Automatic Segmentation of the Aortic Dissection Membrane from 3D CTA Images
|
|
|
- Junior Montgomery
- 6 years ago
- Views:
Transcription
1 Automatic Segmentation of the Aortic Dissection Membrane from 3D CTA Images Tamás Kovács 1, Philippe Cattin 1, Hatem Alkadhi 2, Simon Wildermuth 3, and Gábor Székely 1 1 Computer Vision Group, ETH Zurich, 8092 Zurich, Switzerland 2 Institute of Diagnostic Radiology, University Hospital, 8092 Zürich 3 Institut fuer Radiologie, Kantonsspital St. Gallen, 9000 St. Gallen, Switzerland {kovacs,cattin,szekely}@vision.ee.ethz.ch, hatem.alkadhi@usz.ch, simon.wildermuth@kssg.ch Abstract. Acute aortic dissection is a life-threatening condition and must be diagnosed and treated promptly. For treatment planning the reliable identification of the true and false lumen is crucial. However, a fully automatic Computer Aided Diagnosis system capable to display the different lumens in an easily comprehensible and timely manner is still not available. In this paper we present a method that segments the entire aorta and then identifies the two lumens separated by the dissection membrane. The algorithm misdetected part of the membrane in only one of the 15 cases tested, where the aorta has not been significantly altered by the presence of aneurisms. 1 Introduction Acute aortic dissection is a life-threatening condition and must be diagnosed and treated promptly. Exact knowledge of the lumen and dissection membrane topology is vital for treatment planning. This is unfortunately a difficult task, even for trained professionals. A fully automatic Computer Aided Diagnosis (CAD) system capable to display the different lumens in an easily comprehensible way is still not available. It is thus the aim of this research to provide the radiologist generic tools to support diagnosis and treatment planning. In order to implement such a CAD system, firstly an accurate segmentation of the entire aorta including the aortic arch as well as the ascending- and descending sections must be performed. Secondly the dissection membrane must be detected in order to identify the true and false lumen. Different solutions for the aorta segmentation can be found in literature. These include vessel axis extraction and border estimation, neural networks, region growing or watershed- and level set-based approaches. An extensive survey of these approaches is given in [1]. All of these methods have serious limitations like segmenting just a well defined part of the aorta or requiring manual initialization, sometimes even user intervention. Moreover, these methods are not robust against the disturbing artifacts in the image data like the inhomogeneous
2 density of the contrast agent caused by flow variations in the lumens, reconstruction errors due to high contrast agent density [2] and last but not least they are not able to deal with the dissection membrane within the aortic lumen. Behrens described in [3] a Hough transform based algorithm combined with a Kalman filter. The drawback of this approach was, however, the need for three user-selected parameters namely, the starting point of the aorta, the aorta radius, and an approximate axis direction. In [4] we presented a fully automatic segmentation method of the aorta using a Hough transformation based initialization of a deformable surface model. In comparison to Behrens approach, our method incorporates more a priori anatomical knowledge about the shape of the aortic arch and therefore needs no manual initialization. In this paper we focus on the detection of the aortic dissection membrane, which is the final step towards a fully automatic CAD system. As the membrane is a flat elongated structure, sheet detection methods can be used for its localization. Only very few sheet detection methods have been reported in the literature and almost all of them are based on the eigenvalue analysis of the Hessian matrix [5 7]. Descoteaux proposed in [8] a method that combines the eigenvalues of the Hessian matrix in one single measure. A completely different multiscale line detector based on steerable filters was proposed by Koller in [9]. The approach can be extended to 3D for sheet detection in a straightforward manner. While the resulting non-linear filter is strictly selective to sheet-like structures and does not respond to edges, our experiments showed that Koller s method is computationally less efficient than Hessian matrix based methods. In the following section we describe the newly developed method for the automatic identification of the true and false aortic lumens. Section 3 presents the results of our approach on 19 clinical cases and their validation against manual segmentation. The paper is then concluded