RESOURCES AND REPORTS CTSU WEBSITE ON THE. Alliance Breakout Session November 3, 2017 Martha Hering
|
|
|
- Lorena Jefferson
- 5 years ago
- Views:
Transcription
1 RESOURCES AND REPORTS ON THE CTSU WEBSITE Alliance Breakout Session November 3, 2017 Martha Hering Nov. 3,
2 Agenda Goals of this Presentation Dashboard Quick Views CRISP Accrual Reports Funding Information Regulatory Tab Roster Information Data Quality Portal Patients Subtab Other Resources Nov. 3, 2017 Cancer Trials Support Unit 2
3 Goals Provide a high-level overview of resources on the CTSU website Demonstrate how to find resources and their uses Show best practices via user demonstration Nov. 3, 2017 Cancer Trials Support Unit 3
4 Dashboard Tab Allows customized views of data related to associated site(s) May set as your landing page Use default profiles or customize view from a selection of portlets Default Profiles: Protocol, Enrollment, Regulatory and General Nov. 3, 2017 Cancer Trials Support Unit 4
5 CTSU Report and Information Subscription Portal (CRISP) Allows subscriptions to a variety of alerts Located on main tool bar Includes: Enrollment Updates: multi-step enrollments, accrual updates and step changes Person Roster: changes at selected rosters Protocol Updates: protocols in selected area of interest Regulatory Updates: expiring approvals, changes site registration status, IRB approval received, registration status change on selected protocols 5
6 User Experience We designed this Are you using this? Nov. 3, 2017 Cancer Trials Support Unit 6
7 Accrual Reports Located at the bottom of the Protocol Browser Tree Categories: NCTN, ETCTN, NCORP and AYA Filters: protocol status and phase Keyword search Group by: study type, disease, Lead Org Export to: PDF, CSV or Excel or print Nov. 3, 2017 Cancer Trials Support Unit 7
8 Limited View Reports Located under the Reports tab Limited to Lead Academic Participating Site (LAPS) sites Includes subtabs LAPS Accrual Report: accruals to protocols under the LAPS grant LAPS Budget Analysis: summary of accrual costs and percentages towards grant funding goals 8
9 Upcoming Accrual Reports Coming Soon - Accrual Graph and Details On Protocol-Specific Home Pages 9
10 Funding Information (1) NCI Funding Sources Lists available funds per protocols Located under funding tab for each protocol 10
11 Funding Information (2) OPEN Funding Reports Reimbursements for QOL and Biospecimens 11
12 Regulatory Tab Site Registration view regulatory status by site Protocol Requirements view protocol specific requirements (PSR) by study Regulatory Submissions track processing status of regulatory submissions CIRB Site Preferences view application of CIRB approval across affiliated sites Provider Association view aligned radiation/imaging providers 12
13 Roster Information Person and Org Roster Browser View persons and org rosters associated with affiliated sites for the NCTN, ETCTN, CITN, and CIRB Filter on site, role, and CTEP status Perform following actions Add/Remove role Add/Remove IVR, NPIVR, AP, and A Add affiliate/sub-affiliate sites to rostered swim lane 13
14 RUMS Person Roster Browser Use i buttons to view detailed person data Practice sites, IRBs, and HSP/GCP training Future enhancements Filter by Registration Type NCI Registration Expiry date in Excel output 14
15 User Experience 15
16 Data Quality Portal (DQP) One Stop Shopping Access all Rave studies Direct link into Rave URLs and directly manage queries/delinquencies Monitor data quality and timeliness Review metrics and performance Standardized Experience Consistent experience across LPOs and Rave studies Reports and other tools 16
17 DQP Metrics and Reports Metric Tables Grid/Table layout and export features Metric Reports Graphic layout, aging report, and totals by form and site Timeliness Reports Quarterly updates for query and timeliness 17
18 User Experience 18
19 Additional Resources (1) Patient Subtab Relocated from Clinical Data Tab (CDT) to Rave/DQP Provides summary of all registration steps Study Agent Report Located in Protocol Browser tree Tags Upcoming feature which allows you to add notes to the website 19
20 Additional Resources (2) Other OPEN Reports Screening Log enrollments Accrual Maps and charts 20
21 Nov. 3, 2017 Cancer Trials Support Unit 21
22 Extra Screen Shots 22
23 23
MULTI-GROUP AUDITS, THE CENTRAL MONITORING PORTAL, AND OTHER CTSU UPDATES. Agenda MULTI-GROUP AUDITS 10/5/2017 OISHI SYMPOSIUM. Multi-Group Audits
 MULTI-GROUP AUDITS, THE CENTRAL MONITORING PORTAL, AND OTHER CTSU UPDATES OISHI SYMPOSIUM 1 Agenda Multi-Group Audits Central Monitoring Portal Website and Administrative Updates 2 MULTI-GROUP AUDITS 3
MULTI-GROUP AUDITS, THE CENTRAL MONITORING PORTAL, AND OTHER CTSU UPDATES OISHI SYMPOSIUM 1 Agenda Multi-Group Audits Central Monitoring Portal Website and Administrative Updates 2 MULTI-GROUP AUDITS 3
ALLIANCE CLINICAL RESEARCH PROFESSIONAL INFORMATION SESSION CTSU WEBSITE AND OPEN UPDATES
 ALLIANCE CLINICAL RESEARCH PROFESSIONAL INFORMATION SESSION CTSU WEBSITE AND OPEN UPDATES Kendra Godfrey Barrow, BS, CCRP Martha Hering, RN, BA, MHA, CCRP 1 1. CTSU Website Updates 1. Protocol Tab New
ALLIANCE CLINICAL RESEARCH PROFESSIONAL INFORMATION SESSION CTSU WEBSITE AND OPEN UPDATES Kendra Godfrey Barrow, BS, CCRP Martha Hering, RN, BA, MHA, CCRP 1 1. CTSU Website Updates 1. Protocol Tab New
CTSU UPDATES ALLIANCE FALL MEETING
