State University Protection of Human Subjects Committee (PHSC/IRB) College Deans, Department Chairs & Supervising Faculty Sam Houston State University has developed a new online
Institutional Review Board (IRB)
 IRBNet User s Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org If you have any questions regarding the submission of an IRB protocol contact:
IRBNet User s Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org If you have any questions regarding the submission of an IRB protocol contact:
ERM User guide. The Royal Children s Hospital, Melbourne
 ERM User guide The Royal Children s Hospital, Melbourne Compiled by Katherine Lieschke Ethics Submissions Officer, Melbourne Children s Trials Centre With thanks to the Mater Research Ethics Review Manager
ERM User guide The Royal Children s Hospital, Melbourne Compiled by Katherine Lieschke Ethics Submissions Officer, Melbourne Children s Trials Centre With thanks to the Mater Research Ethics Review Manager
ANNUAL PROGRESS REPORT SUBMISSIONS
 SUBMISSIONS The submission of an annual report of an open study requires the creation of a subsequent PACKAGE in a project. As part of the DU IACUC post-approval monitoring program, all IACUC-approved
SUBMISSIONS The submission of an annual report of an open study requires the creation of a subsequent PACKAGE in a project. As part of the DU IACUC post-approval monitoring program, all IACUC-approved
Guide for Researchers: Online Human Ethics Application Form
 Guide for Researchers: Online Human Ethics Application Form What is Quest Quest is our comprehensive research management system used to administer and support research activity at Victoria University.
Guide for Researchers: Online Human Ethics Application Form What is Quest Quest is our comprehensive research management system used to administer and support research activity at Victoria University.
eresearch IBC Staff Review Step-by-Step Procedures Core Committee Staff Home Page
 1 Core Committee Staff Home Page 2 1. Verify Core Committee Staff is displayed on the home workspace. 2. Note that the Inbox is active. 3. Click the Name of the Registration to be reviewed. 3 4 6 5 4.
1 Core Committee Staff Home Page 2 1. Verify Core Committee Staff is displayed on the home workspace. 2. Note that the Inbox is active. 3. Click the Name of the Registration to be reviewed. 3 4 6 5 4.
My Research, Human Protocols (MRHP) PI User Guide October 2017
 My Research, Human Protocols (MRHP) PI User Guide October 2017 October, 2017 Page 1 of 22 Contents System Requirements... 3 Browsers... 3 Pop-ups... 3 Chrome... 3 Internet Explorer... 4 Firefox... 4 Safari...
My Research, Human Protocols (MRHP) PI User Guide October 2017 October, 2017 Page 1 of 22 Contents System Requirements... 3 Browsers... 3 Pop-ups... 3 Chrome... 3 Internet Explorer... 4 Firefox... 4 Safari...
erequest How to apply guide
 Overview is an application that assists UCB in request life cycle management. UCB has clear guidance in place on what they can support or sponsor. Online requests will go through an internal review and
Overview is an application that assists UCB in request life cycle management. UCB has clear guidance in place on what they can support or sponsor. Online requests will go through an internal review and
Nova Southeastern University IRBManager User Manual IRBMANAGER USER MANUAL
 IRBMANAGER USER MANUAL Contents 1. What is IRBManager?...1 2. IRBManager User Accounts/Login...1 a. Creating an IRBManager Account...1 b. Logging into IRBManager...2 c. Forgot Password...2 d. Locked Out
IRBMANAGER USER MANUAL Contents 1. What is IRBManager?...1 2. IRBManager User Accounts/Login...1 a. Creating an IRBManager Account...1 b. Logging into IRBManager...2 c. Forgot Password...2 d. Locked Out
Student Guide. Click here to log in. A: Log in
 Middlesex Online Research Ethics (MORE) Applicant Guidance Notes (Student & Staff) Note: PhD ethics applications must be submitted as Student Applications. Creating, completing and submitting a Research
Middlesex Online Research Ethics (MORE) Applicant Guidance Notes (Student & Staff) Note: PhD ethics applications must be submitted as Student Applications. Creating, completing and submitting a Research
eiacuc Reference for Veterinarians
 1. Go to https://eiacuc.rutgers.edu 2. Enter your Rutgers NetID and Password. 3. Click Login to enter the site. Logging In My Inbox My Inbox displays all eiacuc submissions associated with you. Identify
1. Go to https://eiacuc.rutgers.edu 2. Enter your Rutgers NetID and Password. 3. Click Login to enter the site. Logging In My Inbox My Inbox displays all eiacuc submissions associated with you. Identify
Chapter 10: Regulatory Documentation
 Table of Contents Chapter 10: Regulatory Documentation... 10-1 10.1 Regulatory Requirements:... 10-1 10.2 Ongoing Regulatory Documents:... 10-4 10.3 After study completion or termination of the trial...