by the discussion of the achievements and ongoing and future work. 2 Method For completeness we first summarize the aorta segmentation method we use [4]. The method is based on the observation that on the one hand the aorta lumen has an approximately circular cross section and on the other hand the aortic arch forms a 180 -sector of a torus. In the first step an initial slice superior to the heart has to be found, on which two seed points for the ascending and the descending segment can be marked. In order to obtain this initial slice the region of the heart is detected by calculating the average intensity of all axial slices. Starting from the slice with the highest average intensity, 20 axial slices are taken with an inter slice distance of 1 cm. On each of these slices the two circles corresponding to the strongest Hough-peaks are selected, as these are mostly generated by the ascending and descending aorta. These Hough-peaks are then projected to an axial plane. The set of the points on this plane is then spatially clustered with the K-mean algorithm. The centers of the two largest clusters are taken as the centers of the ascending and
3 descending aorta and the slice with these two clusters closest to the heart is taken as the initial slice as illustrated by Fig. 1(a)/1. The relative position of these points is then used as a first rough estimate for the position of the aortic arch (Fig. 1(a)/2). This approximation is then refined by using an additional Hough transformation on a reformatted slice perpendicular to the line connecting the two circle centers (Fig. 1(a)/3,4). The exact location of the aortic arch is then found by Hough transformations on reformatted slices perpendicular to this first centerline estimate as shown in. An initial mesh of the lumen is generated using the centers (Fig. 1(b)/5) and the radii of the detected circles. The lumen is finally identified by elastically deforming the initial mesh to match the edges corresponding to the vessel wall. (a) (b) Fig. 1. (a) The detection of the three initial circles to estimate the parameters of the aortic arch (b) the refinement of the aortic arch and the descending aorta with additional circles. The steps described in the text are highlighted with numbers Once the aorta lumen is segmented, it is used as a region of interest (ROI) for the detection of the dissection membrane. To further ease the membrane detection, the aorta is straightened along its centerline. The resulting stack consist of reformatted slices, locally perpendicular to the vessel. In order to localize the membrane we use Descoteaux s sheetness measure [8], which is making simultaneous use of the three Hessian eigenvalues in a comprehensive way. Based on the ellipsoid model described in [7], this detector assigns a score S(p, σ) [0, 1] to each voxel p at scale σ which can be interpreted as the probability to be part of a sheet-like structure, S(p) = max σ Ω S(σ) = 0 if λ 3 < 0 «[ «] [ «] R sheet 2 R 2 2α e 2 blob R 2β 1 e 2 noise 2 2c 1 e 2 else (1)
4 where R sheet = λ 2 λ 3 (2) R blob = (2 λ 3 λ 2 λ 1 ) (3) λ 3 R noise = 3 λ 2 j (4) j=1 with λ 1 λ 2 λ 3 the eigenvalues of the Hessian matrix computed at point p with scale σ. Ω is a finite set of scales covering the range between the smallest and the largest expected membrane thickness. For our experiments we have chosen three different scales Ω={1 mm, 1.5 mm, 2 mm}. The parameters α, β and c control the sensitivity of the sheet detector and were set to α = 0.5, β = 0.5, for c half the value of the maximum Hessian norm has been selected. To avoid the big computational overhead required for the calculation of c Felkel et al. proposed in [10] to use 0.25σ 2 I max instead of 2c 2 for the normalization of the third exponent. According to our experience this results in faster but less accurate membrane detection, therefore we decided not to use this alternative for the calculation of the sheetness measure. As the approach described above is quite sensitive to image noise, the ROI is pre-processed with a curvature flow filter [11] to increase the robustness of the membrane detection. Several factors influence the detection of the membrane, leading often to the generation of false positive or false negative candidates. On the one hand, image noise and inhomogeneous contrast agent distribution can create fake membrane structures. As these are only visible in one or a few slices, they can be efficiently filtered out using 3D connected component labeling. In fact, in all our experiments the largest connected component was always the dissection membrane itself. On the other hand, reformatting blur, partial volume effects, thin membranes, and previous surgical