 CTSU UPDATES ALLIANCE FALL MEETING November 2, 2017 Martha Hering, RN, MHA, CCRP Nov. 2, 2017 1 Agenda Central Monitoring Multi-Group Audits Series Adverse Event (SAE) Integration Electronic Medical Record
CTSU UPDATES ALLIANCE FALL MEETING November 2, 2017 Martha Hering, RN, MHA, CCRP Nov. 2, 2017 1 Agenda Central Monitoring Multi-Group Audits Series Adverse Event (SAE) Integration Electronic Medical Record
CTSU UPDATES. Spring Site Operations Meeting 4/11/2018 1
 CTSU UPDATES Spring Site Operations Meeting 4/11/2018 1 Today s Updates Delegation of Tasks Log (DTL) Imaging and Radiation Oncology Core (IROC) Integration 4/11/2018 Cancer Trials Support Unit 2 DELEGATION
CTSU UPDATES Spring Site Operations Meeting 4/11/2018 1 Today s Updates Delegation of Tasks Log (DTL) Imaging and Radiation Oncology Core (IROC) Integration 4/11/2018 Cancer Trials Support Unit 2 DELEGATION
Registration and Credential Repository (RCR) Donna A Shriner, PharmD, MPH
 Registration and Credential Repository (RCR) Donna A Shriner, PharmD, MPH April 21, 2017 Introduction The Registration and Credential Repository (RCR) will: Provide a self-service online person registration
Registration and Credential Repository (RCR) Donna A Shriner, PharmD, MPH April 21, 2017 Introduction The Registration and Credential Repository (RCR) will: Provide a self-service online person registration
OPEN v8.1 Site User Guide Revision 14
 CTSU Cancer Trials Support Unit Revision 14 December 7, 2017 Overview of OPEN Document Information Revision Information for the Revision History # Date By Description 1.0 19-SEP-2013 Mark Stauffer Updated
CTSU Cancer Trials Support Unit Revision 14 December 7, 2017 Overview of OPEN Document Information Revision Information for the Revision History # Date By Description 1.0 19-SEP-2013 Mark Stauffer Updated
OPEN Portal v8.1 Group User Guide Revision 16
 CTSU Cancer Trials Support Unit Revision 16 December 7, 2017 Document Information Revision Information for the Revision History # Date By Description 1 10-JUL-2013 Mark Stauffer Updated document to have
CTSU Cancer Trials Support Unit Revision 16 December 7, 2017 Document Information Revision Information for the Revision History # Date By Description 1 10-JUL-2013 Mark Stauffer Updated document to have
OPEN T&UM Quick Reference Site User Guide Revision 13
 CTSU Cancer Trials Support Unit OPEN T&UM Quick Reference Site User Guide Revision 13 August 21, 2017 Document Information Revision Information for the Revision History # Date By Description 1 2014/07/29
CTSU Cancer Trials Support Unit OPEN T&UM Quick Reference Site User Guide Revision 13 August 21, 2017 Document Information Revision Information for the Revision History # Date By Description 1 2014/07/29
Registration and Credential Repository (RCR) Update and Demo
 Registration and Credential Repository (RCR) Update and Demo Matthew Boron, RPh PMB, CTEP, NCI November 2-3, 2017 Goals and Objectives Describe why the Registration and Credential Repository was implemented
Registration and Credential Repository (RCR) Update and Demo Matthew Boron, RPh PMB, CTEP, NCI November 2-3, 2017 Goals and Objectives Describe why the Registration and Credential Repository was implemented
The edcf Program Helpful hints and a walk through (updated Sept. 09)
 The edcf Program Helpful hints and a walk through (updated Sept. 09) Accessing edcf Program Below is the link to the ECOG Web Application Portal: https://webapps.ecog.org/ webapps.ecog.org/authworld/ Note:
The edcf Program Helpful hints and a walk through (updated Sept. 09) Accessing edcf Program Below is the link to the ECOG Web Application Portal: https://webapps.ecog.org/ webapps.ecog.org/authworld/ Note:
Office of Human Research
 Office of Human Research JeffTrial End-User Training Document Regulatory Coordinator Training for Non-Oncology personnel Office of Human Research 8/16/2013 Ver. 1.0 Contents The REG Role: Completing Basic
Office of Human Research JeffTrial End-User Training Document Regulatory Coordinator Training for Non-Oncology personnel Office of Human Research 8/16/2013 Ver. 1.0 Contents The REG Role: Completing Basic
TRIAD. What is TRIAD. Accessing TRIAD. TRIAD Presentation 2/6/2014
 TRIAD Denise Manfredi Assistant Director RTQA February 2014 What is TRIAD TRIAD is the American College of Radiology s (ACR s) image and data exchange platform. TRIAD is a standards based open architecture
TRIAD Denise Manfredi Assistant Director RTQA February 2014 What is TRIAD TRIAD is the American College of Radiology s (ACR s) image and data exchange platform. TRIAD is a standards based open architecture
CRA Workbench: Access DATA SUBMISSION PRE-RAVE & RAVE. Clinical Trials Training Course Data Submission. Pre-RAVE
 DATA SUBMISSION PRE-RAVE & RAVE JACQUELINE SCURLOCK & SEAN O BRYAN CLINICAL RESEARCH DATA COORDINATORS SDMC 1 Pre-RAVE Also known as: Chart Manager The Legacy System CRA Workbench Used for studies activated
DATA SUBMISSION PRE-RAVE & RAVE JACQUELINE SCURLOCK & SEAN O BRYAN CLINICAL RESEARCH DATA COORDINATORS SDMC 1 Pre-RAVE Also known as: Chart Manager The Legacy System CRA Workbench Used for studies activated
OnCore Enterprise Research. Basics: Reporting and Searching
 OnCore Enterprise Research Basics: Reporting and Searching All Access Roles June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Table of Contents What is the OnCore Enterprise Research System?...
OnCore Enterprise Research Basics: Reporting and Searching All Access Roles June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Table of Contents What is the OnCore Enterprise Research System?...