Table of Contents Chapter 10: Regulatory Documentation... 10-1 10.1 Regulatory Requirements:... 10-1 10.2 Ongoing Regulatory Documents:... 10-4 10.3 After study completion or termination of the trial...
Institutional Review Board (IRB)
 IRBNet User s Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org If you have any questions regarding the submission of an IRB protocol, contact:
IRBNet User s Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org If you have any questions regarding the submission of an IRB protocol, contact:
IACUC Module. March 2018
 z IACUC Module March 2018 1 Page left intentionally blank 2 Contents Logins Required for Exercises... 4 Navigation Exercises... 6 Log into the Click IACUC Module... 6 Explore the Inbox... 6 Explore Submissions...
z IACUC Module March 2018 1 Page left intentionally blank 2 Contents Logins Required for Exercises... 4 Navigation Exercises... 6 Log into the Click IACUC Module... 6 Explore the Inbox... 6 Explore Submissions...
How to Use the Comprehensive Ancillary Forms Engine (CAFÉ) for DSMB/ISPRC Initial Submission
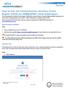 How to Use the Comprehensive Ancillary Forms Engine (CAFÉ) for DSMB/ISPRC Initial Submission The Comprehensive Ancillary Forms Engine (CAFÉ) allows s to submit DSMB and ISPRC submissions to the Jonsson
How to Use the Comprehensive Ancillary Forms Engine (CAFÉ) for DSMB/ISPRC Initial Submission The Comprehensive Ancillary Forms Engine (CAFÉ) allows s to submit DSMB and ISPRC submissions to the Jonsson
Buck-IRB Amendment User Guide
 Office of Research Buck-IRB Amendment User Guide Office of Responsible Research Practices 1960 Kenny Road, Columbus, OH 43210-1016 Institutional Review Board General Guidance for Amendments: The Start
Office of Research Buck-IRB Amendment User Guide Office of Responsible Research Practices 1960 Kenny Road, Columbus, OH 43210-1016 Institutional Review Board General Guidance for Amendments: The Start
Office of Human Research
 Office of Human Research JeffTrial End-User Training Document Regulatory Coordinator Training for Non-Oncology personnel Office of Human Research 8/16/2013 Ver. 1.0 Contents The REG Role: Completing Basic
Office of Human Research JeffTrial End-User Training Document Regulatory Coordinator Training for Non-Oncology personnel Office of Human Research 8/16/2013 Ver. 1.0 Contents The REG Role: Completing Basic
User Guide. Avery Dennison Web Ordering Solution for Steinmart
 User Guide Avery Dennison Web Ordering Solution for Steinmart March 2012 Copyright 2012 A very Dennison The information in this document is subject to change without notice and should not be construed
User Guide Avery Dennison Web Ordering Solution for Steinmart March 2012 Copyright 2012 A very Dennison The information in this document is subject to change without notice and should not be construed
WISER. Protocol Creation, Activation, and Management TRAINING MANUAL. Wake Integrated Solution for Enterprise Research. For Oncology Studies
 WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Oncology Studies TRAINING MANUAL Version June 11, 2018 WELCOME to WISER! Navigation and Home Page
WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Oncology Studies TRAINING MANUAL Version June 11, 2018 WELCOME to WISER! Navigation and Home Page
Guide for Researchers: Online Human Ethics Application Form
 Ethics & Integrity Research Office HUMAN RESEARCH ETHICS ONLINE APPLICATION October 2016/V1.03 Guide for Researchers: Online Human Ethics Application Form ENQUIRIES Senior Human Ethics Officer University
Ethics & Integrity Research Office HUMAN RESEARCH ETHICS ONLINE APPLICATION October 2016/V1.03 Guide for Researchers: Online Human Ethics Application Form ENQUIRIES Senior Human Ethics Officer University
Animal Protocol Development Instructions for Researchers
 Animal Protocol Development Instructions for Researchers OFFICE OF THE VICE PRESIDENT FOR RESEARCH TOPAZ Elements - Protocol Development Instructions for Researchers Page 1 Topaz Elements Animal Protocol
Animal Protocol Development Instructions for Researchers OFFICE OF THE VICE PRESIDENT FOR RESEARCH TOPAZ Elements - Protocol Development Instructions for Researchers Page 1 Topaz Elements Animal Protocol
Click IRB 7.3 IRB Ancillary Reviewer s Guide
 Click IRB 7.3 IRB Ancillary Reviewer s Guide October 2013 Table of Contents Logging In... 3 Login issues... 3 Locating Your To-Do List... 4 Understanding My Inbox... 5 Viewing the Study Details... 6 Viewing
Click IRB 7.3 IRB Ancillary Reviewer s Guide October 2013 Table of Contents Logging In... 3 Login issues... 3 Locating Your To-Do List... 4 Understanding My Inbox... 5 Viewing the Study Details... 6 Viewing
How to Create and Submit a Continuing Review Form (Progress Report) in INSPIR II
 How to Create and Submit a Continuing Review Form (Progress Report) in INSPIR II Creating and Submitting a Continuing Review Form If you are listed as the Study Contact on the study, the system will send
How to Create and Submit a Continuing Review Form (Progress Report) in INSPIR II Creating and Submitting a Continuing Review Form If you are listed as the Study Contact on the study, the system will send
era(infoed) Electronic Submission Instructions and Tips: Initial Application
 era(infoed) Electronic Submission Instructions and Tips: Initial Application This instructional document introduces users to submitting NEW research protocols electronically through the era(infoed) system.