interventions can lead to gaps in the detected dissection membrane. At least one tear in the dissection membrane, where the blood enters the inner lining of the aorta (intima) and thus creates the false lumen, has to be present near to the proximal side of the dissection. Often more gaps and tears appear due to the aforementioned effects. A robust method has therefore to be found to replace the missing parts of the membrane. Since the above described sheetness measure is not able to detect the membrane close to other structures (see Fig. 2(a)), extrapolation is necessary to connect the membrane with the aortic wall. For this, the centerline of the membrane is generated on every slice i using binary thinning. If the centerline is not connected on the slice, it is skipped and the connection points with the wall (wallpoints) are interpolated at a later stage from adjacent slices. Otherwise the two endpoints (P i, P i ) are linearly extrapolated towards the aortic wall. To incorporate the orientation of the membrane into the extrapolation, the directions of continuation v i,0 and v i,0 are first set according to the estimated tangent of the two endpoints, see Fig. 2(a). By pivoting v i,0 around P i with an angle of
5 (a) (b) Fig. 2. (a) Extrapolation of the membrane to the aortic wall as a straight continuation. (b) The determination of the real connection point used to initialize the sheet mesh ω = k 5 k Z ω 30 several wallpoint candidates {W i,30, W i,25... W i,ω... W i, 30 } are generated. The wallpoint candidate with the lowest average intensity along the line segment connecting to P i is then finally chosen. v i,0 is processed in an identical fashion. Once the wallpoints for all slices are calculated, the initial mesh of the membrane can be generated using the wallpoints as constraining nodes. On each slice the connecting line between the two wallpoints is split in equidistant points. The mesh is generated by triangulating between these points of adjacent slices. It is then iteratively fitted to the dissection membrane using the mass-spring approach presented by Brown in [12]. The segmentation process follows the procedure described by [13]. According to Zsemlye, mass points are associated with each node and springs with each edge of the mesh. The nodes are under the influence of internal and external forces. The internal forces keep the surface smooth, whereas the external forces deform the mesh in order to match the underlying CT dataset. In order to obtain accurate membrane segmentation, we used the Gradient Vector Flow (GVF) proposed by Xu et al. [14] as external force. 3 Results The proposed method for membrane detection was tested on 19 patients with an abdominal aortic dissection. Of these 19 patients 4 had in addition to the dissection an aortic aneurism. The CT datasets typically consisted of slices of pixels using 16-bit quantization, with a pixel size of mm, and an effective slice thickness of 2 mm. The method was successful on 15 cases, but failed for the 4 patients with the aneurism. The reason was that the aorta segmentation method can not yet handle the varying lumen diameter through the aneurisms. The membrane detection could therefore not be tested, as the aorta lumen has to be known
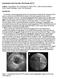
6 beforehand to restrict the ROI. Additionally, the proposed approach could not properly segment the dissection membrane in one case, where over a range of 12 slices three lumens are visible, see Fig. 4(a). This type of split or highly folded membranes can of course not be modeled with our sheet mesh. In spite of the sometimes bad signal-to-noise ratio, the detection process was successful in all other cases, as demonstrated by some examples in Fig. 3. (a) (b) (c) Fig. 3. (a) Accurate segmentation of the lumens in the aortic arch displayed on an axial slice (b) membrane detection in the ascending as well as in the descending aorta (c) 3D rendering of the true and false lumen To obtain a quantitative assessment, the aorta and the membrane in every dataset were manually segmented by an expert observer. As the dissection membrane is generally a several voxels wide thick structure, manual segmentation is highly subjective and observer dependent. To avoid this ambiguity the expert was instructed to place the borderline between the lumens in the middle of the membrane wherever possible. The mesh of the segmented true and false lumens were then taken as the ground truth and compared to the results of the 15 successful automatic segmentations. To quantify the performance, we calculated the average distance of the two meshes. Our method resulted in an overall average error of 1.71 ± 1.89 mm. The best segmentation had