RTOG Common Data Management System Implementation. Shashi Solipuram ACR IT Tao Wang ACR IT
 RTOG Common Data Management System Implementation Shashi Solipuram ACR IT Tao Wang ACR IT Radiation Therapy Oncology Group (RTOG) Implemented three trials in Medidata Rave Single and multi-step registration
RTOG Common Data Management System Implementation Shashi Solipuram ACR IT Tao Wang ACR IT Radiation Therapy Oncology Group (RTOG) Implemented three trials in Medidata Rave Single and multi-step registration
JeffTrial Training Document Protocol Management. Jefferson Coordinating Center for Clinical Research 9/10/2013 Ver. 1.1
 JeffTrial Training Document Protocol Management Jefferson Coordinating Center for Clinical Research 9/10/2013 Ver. 1.1 Table of Contents JCCCR Process Flow Diagram... 3 The REG Role, PART I: Entering Basic
JeffTrial Training Document Protocol Management Jefferson Coordinating Center for Clinical Research 9/10/2013 Ver. 1.1 Table of Contents JCCCR Process Flow Diagram... 3 The REG Role, PART I: Entering Basic
PRIDE: Guidelines for CRAs to Access the ECOG-ACRIN Systems for EASY Entry of PROs (EASEE-PRO)
 to Access the ECOG-ACRIN Systems for EASY Entry of PROs (EASEE-PRO) The Participant Reporting Interface and Data Entry (PRIDE https://pride.stat.brown.edu) portal is the secure web interface that both
to Access the ECOG-ACRIN Systems for EASY Entry of PROs (EASEE-PRO) The Participant Reporting Interface and Data Entry (PRIDE https://pride.stat.brown.edu) portal is the secure web interface that both
Records and Enrollment Participants Guide
 IBM Cognos Analytics Welcome to Cognos Records and Enrollment Training! Today s objectives include: Understand University policies on data security including FERPA Understand which package/report to use
IBM Cognos Analytics Welcome to Cognos Records and Enrollment Training! Today s objectives include: Understand University policies on data security including FERPA Understand which package/report to use
NHPNet User Guide Updated August 2017
 NHPNet User Guide Updated August 2017 Table of Contents Introduction... 3 How to Register for NHPNet... 6 Your Profile... 16 User Administration... 22 Authorizations & Referrals... 33 Provider Enrollment
NHPNet User Guide Updated August 2017 Table of Contents Introduction... 3 How to Register for NHPNet... 6 Your Profile... 16 User Administration... 22 Authorizations & Referrals... 33 Provider Enrollment
TrialSearch v2.0 User Guide
 TrialSearch v2.0 User Guide Revised August 2016 by Robin Richardson Contents 1.0. Introduction to TrialSearch... 3 2.0. Searching or Browsing Studies... 3 2.1. Locating Trials and Viewing Trial Details
TrialSearch v2.0 User Guide Revised August 2016 by Robin Richardson Contents 1.0. Introduction to TrialSearch... 3 2.0. Searching or Browsing Studies... 3 2.1. Locating Trials and Viewing Trial Details
Records and Enrollment Participants Guide
 IBM Cognos Analytics Welcome to Cognos Records and Enrollment Training! Today s objectives include: Gain a Basic Understanding of Cognos Understand University policies on data security including FERPA
IBM Cognos Analytics Welcome to Cognos Records and Enrollment Training! Today s objectives include: Gain a Basic Understanding of Cognos Understand University policies on data security including FERPA
Office of Human Research
 Office of Human Research JeffTrial OHR Training Document OHR Workflow and Data Entry Office of Human Research 8/16/2013 Ver. 1.0 Table of Contents OHR and JeffTrial... 2 JeffTrial Workflow... 2 Workflow
Office of Human Research JeffTrial OHR Training Document OHR Workflow and Data Entry Office of Human Research 8/16/2013 Ver. 1.0 Table of Contents OHR and JeffTrial... 2 JeffTrial Workflow... 2 Workflow
JeffTrial Training Document Coordinator Training. Thomas Jefferson University 1/15/2016 Ver. 2.0
 JeffTrial Training Document Coordinator Training Thomas Jefferson University 1/15/2016 Ver. 2.0 Table of Contents Clinical Study Process Flow Diagram... 4 Navigation... 5 Getting Started... 5 User Roles...
JeffTrial Training Document Coordinator Training Thomas Jefferson University 1/15/2016 Ver. 2.0 Table of Contents Clinical Study Process Flow Diagram... 4 Navigation... 5 Getting Started... 5 User Roles...
MicroStrategy Quick Guide: Running the PI Report
 MicroStrategy Quick Guide: Running the PI Report ITS Business Intelligence Group Go to reporting.gmu.edu and click on Login to Microstrategy ACTION Enter User name and Password. Keep default selection
MicroStrategy Quick Guide: Running the PI Report ITS Business Intelligence Group Go to reporting.gmu.edu and click on Login to Microstrategy ACTION Enter User name and Password. Keep default selection
ACRIN 4703 Detection of Early lung Cancer Among Military Personnel Study 1 (DECAMP 1): Diagnosis and Surveillance of Indeterminate Pulmonary Nodules
 ACRIN 4703 Detection of Early lung Cancer Among Military Personnel Study 1 (DECAMP 1): Diagnosis and Surveillance of Indeterminate Pulmonary Nodules Data Management Training Lindsey Dymond, Senior Research
ACRIN 4703 Detection of Early lung Cancer Among Military Personnel Study 1 (DECAMP 1): Diagnosis and Surveillance of Indeterminate Pulmonary Nodules Data Management Training Lindsey Dymond, Senior Research
WISER. Protocol Creation, Activation, and Management TRAINING MANUAL. Wake Integrated Solution for Enterprise Research. For Oncology Studies
 WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Oncology Studies TRAINING MANUAL Version June 11, 2018 WELCOME to WISER! Navigation and Home Page
WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Oncology Studies TRAINING MANUAL Version June 11, 2018 WELCOME to WISER! Navigation and Home Page
Provider Portal User Guide
 Provider Portal User Guide Updated: January 1, 2019 Table of Contents Introduction... 1 How to Register for the Provider Portal... 3 Manage Your Profile... 5 User Administration... 8 Authorizations & Referrals...
Provider Portal User Guide Updated: January 1, 2019 Table of Contents Introduction... 1 How to Register for the Provider Portal... 3 Manage Your Profile... 5 User Administration... 8 Authorizations & Referrals...
MicroStrategy Quick Guide: Running the PI Report ITU Data Mart Support Group Go to reporting.gmu.edu and click on Login to Microstrategy
 MicroStrategy Quick Guide: Running the PI Report ITU Data Mart Support Group Go to reporting.gmu.edu and click on Login to Microstrategy ACTION Enter User name and Password. Keep default selection of LDAP
MicroStrategy Quick Guide: Running the PI Report ITU Data Mart Support Group Go to reporting.gmu.edu and click on Login to Microstrategy ACTION Enter User name and Password. Keep default selection of LDAP
Admissions & Intro to Report Editing Participants Guide
 IBM Cognos Analytics Admissions & Intro to Report Editing Participants Guide Welcome to Cognos - Admissions and Introduction to Report Editing! Today s objectives include: Gain a Basic Understanding of
IBM Cognos Analytics Admissions & Intro to Report Editing Participants Guide Welcome to Cognos - Admissions and Introduction to Report Editing! Today s objectives include: Gain a Basic Understanding of
Tutorial 8 Sharing, Integrating and Analyzing Data
 Tutorial 8 Sharing, Integrating and Analyzing Data Microsoft Access 2013 Objectives Session 8.1 Export an Access query to an HTML document and view the document Import a CSV file as an Access table Use
Tutorial 8 Sharing, Integrating and Analyzing Data Microsoft Access 2013 Objectives Session 8.1 Export an Access query to an HTML document and view the document Import a CSV file as an Access table Use
WISER. Wake Integrated Solution for Enterprise Research. Protocol Creation, Activation, and Management. For Non-Oncology Studies.
 WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Non-Oncology Studies Version 6/11/2018 TRAINING MANUAL WELCOME to WISER! Navigation and Home Page
WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Non-Oncology Studies Version 6/11/2018 TRAINING MANUAL WELCOME to WISER! Navigation and Home Page
Key. General. Host Account Functionality. Included, no-cost Not Included Add-on/Low cost $ Add-on/High cost $$$
 Key Included, no-cost Not Included Add-on/Low cost $ Add-on/High cost $$$ General X Cost $ $ $$$ $$$ Complete cross-functionality between native mobile app and web-based system Ability to create host accounts
Key Included, no-cost Not Included Add-on/Low cost $ Add-on/High cost $$$ General X Cost $ $ $$$ $$$ Complete cross-functionality between native mobile app and web-based system Ability to create host accounts
The link to access the ESR Portal is https://my.esr.nhs.uk or you can access it via the Applications age on the Trust Intranet
 This guide is intended to provide you will an overview of the My ESR Dashboard. The My ESR dashboard is available to all users and the default dashboard displayed when accessing the ESR Portal Accessing
This guide is intended to provide you will an overview of the My ESR Dashboard. The My ESR dashboard is available to all users and the default dashboard displayed when accessing the ESR Portal Accessing
Physician Time Study System. User Guide for Supporting Staff
 Physician Time Study System User Guide for Supporting Staff 1 Table of Contents 1. Introduction... 3 2. About your Support Staff Account... 3 3. Getting Started... 4 4. Your Main Navigation Area... 5 5.
Physician Time Study System User Guide for Supporting Staff 1 Table of Contents 1. Introduction... 3 2. About your Support Staff Account... 3 3. Getting Started... 4 4. Your Main Navigation Area... 5 5.
CTSU Image Portal Rave Custom Function Setup Guide Revision 1.5
 CTSU Cancer Trials Support Unit CTSU Image Portal Rave Custom Function Setup Guide Revision 1.5 July 12, 2013 Document Information Revision Information for the Version 1.5 Revision History # Date By Description
CTSU Cancer Trials Support Unit CTSU Image Portal Rave Custom Function Setup Guide Revision 1.5 July 12, 2013 Document Information Revision Information for the Version 1.5 Revision History # Date By Description
Insight 3.4: Release Date September 27, Help Desk/ Service Center # (If Available) Internal TFS # Description
 Insight 3.4: Release Date September 27, 2013 Internal TFS # Help Desk/ Service Center # (If Available) Description Agreements- Patient Care Implement Patient Care Details for BWH Patient Care Details are
Insight 3.4: Release Date September 27, 2013 Internal TFS # Help Desk/ Service Center # (If Available) Description Agreements- Patient Care Implement Patient Care Details for BWH Patient Care Details are
Getting started. Create event content. Quick Start Guide. Quick start Adobe Connect for Webinars
 Quick start Adobe Connect for Webinars Adobe Connect Event enables you to manage the full life cycle of large or small events, including registration, invitations, reminders, and reports. Adobe Connect
Quick start Adobe Connect for Webinars Adobe Connect Event enables you to manage the full life cycle of large or small events, including registration, invitations, reminders, and reports. Adobe Connect
You can access ProQuest, as well as any of the other available subscription databases, from the library Web page,
 Harold Washington College Library Using ProQuest to Search for Articles About ProQuest ProQuest is one of many subscription databases available through the Harold Washington College Library. ProQuest offers
Harold Washington College Library Using ProQuest to Search for Articles About ProQuest ProQuest is one of many subscription databases available through the Harold Washington College Library. ProQuest offers
ESR Portal Familiarisation User Guide
 ESR Portal Familiarisation User Guide Page 1 April 2017 It s your ESR ESR Portal Familiarisation - User Guide This guide is intended to provide you with an overview of the My ESR Dashboard. The My ESR
ESR Portal Familiarisation User Guide Page 1 April 2017 It s your ESR ESR Portal Familiarisation - User Guide This guide is intended to provide you with an overview of the My ESR Dashboard. The My ESR
Investigator Activities Quick Reference Guide. Sanofi/Genzyme October 2013
 Investigator Activities Quick Reference Guide Sanofi/Genzyme October 2013 Contents INVESTIGATOR ACTIVITIES QUICK REFERENCE GUIDE... 1 I. INTRODUCTION... 3 II. HOW TO REGISTER AND LOG IN... 6 III. HOW TO
Investigator Activities Quick Reference Guide Sanofi/Genzyme October 2013 Contents INVESTIGATOR ACTIVITIES QUICK REFERENCE GUIDE... 1 I. INTRODUCTION... 3 II. HOW TO REGISTER AND LOG IN... 6 III. HOW TO
Health Messenger User Guide
 Health Messenger User Guide V 2.6 3/09/2018 USER GUIDE TABLE OF CONTENTS 1.0 GENERAL INFORMATION 1.1 System Overview 1.2 Support 1.3 Uploading Export Files 1.4 Minimum System Requirements 2.0 GETTING STARTED
Health Messenger User Guide V 2.6 3/09/2018 USER GUIDE TABLE OF CONTENTS 1.0 GENERAL INFORMATION 1.1 System Overview 1.2 Support 1.3 Uploading Export Files 1.4 Minimum System Requirements 2.0 GETTING STARTED
NCI s Learning Management System (LMS) Instructor-Led Training (ILT) Learner Guide
 NCI s Learning Management System (LMS) Instructor-Led Training (ILT) Learner Guide 1 CONTENTS Terms Definitions.3 Access Instructor-Led Training (ILT).. 3 Register for Instructor-Led Training (ILT)...5
NCI s Learning Management System (LMS) Instructor-Led Training (ILT) Learner Guide 1 CONTENTS Terms Definitions.3 Access Instructor-Led Training (ILT).. 3 Register for Instructor-Led Training (ILT)...5
Sophos Central Enterprise. Help
 Help Contents About Sophos Central Enterprise...1 Dashboard... 2 Alerts... 3 Logs... 4 Audit Logs...4 Sub-Estates... 6 Unlink sub-estates... 7 Delete sub-estates...8 Licenses...9 Trials...10 Manage Administrators...
Help Contents About Sophos Central Enterprise...1 Dashboard... 2 Alerts... 3 Logs... 4 Audit Logs...4 Sub-Estates... 6 Unlink sub-estates... 7 Delete sub-estates...8 Licenses...9 Trials...10 Manage Administrators...