era(infoed) Electronic Submission Instructions and Tips: Initial Application This instructional document introduces users to submitting NEW research protocols electronically through the era(infoed) system.
Maximo Self Service Center
 Maximo Self Service Center Once you have received an email regarding your registration approval, go to the following web address: https://maximo.mysodexo.com Log in to the Self Service Center: Your User
Maximo Self Service Center Once you have received an email regarding your registration approval, go to the following web address: https://maximo.mysodexo.com Log in to the Self Service Center: Your User
STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018. I. Protocol Application & IRB Certification Tutorial...2
 STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018 I. Protocol Application & IRB Certification Tutorial...2 II. Logging In (Password)... 2 III. Creating a New Protocol Record / Menu Bar...
STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018 I. Protocol Application & IRB Certification Tutorial...2 II. Logging In (Password)... 2 III. Creating a New Protocol Record / Menu Bar...
ROCHE/GENENTECH PRACTICAL WORKING GUIDE FOR REQUESTORS
 ROCHE/GENENTECH PRACTICAL WORKING GUIDE FOR REQUESTORS Contents I. INTRODUCTION... 3 II. HOW TO REGISTER AND LOG-IN... 3 III. HOW TO SUBMIT AN APPLICATION... 7 IV. HOW TO PROVIDE ADDITIONAL STUDY INFORMATION...
ROCHE/GENENTECH PRACTICAL WORKING GUIDE FOR REQUESTORS Contents I. INTRODUCTION... 3 II. HOW TO REGISTER AND LOG-IN... 3 III. HOW TO SUBMIT AN APPLICATION... 7 IV. HOW TO PROVIDE ADDITIONAL STUDY INFORMATION...
Once you have created a Project, you must submit the Project s first Package to the ORA.
 CREATING, SIGNING, AND SUBMITTING PACKAGE #1 Once you have created a Project, you must submit the Project s first Package to the ORA. CREATE PACKAGE #1 1. Go to the Designer page. This is where an IRB
CREATING, SIGNING, AND SUBMITTING PACKAGE #1 Once you have created a Project, you must submit the Project s first Package to the ORA. CREATE PACKAGE #1 1. Go to the Designer page. This is where an IRB
eprotocol - Protocol Management System (PMS) Investigator User Guide Version 2.0
 eprotocol - Protocol Management System (PMS) Investigator User Guide Version 2.0 Last Updated: 05/18/2011 Product Version: 2.0.16 eprotocol - PMS - Investigator User Guide 2 Table of Contents 1. INTRODUCTION
eprotocol - Protocol Management System (PMS) Investigator User Guide Version 2.0 Last Updated: 05/18/2011 Product Version: 2.0.16 eprotocol - PMS - Investigator User Guide 2 Table of Contents 1. INTRODUCTION
NAU New Project Submission. Guidance for creating a new project
 NAU New Project Submission Guidance for creating a new project Go to IRBNet Website address: https://www.irbnet.org/release/index.html Login to your account Enter your Username and Password and select
NAU New Project Submission Guidance for creating a new project Go to IRBNet Website address: https://www.irbnet.org/release/index.html Login to your account Enter your Username and Password and select
IRBNet Picture Guide
 IRBNet Picture Guide Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org For any questions or concerns regarding your submission, visit CSUSM
IRBNet Picture Guide Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org For any questions or concerns regarding your submission, visit CSUSM
Velos ecompliance. Researcher User Manual
 Velos ecompliance Researcher User Manual Table of Contents Disclaimer... 4 ecompliance Overview... 5 Contacts... 6 Administrator... 6 Super Users... 6 IT/Technical Support... 6 Quick Help... 7 Getting
Velos ecompliance Researcher User Manual Table of Contents Disclaimer... 4 ecompliance Overview... 5 Contacts... 6 Administrator... 6 Super Users... 6 IT/Technical Support... 6 Quick Help... 7 Getting
InForm Functionality Reference Manual for Sites. Version 1.0
 InForm Functionality Reference Manual for Sites Version 1.0 1-Mar-2012 2012 by Merck & Co., Inc., Whitehouse Station, New Jersey, USA All Rights Reserved No part of this book may be reproduced in any form
InForm Functionality Reference Manual for Sites Version 1.0 1-Mar-2012 2012 by Merck & Co., Inc., Whitehouse Station, New Jersey, USA All Rights Reserved No part of this book may be reproduced in any form
eprotocol - Protocol Management System (PMS) Reviewer User Guide Version 2.0
 eprotocol - Protocol Management System (PMS) Reviewer User Guide Version 2.0 Last Updated: 05/18/2011 Product Version: 2.0.16 eprotocol - PMS - Reviewer User Guide 2 Table of Contents 1. INTRODUCTION 3
eprotocol - Protocol Management System (PMS) Reviewer User Guide Version 2.0 Last Updated: 05/18/2011 Product Version: 2.0.16 eprotocol - PMS - Reviewer User Guide 2 Table of Contents 1. INTRODUCTION 3
WISER. Wake Integrated Solution for Enterprise Research. Protocol Creation, Activation, and Management. For Non-Oncology Studies.
 WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Non-Oncology Studies Version 6/11/2018 TRAINING MANUAL WELCOME to WISER! Navigation and Home Page
WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Non-Oncology Studies Version 6/11/2018 TRAINING MANUAL WELCOME to WISER! Navigation and Home Page
TRAQ DSS Form. Researchers - User Manual
 TRAQ DSS Form Researchers - User Manual January 2014 Introduction to TRAQ TRAQ (Tools for Research at Queen s) is an electronic research management system which replaced all internal certification paper
TRAQ DSS Form Researchers - User Manual January 2014 Introduction to TRAQ TRAQ (Tools for Research at Queen s) is an electronic research management system which replaced all internal certification paper
Submitting a Response to Modifications Required by the IRB to Secure Approval
 Submitting a Response to Modifications Required by the IRB to Secure Approval If you have submitted an item to the IRB, and the IRB has responded with a Modifications Required to Secure Approval memorandum,
Submitting a Response to Modifications Required by the IRB to Secure Approval If you have submitted an item to the IRB, and the IRB has responded with a Modifications Required to Secure Approval memorandum,
Reference Card for ORSP
 Reference Card for ORSP https://eresearch.umich.edu Log in from eresearch Homepage 1. Go to https://eresearch.umich.edu. 2. Click Login in the Proposal Management box. 3. Enter your Login ID (uniqname)
Reference Card for ORSP https://eresearch.umich.edu Log in from eresearch Homepage 1. Go to https://eresearch.umich.edu. 2. Click Login in the Proposal Management box. 3. Enter your Login ID (uniqname)
eprotocol Investigator Role Manual
 eprotocol Investigator Role Manual Table of Contents Table of Contents Auto-Population of Stored User Data...17 5.4 Module Specific Work Flow Sections...18 1 Overview...4 1.1 Things to Remember...4 2 Creating
eprotocol Investigator Role Manual Table of Contents Table of Contents Auto-Population of Stored User Data...17 5.4 Module Specific Work Flow Sections...18 1 Overview...4 1.1 Things to Remember...4 2 Creating
User s Guide. Biomedical Research Alliance of New York
 Using IRBManager to Maintain Your BRANY Studies User s Guide Biomedical Research Alliance of New York Institutional Review Board 1981 Marcus Avenue, Suite 210 Lake Success, NY 11042 P: (516) 470-6900 F:
Using IRBManager to Maintain Your BRANY Studies User s Guide Biomedical Research Alliance of New York Institutional Review Board 1981 Marcus Avenue, Suite 210 Lake Success, NY 11042 P: (516) 470-6900 F:
BOULDER IRB era InfoEd Continuing Review
 BOULDER IRB era InfoEd Continuing Review Last Update: 2017/11/30 Preface: This guide explains how to submit a continuing review for a previously approved expedited or full board study that is nearing expiration.
BOULDER IRB era InfoEd Continuing Review Last Update: 2017/11/30 Preface: This guide explains how to submit a continuing review for a previously approved expedited or full board study that is nearing expiration.
Investigator Activities Quick Reference Guide. Sanofi/Genzyme October 2013
 Investigator Activities Quick Reference Guide Sanofi/Genzyme October 2013 Contents INVESTIGATOR ACTIVITIES QUICK REFERENCE GUIDE... 1 I. INTRODUCTION... 3 II. HOW TO REGISTER AND LOG IN... 6 III. HOW TO
Investigator Activities Quick Reference Guide Sanofi/Genzyme October 2013 Contents INVESTIGATOR ACTIVITIES QUICK REFERENCE GUIDE... 1 I. INTRODUCTION... 3 II. HOW TO REGISTER AND LOG IN... 6 III. HOW TO