an average distance of 1.13 ± 0.63 mm and the worst one of 1.92 ± 2.00 mm. Several problems limit the performance of the membrane detection. Firstly, the exact position of the membrane close to the aortic wall is ambiguous, see Fig. 4(b). The membranes generally widen near the wall and it is therefore not possible to determine its exact location. Fig. 4(c) depicts the manual delineation of the membrane on the same slice. It can be seen that the observer had exact knowledge about the true lumen and this was incorporated into the segmentation process, as the membrane usually curves towards the true lumen. Secondly, in areas where the membrane has no visible attachment to the wall as in Fig. 4(d) the extrapolation method often leads to unintuitive results. A smoothing along
7 the wallpoints should reduce the deviations from the wallpoints a human expert would have selected. Thirdly, the membrane can not yet be accurately detected close to calcified plaques as can be seen in Fig. 4(e). In the worst case a membrane is recognized but the plaque being assigned to the wrong lumen as shown in Fig. 4(f). As plaques can only appear in the true lumen such membrane misdetections can lead to wrong identification and consequently lead to false treatment plans. (a) (b) (c) (d) (e) (f) Fig. 4. (a) Special case with three visible lumens and the result of membrane segmentation in black (b) Comparison of the automatic detection and (c) the manual segmentation of the membrane. (d) Unintuitive membrane extrapolation. (e,f) Erroneous membrane detections due to the presence of calcified plaques. Manual segmentation of the aorta and the membrane required min per dataset. In contrast, the processing time was less than 6 min for the fully automatic aorta segmentation and 7 9 min for the membrane detection procedure using a standard 2.4 GHz PC. 4 Conclusions In this paper we presented a fully automatic method for the detection of the membrane in abdominal aortic dissections. The detection method is based on eigenvalue analysis of the Hessian matrix and a related sheetness measure. To close the inevitable gaps in the segmented dissection membrane, an elastically
8 deformable 3D sheet model is used. The method has been evaluated on angiographic CT images and the results indicate that the proposed method has a high reproducibility, clinically acceptable accuracy and is faster than manual segmentation. Our future work will concentrate on the identification of the true and false lumen based on indicators such as (1) calcifications (should be only in the true lumen), (2) the angle between the aortic wall and the membrane and (3) the different densities of contrast agent caused by flow variations in the two lumens. A larger evaluation study with manually segmented lumens from multiple experts is planned to asses the reliability of the proposed aortic dissection analysis tool. Acknowledgments The authors thank Dr. Tobias Boskamp and Olaf Konrad-Verse from MeVis (Center for Medical Diagnostic Systems and Visualization) Bremen, Germany for providing and supporting the MeVisLab working environment. This work has been supported by the CO-ME/NCCR research network of the Swiss National Science Foundation ( References 1. B.H.Sollie: Automatic segmentation and registration of CT and US images of abdominal aortic aneurysm using ITK (2002) Masterthesis: Norwegian University of Science and Technology, Norway. 2. R.Baissalov, G.A.Sandison, B.J.Donnelly, et al.: Suppression of high-density artefacts in x-ray CT images using temporal digital subtraction with application to cryotherapy. Phys Med Biol 45 (2000) T.Behrens, K.Rohr, H.S.Stiehl: Robust segmentation of tubular structures in 3-D medical images by parametric object detection and traking. IEEE Trans Syst Man Cybern B 33 (2003) Y.Sato, C.Westin, A.Bhalerao, et al.: Tissue classification based on 3d local intensity structures for volume rendering. IEEE Trans Vis Comput Graph 6 (2000) K.Krissian, G.Malandain, N.Ayache, et al.: Model-based detection of tubular structures in 3d images. Comput Vis Image Underst 80 (2000) A.F.Frangi, W.J.Niessen, K.L.Vincken, et al.: Multiscale vessel enhancement filtering. Lecture Notes in Comput Science 1496 (1998) M.Descoteaux, M.Audette, K.Chinzei, el al.: Bone enhancement filtering: Application to sinus bone segmentation and simulation of pituitary surgery. In: MICCAI. (2005) T.Koller, G.Gerig, G.Szekely, et al.: Multiscale detection of curvilinear structures in 2d and 3d image data. In: ICCV. (1995) P.Felkel, R.Wegenkittl, A.Kanitsar: Vessel tracking in peripheral cta datasets an overview. In: SCCG 01. (2001) Sethian, J.: Level Set Methods and Fast Marching Methods. Cambridge Press (1999) Chapter 16.