Comprehensive Capabilities Comparison
 page 1 of 9 Comprehensive Capabilities Comparison General Key Included, no added cost Add-on/Low cost $ Not Available X Add-on/High cost $$$ Cost $ $ $$$ $$$ Complete cross-functionality between native
page 1 of 9 Comprehensive Capabilities Comparison General Key Included, no added cost Add-on/Low cost $ Not Available X Add-on/High cost $$$ Cost $ $ $$$ $$$ Complete cross-functionality between native
Submission Guidelines
 Submission Guidelines Clinical Trial Results invites the submission of phase I, II, and III clinical trials for publication in a brief print format, with full trials results online. We encourage the submission
Submission Guidelines Clinical Trial Results invites the submission of phase I, II, and III clinical trials for publication in a brief print format, with full trials results online. We encourage the submission
Office of Clinical Research. CTMS Reference Guide Report Building
 Office of Clinical Research CTMS Reference Guide Report Building Reports Reports Reports Overview The Reports module provides you with the ability to quickly create and edit reports based on data stored
Office of Clinical Research CTMS Reference Guide Report Building Reports Reports Reports Overview The Reports module provides you with the ability to quickly create and edit reports based on data stored
PQRS - Reports Monday, June 17, 2013
 Copyright 2012 Pulse Systems, Inc. Page 1 of 36 Welcome to the Reports lesson using PQRS. Click Continue to proceed. Copyright 2012 Pulse Systems, Inc. Page 2 of 36 The PQRS Reports screen enables users
Copyright 2012 Pulse Systems, Inc. Page 1 of 36 Welcome to the Reports lesson using PQRS. Click Continue to proceed. Copyright 2012 Pulse Systems, Inc. Page 2 of 36 The PQRS Reports screen enables users
LOCATION TRAINING DATE. For operating AfterSchool21 Data Program Management Software. P : opt. 1 E :
 ADDRESS 7100 W. Center Street Milwaukee, WI 53210 CONTACT P : +1 414 257 9400 opt. 1 E : support@cayen.net A division of TransACT Communications, LLC For operating AfterSchool21 Data Program Management
ADDRESS 7100 W. Center Street Milwaukee, WI 53210 CONTACT P : +1 414 257 9400 opt. 1 E : support@cayen.net A division of TransACT Communications, LLC For operating AfterSchool21 Data Program Management
Welcome to ChildWare 2.0! Let s get started!
 1 Welcome to ChildWare 2.0! This first phase of the newly redeveloped ChildWare is designed to support child care providers that use ChildWare for the PHLpreK program. Logging into ChildWare 2.0 Let s
1 Welcome to ChildWare 2.0! This first phase of the newly redeveloped ChildWare is designed to support child care providers that use ChildWare for the PHLpreK program. Logging into ChildWare 2.0 Let s
Affinity Provider Portal Training Manual
 Training Manual Provider Homepage The portal enables users to easily access information on Affinity members across all lines of business. The improved design, enhanced security, registration and navigation
Training Manual Provider Homepage The portal enables users to easily access information on Affinity members across all lines of business. The improved design, enhanced security, registration and navigation
Instructional Improvement System (IIS) Dashboard District User Guide Statewide Longitudinal Data System (SLDS)
 Instructional Improvement System (IIS) Dashboard District User Guide Statewide Longitudinal Data System (SLDS) June 10, 2014 Page 1 of 36 IIS Dashboard District User Guide 2 Contents Project Overview...
Instructional Improvement System (IIS) Dashboard District User Guide Statewide Longitudinal Data System (SLDS) June 10, 2014 Page 1 of 36 IIS Dashboard District User Guide 2 Contents Project Overview...
Important items to note before you get started:
 Engineering econnection Students Help http://www.egr.uh.edu/careert The Table of Contents below is hyperlinked to the section described. Important items to note before you get started: Navigate the Homepage
Engineering econnection Students Help http://www.egr.uh.edu/careert The Table of Contents below is hyperlinked to the section described. Important items to note before you get started: Navigate the Homepage
VTC FY19 CO-OP GOOGLE QUALIFICATIONS PARAMETERS & REIMBURSEMENT DOCUMENTATION HOW-TO
 VTC FY19 CO-OP GOOGLE QUALIFICATIONS PARAMETERS & REIMBURSEMENT DOCUMENTATION HOW-TO 1 TABLE OF CONTENTS 01 CO-OP QUALIFICATIONS 02 REQUIRED DOCUMENTATION 03 REPORTING HOW-TO 04 REIMBURSEMENT PROCESS 2
VTC FY19 CO-OP GOOGLE QUALIFICATIONS PARAMETERS & REIMBURSEMENT DOCUMENTATION HOW-TO 1 TABLE OF CONTENTS 01 CO-OP QUALIFICATIONS 02 REQUIRED DOCUMENTATION 03 REPORTING HOW-TO 04 REIMBURSEMENT PROCESS 2
PIVOT. Pivot: Represents over 40,000 private, federal, and international funding opportunities Edited daily by group of editors
 1 PIVOT Pivot: Is a multi-disciplinary searchable funding opportunities database Matches faculty with funding opportunities Helps identify potential collaborators Represents over 40,000 private, federal,
1 PIVOT Pivot: Is a multi-disciplinary searchable funding opportunities database Matches faculty with funding opportunities Helps identify potential collaborators Represents over 40,000 private, federal,
Topics in this guide include:
 WORKSHOP INSTRUCTOR GUIDE This resource provides instructions on how to access and manage rosters and sessions for workshops or events within the College or Marin ProLearning portal. For information on
WORKSHOP INSTRUCTOR GUIDE This resource provides instructions on how to access and manage rosters and sessions for workshops or events within the College or Marin ProLearning portal. For information on
Molina Agent User Guide
 Molina Agent User Guide REGISTERING and LOGGING IN Welcome to Sentinel Elite! Sentinel is accessible at www.sentinelelite.com. If you are new to Sentinel please register as a new agent. Once selected,
Molina Agent User Guide REGISTERING and LOGGING IN Welcome to Sentinel Elite! Sentinel is accessible at www.sentinelelite.com. If you are new to Sentinel please register as a new agent. Once selected,
Windows Client Installation and User Guide. TRIAD v4 Clinical Trials
 Windows Client Installation and User Guide TRIAD v4 Clinical Trials Hardware & Software Requirements Hardware requirements (recommended): 4 GB RAM 2.4 GHz Processor 60 GB Hard Disk space Software requirements:
Windows Client Installation and User Guide TRIAD v4 Clinical Trials Hardware & Software Requirements Hardware requirements (recommended): 4 GB RAM 2.4 GHz Processor 60 GB Hard Disk space Software requirements:
eirb Training Investigators/Research Coordinators Topics Covered:
 eirb Training Investigators/Research Coordinators Topics Covered: 1. Home Page 2. My IRB Protocols 3. Completing New Protocols 4. Amendments & Continuing Reviews 5. Submitting a Protocol 6. Certifying
eirb Training Investigators/Research Coordinators Topics Covered: 1. Home Page 2. My IRB Protocols 3. Completing New Protocols 4. Amendments & Continuing Reviews 5. Submitting a Protocol 6. Certifying
Clinical Research Professionals Educa3on Session Barb Greguson Alliance Sta3s3cal and Data Center
 Clinical Research Professionals Educa3on Session Barb Greguson Alliance Sta3s3cal and Data Center greguson.barbara@mayo.edu Alliance for Clinical Trials in Oncology Spring 2015 Group Mee3ng Presenta3on
Clinical Research Professionals Educa3on Session Barb Greguson Alliance Sta3s3cal and Data Center greguson.barbara@mayo.edu Alliance for Clinical Trials in Oncology Spring 2015 Group Mee3ng Presenta3on
Creating a grant submission record in Themis
 Creating a grant submission record in Themis Creating a submission record for a grant requires completion of a number of screens. You will be able to monitor your progress on the progress train that displays
Creating a grant submission record in Themis Creating a submission record for a grant requires completion of a number of screens. You will be able to monitor your progress on the progress train that displays
Online Services for Employers User Guide. Mayo Clinic Health Solutions
 Online Services for Employers User Guide Mayo Clinic Health Solutions Table of Contents 1.0 Registering and Signing In... 1 Registration... 1 User Definitions... 1 Signing In... 1 2.0 Enrollment... 3 3.0
Online Services for Employers User Guide Mayo Clinic Health Solutions Table of Contents 1.0 Registering and Signing In... 1 Registration... 1 User Definitions... 1 Signing In... 1 2.0 Enrollment... 3 3.0
Routing a BennyBuy Access Form Using DocuSign
 Routing a BennyBuy Access Form Using DocuSign Become a DocuSign sender If you have not already done so, review the overview, signing and sending videos on the Resources/Senders page of the OSU DocuSign
Routing a BennyBuy Access Form Using DocuSign Become a DocuSign sender If you have not already done so, review the overview, signing and sending videos on the Resources/Senders page of the OSU DocuSign
Instructions for applying online for CAS
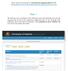 Please visit the following link for Instructions for applying online for CAS http://kashmiruniversity.net/recruitment/files/otherlist/instructionsforcas.pdf Step -1 The following screen will display all
Please visit the following link for Instructions for applying online for CAS http://kashmiruniversity.net/recruitment/files/otherlist/instructionsforcas.pdf Step -1 The following screen will display all
LabVantage User Manual: Registering Subjects Enrolling Subjects on an IRB Protocol Documenting Consent
 LabVantage User Manual: Registering Subjects Enrolling Subjects on an IRB Protocol Documenting Consent Disclaimer: Printed copies are not the official document. Please see the online PDF file for the most
LabVantage User Manual: Registering Subjects Enrolling Subjects on an IRB Protocol Documenting Consent Disclaimer: Printed copies are not the official document. Please see the online PDF file for the most
Introduction to Cognos Participants Guide. Table of Contents: Guided Instruction Overview of Welcome Screen 2
 IBM Cognos Analytics Welcome to Introduction to Cognos! Today s objectives include: Gain a Basic Understanding of Cognos View a Report Modify a Report View a Dashboard Request Access to Cognos Table of
IBM Cognos Analytics Welcome to Introduction to Cognos! Today s objectives include: Gain a Basic Understanding of Cognos View a Report Modify a Report View a Dashboard Request Access to Cognos Table of
OnCore Enterprise Research. Exercises: Subject Administration
 OnCore Enterprise Research Exercises: Subject Administration Clinical Research Coordinator August 2018 P a g e 1 This page is intentionally blank. P a g e 2 Exercises: Subject Administration Contents OnCore
OnCore Enterprise Research Exercises: Subject Administration Clinical Research Coordinator August 2018 P a g e 1 This page is intentionally blank. P a g e 2 Exercises: Subject Administration Contents OnCore
Introduction to. Sponsored by the Pediatric Research Office (PRO)
 Introduction to Sponsored by the Pediatric Research Office (PRO) Agenda Overview of REDCap Basic project work flow Creating a project with REDCap Interactive demonstration Questions and Answers Overview
Introduction to Sponsored by the Pediatric Research Office (PRO) Agenda Overview of REDCap Basic project work flow Creating a project with REDCap Interactive demonstration Questions and Answers Overview
eprmc User Manual For Investigators and Research Staff
 eprmc User Manual For Investigators and Research Staff Table of Contents How to Get an Account... 3 Logging In... 3 Overview of the eprmc Database... 4 Searching for a Protocol... 5 Adding a New Protocol...
eprmc User Manual For Investigators and Research Staff Table of Contents How to Get an Account... 3 Logging In... 3 Overview of the eprmc Database... 4 Searching for a Protocol... 5 Adding a New Protocol...
NRG Oncology and VisionTree Optimal Care (VTOC) Frequently Asked Questions
 NRG Oncology and VisionTree Optimal Care (VTOC) Frequently Asked Questions Overview VisionTree Optimal Care (VTOC) v4.1 is a secure, encrypted cloud-based platform to collect/report patient reported outcomes
NRG Oncology and VisionTree Optimal Care (VTOC) Frequently Asked Questions Overview VisionTree Optimal Care (VTOC) v4.1 is a secure, encrypted cloud-based platform to collect/report patient reported outcomes
Windows Client Installation and User Guide. TRIAD v4 Clinical Trials (Clinical Trials, Clinical Trials (NCI Oncology) and IDEAS)
 Windows Client Installation and User Guide TRIAD v4 Clinical Trials (Clinical Trials, Clinical Trials (NCI Oncology) and IDEAS) Version January 22, 2016 Table of Contents: Slide 5: Hardware & Software
Windows Client Installation and User Guide TRIAD v4 Clinical Trials (Clinical Trials, Clinical Trials (NCI Oncology) and IDEAS) Version January 22, 2016 Table of Contents: Slide 5: Hardware & Software
CTMS. Quick Reference Card. Using the CTMS Dashboard. Accessing the CTMS Dashboard. Viewing All Metrics
 Overview The Dashboard reports key metrics about DCRI protocols. This Quick Reference Card (QRC) summarizes Dashboard operations for Project Leaders. All protocol names and data in this QRC are fictitious.
Overview The Dashboard reports key metrics about DCRI protocols. This Quick Reference Card (QRC) summarizes Dashboard operations for Project Leaders. All protocol names and data in this QRC are fictitious.