9 12. J.Brown, K.Montgomery, J.C.Latombe, et al.: A microsurgery simulation system. Lecture Notes in Computer Science 2208 (2001) Zsemlye, G.: Shape Prediction from Partial Information. PhD thesis, Computer Vision Laboratory, ETH Zurich, Switzerland (2005) 14. C.Xu, J.Prince: Snakes, shapes, and gradient vector flow. (1998)
Automatic Ascending Aorta Detection in CTA Datasets
 Automatic Ascending Aorta Detection in CTA Datasets Stefan C. Saur 1, Caroline Kühnel 2, Tobias Boskamp 2, Gábor Székely 1, Philippe Cattin 1,3 1 Computer Vision Laboratory, ETH Zurich, 8092 Zurich, Switzerland
Automatic Ascending Aorta Detection in CTA Datasets Stefan C. Saur 1, Caroline Kühnel 2, Tobias Boskamp 2, Gábor Székely 1, Philippe Cattin 1,3 1 Computer Vision Laboratory, ETH Zurich, 8092 Zurich, Switzerland
Contrast Enhancement with Dual Energy CT for the Assessment of Atherosclerosis
 Contrast Enhancement with Dual Energy CT for the Assessment of Atherosclerosis Stefan C. Saur 1, Hatem Alkadhi 2, Luca Regazzoni 1, Simon Eugster 1, Gábor Székely 1, Philippe Cattin 1,3 1 Computer Vision
Contrast Enhancement with Dual Energy CT for the Assessment of Atherosclerosis Stefan C. Saur 1, Hatem Alkadhi 2, Luca Regazzoni 1, Simon Eugster 1, Gábor Székely 1, Philippe Cattin 1,3 1 Computer Vision
Modeling and preoperative planning for kidney surgery
 Modeling and preoperative planning for kidney surgery Refael Vivanti Computer Aided Surgery and Medical Image Processing Lab Hebrew University of Jerusalem, Israel Advisor: Prof. Leo Joskowicz Clinical
Modeling and preoperative planning for kidney surgery Refael Vivanti Computer Aided Surgery and Medical Image Processing Lab Hebrew University of Jerusalem, Israel Advisor: Prof. Leo Joskowicz Clinical
Comparison of Vessel Segmentations using STAPLE
 Comparison of Vessel Segmentations using STAPLE Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab The University of North Carolina at Chapel Hill, Department
Comparison of Vessel Segmentations using STAPLE Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab The University of North Carolina at Chapel Hill, Department
Comparison of Vessel Segmentations Using STAPLE
 Comparison of Vessel Segmentations Using STAPLE Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab, The University of North Carolina at Chapel Hill, Department
Comparison of Vessel Segmentations Using STAPLE Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab, The University of North Carolina at Chapel Hill, Department
Calculating the Distance Map for Binary Sampled Data
 MITSUBISHI ELECTRIC RESEARCH LABORATORIES http://www.merl.com Calculating the Distance Map for Binary Sampled Data Sarah F. Frisken Gibson TR99-6 December 999 Abstract High quality rendering and physics-based
MITSUBISHI ELECTRIC RESEARCH LABORATORIES http://www.merl.com Calculating the Distance Map for Binary Sampled Data Sarah F. Frisken Gibson TR99-6 December 999 Abstract High quality rendering and physics-based
2D Vessel Segmentation Using Local Adaptive Contrast Enhancement
 2D Vessel Segmentation Using Local Adaptive Contrast Enhancement Dominik Schuldhaus 1,2, Martin Spiegel 1,2,3,4, Thomas Redel 3, Maria Polyanskaya 1,3, Tobias Struffert 2, Joachim Hornegger 1,4, Arnd Doerfler
2D Vessel Segmentation Using Local Adaptive Contrast Enhancement Dominik Schuldhaus 1,2, Martin Spiegel 1,2,3,4, Thomas Redel 3, Maria Polyanskaya 1,3, Tobias Struffert 2, Joachim Hornegger 1,4, Arnd Doerfler
Automatic Vascular Tree Formation Using the Mahalanobis Distance
 Automatic Vascular Tree Formation Using the Mahalanobis Distance Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab, Department of Radiology The University
Automatic Vascular Tree Formation Using the Mahalanobis Distance Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab, Department of Radiology The University
FINDING THE TRUE EDGE IN CTA
 FINDING THE TRUE EDGE IN CTA by: John A. Rumberger, PhD, MD, FACC Your patient has chest pain. The Cardiac CT Angiography shows plaque in the LAD. You adjust the viewing window trying to evaluate the stenosis