TechDirect User's Guide for ProSupport Plus Reporting
 TechDirect User's Guide for ProSupport Plus Reporting Notes, cautions, and warnings NOTE: A NOTE indicates important information that helps you make better use of your product. CAUTION: A CAUTION indicates
TechDirect User's Guide for ProSupport Plus Reporting Notes, cautions, and warnings NOTE: A NOTE indicates important information that helps you make better use of your product. CAUTION: A CAUTION indicates
Sanofi Investigator Sponsored Studies (ISS) External Reference Guide. 1 November 2017
 Sanofi Investigator Sponsored Studies (ISS) External Reference Guide 1 November 2017 Sanofi ISS Overview Sanofi is committed to supporting medically and scientifically sound research aimed at the advancement
Sanofi Investigator Sponsored Studies (ISS) External Reference Guide 1 November 2017 Sanofi ISS Overview Sanofi is committed to supporting medically and scientifically sound research aimed at the advancement
The Basics. As of December 12, 2016
 The Basics As of December 12, 2016 Accessing REDCap 1. To access REDCap, enter the URL into your internet browser: https://redcap.wakehealth.edu/ 2. Login using your Medical Center ID and password 3. FAQ
The Basics As of December 12, 2016 Accessing REDCap 1. To access REDCap, enter the URL into your internet browser: https://redcap.wakehealth.edu/ 2. Login using your Medical Center ID and password 3. FAQ
OnCore Enterprise Research. Subject Administration Full Study
 OnCore Enterprise Research Subject Administration Full Study Principal Investigator Clinical Research Coordinator June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Table of Contents What
OnCore Enterprise Research Subject Administration Full Study Principal Investigator Clinical Research Coordinator June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Table of Contents What
Children s Surgery Verification Program - Center Portal Instructions
 Children s Surgery Verification Program - Center Portal Instructions Thank you for your interest in joining the Children s Surgery Verification Quality Improvement Program. This portal guide is designed
Children s Surgery Verification Program - Center Portal Instructions Thank you for your interest in joining the Children s Surgery Verification Quality Improvement Program. This portal guide is designed
GOG-0274-RTOG 1174 UPDATE TO HANDBOOK. GOG and RTOG Legacy Group Edition Version 3.0, July 7, 2014 # Section Page(s) Change
 GOG-0274-RTOG 1174 UPDATE TO HANDBOOK GOG and RTOG Legacy Group Edition Version 3.0, July 7, 2014 # Section Page(s) Change 1 Title page i In the footer US Edition has been replaced with GOG and RTOG Legacy
GOG-0274-RTOG 1174 UPDATE TO HANDBOOK GOG and RTOG Legacy Group Edition Version 3.0, July 7, 2014 # Section Page(s) Change 1 Title page i In the footer US Edition has been replaced with GOG and RTOG Legacy
User Guide for the New Kofax Partner Portal
 User Guide for the New Kofax Partner Portal The purpose of this document is to better acquaint you with the basic features of the new Kofax Partner Portal being released in August 2012. Please note that
User Guide for the New Kofax Partner Portal The purpose of this document is to better acquaint you with the basic features of the new Kofax Partner Portal being released in August 2012. Please note that
OnCore Enterprise Research. Exercises: Subject Administration
 OnCore Enterprise Research Exercises: Subject Administration Clinical Research Coordinator June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Exercises: Subject Administration Contents OnCore
OnCore Enterprise Research Exercises: Subject Administration Clinical Research Coordinator June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Exercises: Subject Administration Contents OnCore
Online Registration FAQs
 Online Registration FAQs o How do we register individuals from the back end? (Over the phone) o How do we send emails to individuals on the roster? o How do we view and print a roster of individuals attending
Online Registration FAQs o How do we register individuals from the back end? (Over the phone) o How do we send emails to individuals on the roster? o How do we view and print a roster of individuals attending
zipform Plus: Exploring the Agent Dashboard
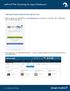 Exploring the Agent Dashboard inside zipform Plus! 1. Begin by signing in to zipform Plus at www.zipformplus.com, or through your Association, MLS, or Brokerage site, if you have single sign on. 2. Once
Exploring the Agent Dashboard inside zipform Plus! 1. Begin by signing in to zipform Plus at www.zipformplus.com, or through your Association, MLS, or Brokerage site, if you have single sign on. 2. Once
Research Participant Registration System (RPRS) Study Creation Guide
 Research Participant Registration System (RPRS) Study Creation Guide Revised August 2016 By Robin Richardson Contents Introduction to RPRS... 3 Creating a Study... 3 1. Details... 4 1.1. Basic Details...
Research Participant Registration System (RPRS) Study Creation Guide Revised August 2016 By Robin Richardson Contents Introduction to RPRS... 3 Creating a Study... 3 1. Details... 4 1.1. Basic Details...
The Volunteer Space Sign-Up Sheet User Manual
 The Volunteer Space Sign-Up Sheet User Manual This document is the property of Web Concepts, Inc. and the use of it other than as a guide to TheVolunteerSpace.com web site is strictly prohibited unless
The Volunteer Space Sign-Up Sheet User Manual This document is the property of Web Concepts, Inc. and the use of it other than as a guide to TheVolunteerSpace.com web site is strictly prohibited unless
13241 Woodland Park Road, Suite 400 Herndon, VA USA A U T H O R : E X O S T A R D ATE: M A R C H V E R S I O N : 3.
 SECURE ACCESS MAN AG E R FIRST TIME LOGIN GUIDE A U T H O R : E X O S T A R D ATE: M A R C H 2 0 1 5 V E R S I O N : 3.0 1 S E C U R E A CCESS M A N A G E R SECURE ACCESS MANAGER OVERVIEW... 3 SUMMARY...
SECURE ACCESS MAN AG E R FIRST TIME LOGIN GUIDE A U T H O R : E X O S T A R D ATE: M A R C H 2 0 1 5 V E R S I O N : 3.0 1 S E C U R E A CCESS M A N A G E R SECURE ACCESS MANAGER OVERVIEW... 3 SUMMARY...