FINDING THE TRUE EDGE IN CTA by: John A. Rumberger, PhD, MD, FACC Your patient has chest pain. The Cardiac CT Angiography shows plaque in the LAD. You adjust the viewing window trying to evaluate the stenosis
Human Heart Coronary Arteries Segmentation
 Human Heart Coronary Arteries Segmentation Qian Huang Wright State University, Computer Science Department Abstract The volume information extracted from computed tomography angiogram (CTA) datasets makes
Human Heart Coronary Arteries Segmentation Qian Huang Wright State University, Computer Science Department Abstract The volume information extracted from computed tomography angiogram (CTA) datasets makes
EE368 Project Report CD Cover Recognition Using Modified SIFT Algorithm
 EE368 Project Report CD Cover Recognition Using Modified SIFT Algorithm Group 1: Mina A. Makar Stanford University mamakar@stanford.edu Abstract In this report, we investigate the application of the Scale-Invariant
EE368 Project Report CD Cover Recognition Using Modified SIFT Algorithm Group 1: Mina A. Makar Stanford University mamakar@stanford.edu Abstract In this report, we investigate the application of the Scale-Invariant
8/3/2017. Contour Assessment for Quality Assurance and Data Mining. Objective. Outline. Tom Purdie, PhD, MCCPM
 Contour Assessment for Quality Assurance and Data Mining Tom Purdie, PhD, MCCPM Objective Understand the state-of-the-art in contour assessment for quality assurance including data mining-based techniques
Contour Assessment for Quality Assurance and Data Mining Tom Purdie, PhD, MCCPM Objective Understand the state-of-the-art in contour assessment for quality assurance including data mining-based techniques
Probabilistic Tracking and Model-based Segmentation of 3D Tubular Structures
 Probabilistic Tracking and Model-based Segmentation of 3D Tubular Structures Stefan Wörz, William J. Godinez, Karl Rohr University of Heidelberg, BIOQUANT, IPMB, and DKFZ Heidelberg, Dept. Bioinformatics
Probabilistic Tracking and Model-based Segmentation of 3D Tubular Structures Stefan Wörz, William J. Godinez, Karl Rohr University of Heidelberg, BIOQUANT, IPMB, and DKFZ Heidelberg, Dept. Bioinformatics
Interactive segmentation of vascular structures in CT images for liver surgery planning
 Interactive segmentation of vascular structures in CT images for liver surgery planning L. Wang¹, C. Hansen¹, S.Zidowitz¹, H. K. Hahn¹ ¹ Fraunhofer MEVIS, Institute for Medical Image Computing, Bremen,
Interactive segmentation of vascular structures in CT images for liver surgery planning L. Wang¹, C. Hansen¹, S.Zidowitz¹, H. K. Hahn¹ ¹ Fraunhofer MEVIS, Institute for Medical Image Computing, Bremen,
Automatic Cerebral Aneurysm Detection in Multimodal Angiographic Images
 Automatic Cerebral Aneurysm Detection in Multimodal Angiographic Images Clemens M. Hentschke, Oliver Beuing, Rosa Nickl and Klaus D. Tönnies Abstract We propose a system to automatically detect cerebral
Automatic Cerebral Aneurysm Detection in Multimodal Angiographic Images Clemens M. Hentschke, Oliver Beuing, Rosa Nickl and Klaus D. Tönnies Abstract We propose a system to automatically detect cerebral
The MAGIC-5 CAD for nodule detection in low dose and thin slice lung CT. Piergiorgio Cerello - INFN
 The MAGIC-5 CAD for nodule detection in low dose and thin slice lung CT Piergiorgio Cerello - INFN Frascati, 27/11/2009 Computer Assisted Detection (CAD) MAGIC-5 & Distributed Computing Infrastructure
The MAGIC-5 CAD for nodule detection in low dose and thin slice lung CT Piergiorgio Cerello - INFN Frascati, 27/11/2009 Computer Assisted Detection (CAD) MAGIC-5 & Distributed Computing Infrastructure
The VesselGlyph: Focus & Context Visualization in CT-Angiography
 The VesselGlyph: Focus & Context Visualization in CT-Angiography Matúš Straka M. Šrámek, A. La Cruz E. Gröller, D. Fleischmann Contents Motivation:» Why again a new visualization method for vessel data?