Test Information and Distribution Engine
 SC-Alt Test Information and Distribution Engine User Guide 2018 2019 Published January 14, 2019 Prepared by the American Institutes for Research Descriptions of the operation of the Test Information Distribution
SC-Alt Test Information and Distribution Engine User Guide 2018 2019 Published January 14, 2019 Prepared by the American Institutes for Research Descriptions of the operation of the Test Information Distribution
COEUS IRB MODULE Written & Designed by: Maria Martinez Weill Medical College of Cornell
 COEUS IRB MODULE Table of Contents Short Cut Identification Key 3 Searching How To s 6 How to Run Reports 11 Protocol Screen Description 13 How to Enter a New Protocol 18 How to Submit a Protocol for Review
COEUS IRB MODULE Table of Contents Short Cut Identification Key 3 Searching How To s 6 How to Run Reports 11 Protocol Screen Description 13 How to Enter a New Protocol 18 How to Submit a Protocol for Review
Sophos Central Enterprise
 Contents 1 About Sophos Central Enterprise...3 2 Dashboard...4 3 Alerts...5 4 Sub-Estates...6 5 Licenses...8 6 Trials...9 7 Manage Administrators...10 8 Explore Products...11 9 Global Security News...12
Contents 1 About Sophos Central Enterprise...3 2 Dashboard...4 3 Alerts...5 4 Sub-Estates...6 5 Licenses...8 6 Trials...9 7 Manage Administrators...10 8 Explore Products...11 9 Global Security News...12
Clinical Research Professionals Educa3on Session Barb Greguson Alliance Sta3s3cal and Data Center
 Clinical Research Professionals Educa3on Session Barb Greguson Alliance Sta3s3cal and Data Center greguson.barbara@mayo.edu Alliance for Clinical Trials in Oncology Fall 2015 Group Mee3ng Presenta3on Objec3ves
Clinical Research Professionals Educa3on Session Barb Greguson Alliance Sta3s3cal and Data Center greguson.barbara@mayo.edu Alliance for Clinical Trials in Oncology Fall 2015 Group Mee3ng Presenta3on Objec3ves
HOW TO ACCESS ROTARY CLUB CENTRAL
 HOW TO ACCESS ROTARY CLUB CENTRAL 1 Go to My Rotary and select Sign In or Register. Or go to rotary.org/clubcentral to reach the site directly. You ll be prompted to sign in to My Rotary or create an account
HOW TO ACCESS ROTARY CLUB CENTRAL 1 Go to My Rotary and select Sign In or Register. Or go to rotary.org/clubcentral to reach the site directly. You ll be prompted to sign in to My Rotary or create an account
HOW TO ACCESS ROTARY CLUB CENTRAL
 HOW TO ACCESS ROTARY CLUB CENTRAL 1 Go to My Rotary and select Sign In or Register. Or go to rotary.org/clubcentral to reach the site directly. You ll be prompted to sign in to My Rotary or create an account
HOW TO ACCESS ROTARY CLUB CENTRAL 1 Go to My Rotary and select Sign In or Register. Or go to rotary.org/clubcentral to reach the site directly. You ll be prompted to sign in to My Rotary or create an account
Advisor Case Load Self Service Participants Guide
 IBM Cognos Analytics Advisor Case Load Self Service Participants Guide Welcome to Advisor Case Load Self Service Training! Today s objectives include: Gain a Basic Understanding of Cognos Know the Available
IBM Cognos Analytics Advisor Case Load Self Service Participants Guide Welcome to Advisor Case Load Self Service Training! Today s objectives include: Gain a Basic Understanding of Cognos Know the Available
Release Highlights
 Release Highlights WWW.DFNETRESEARCH.COM 2 DFdiscover 2018 Highlights 1. Look & Feel 2. ecrfs 3. Site Definitions 4. Reporting 5. Metadata Editor Panel 6. Export to Excel 7. Batch Edits View 8. Edit Coding
Release Highlights WWW.DFNETRESEARCH.COM 2 DFdiscover 2018 Highlights 1. Look & Feel 2. ecrfs 3. Site Definitions 4. Reporting 5. Metadata Editor Panel 6. Export to Excel 7. Batch Edits View 8. Edit Coding
DBtC
 (DBtC) The DBtC application was launched to help Transplant Centers retrieve the data they have submitted to the through FormsNet TM. DBtC currently provides data from the submission of data on all TED
(DBtC) The DBtC application was launched to help Transplant Centers retrieve the data they have submitted to the through FormsNet TM. DBtC currently provides data from the submission of data on all TED
User Manual. phr.mtbc.com
 User Manual Table of Contents Introduction Appointments Appointment History Claims History CCDA Report Demographics Health History Lab Reports Online Payment Secure Messages Health Recommendation Patient
User Manual Table of Contents Introduction Appointments Appointment History Claims History CCDA Report Demographics Health History Lab Reports Online Payment Secure Messages Health Recommendation Patient
INFocus Basic Table Reporting
 INFocus Basic Table Reporting About This Document This document provides basic information about the INFocus tool, with specific examples of steps required to create reports. The user should log in to
INFocus Basic Table Reporting About This Document This document provides basic information about the INFocus tool, with specific examples of steps required to create reports. The user should log in to
OnCore Enterprise Research. Basics: Components and Navigation
 OnCore Enterprise Research Basics: Components and Navigation All Access Roles August 2018 P a g e 1 This page is intentionally blank. P a g e 2 Table of Contents What is the OnCore Enterprise Research
OnCore Enterprise Research Basics: Components and Navigation All Access Roles August 2018 P a g e 1 This page is intentionally blank. P a g e 2 Table of Contents What is the OnCore Enterprise Research
myvirena.com User Manual / Training Guide
 myvirena.com User Manual / Training Guide Registering as a New User... 3 User Roles... 5 Healthcare Provider Roles... 5 Organizational Administrator... 5 Authorized User... 5 Public Health Roles... 5 Country...
myvirena.com User Manual / Training Guide Registering as a New User... 3 User Roles... 5 Healthcare Provider Roles... 5 Organizational Administrator... 5 Authorized User... 5 Public Health Roles... 5 Country...
FREQUNETLY ASKED QUESTIONS: REQUESTS FOR MEDICAL EDUCATION GRANTS
 FREQUNETLY ASKED QUESTIONS: REQUESTS FOR MEDICAL EDUCATION GRANTS Guidelines for Requesting Medical Education Grants VIVUS competitively reviews all requests to align resources with the highest quality
FREQUNETLY ASKED QUESTIONS: REQUESTS FOR MEDICAL EDUCATION GRANTS Guidelines for Requesting Medical Education Grants VIVUS competitively reviews all requests to align resources with the highest quality
Instructions for the Hearst Foundations Online Grant Application
 Instructions for the Hearst Foundations Online Grant Application NOTE: All applicants should read this document thoroughly BEFORE starting the application process or seeking assistance from the Hearst
Instructions for the Hearst Foundations Online Grant Application NOTE: All applicants should read this document thoroughly BEFORE starting the application process or seeking assistance from the Hearst
Professional Development
 Contents Profile Creation... 2 Forgot My Password?... 4 Forgot My Email?... 5 Dashboards... 6 Transcript & Content... 7 Workshop Search... 7 Registration... 8 Workshop Creation... 8 Global Reports... 12
Contents Profile Creation... 2 Forgot My Password?... 4 Forgot My Email?... 5 Dashboards... 6 Transcript & Content... 7 Workshop Search... 7 Registration... 8 Workshop Creation... 8 Global Reports... 12