The VesselGlyph: Focus & Context Visualization in CT-Angiography Matúš Straka M. Šrámek, A. La Cruz E. Gröller, D. Fleischmann Contents Motivation:» Why again a new visualization method for vessel data?
Image Segmentation and Registration
 Image Segmentation and Registration Dr. Christine Tanner (tanner@vision.ee.ethz.ch) Computer Vision Laboratory, ETH Zürich Dr. Verena Kaynig, Machine Learning Laboratory, ETH Zürich Outline Segmentation
Image Segmentation and Registration Dr. Christine Tanner (tanner@vision.ee.ethz.ch) Computer Vision Laboratory, ETH Zürich Dr. Verena Kaynig, Machine Learning Laboratory, ETH Zürich Outline Segmentation
Phantom-based evaluation of a semi-automatic segmentation algorithm for cerebral vascular structures in 3D ultrasound angiography (3D USA)
 Phantom-based evaluation of a semi-automatic segmentation algorithm for cerebral vascular structures in 3D ultrasound angiography (3D USA) C. Chalopin¹, K. Krissian², A. Müns 3, F. Arlt 3, J. Meixensberger³,
Phantom-based evaluation of a semi-automatic segmentation algorithm for cerebral vascular structures in 3D ultrasound angiography (3D USA) C. Chalopin¹, K. Krissian², A. Müns 3, F. Arlt 3, J. Meixensberger³,
2 Michael E. Leventon and Sarah F. F. Gibson a b c d Fig. 1. (a, b) Two MR scans of a person's knee. Both images have high resolution in-plane, but ha
 Model Generation from Multiple Volumes using Constrained Elastic SurfaceNets Michael E. Leventon and Sarah F. F. Gibson 1 MIT Artificial Intelligence Laboratory, Cambridge, MA 02139, USA leventon@ai.mit.edu
Model Generation from Multiple Volumes using Constrained Elastic SurfaceNets Michael E. Leventon and Sarah F. F. Gibson 1 MIT Artificial Intelligence Laboratory, Cambridge, MA 02139, USA leventon@ai.mit.edu
Image Analysis, Geometrical Modelling and Image Synthesis for 3D Medical Imaging
 Image Analysis, Geometrical Modelling and Image Synthesis for 3D Medical Imaging J. SEQUEIRA Laboratoire d'informatique de Marseille - FRE CNRS 2246 Faculté des Sciences de Luminy, 163 avenue de Luminy,
Image Analysis, Geometrical Modelling and Image Synthesis for 3D Medical Imaging J. SEQUEIRA Laboratoire d'informatique de Marseille - FRE CNRS 2246 Faculté des Sciences de Luminy, 163 avenue de Luminy,
Object Identification in Ultrasound Scans
 Object Identification in Ultrasound Scans Wits University Dec 05, 2012 Roadmap Introduction to the problem Motivation Related Work Our approach Expected Results Introduction Nowadays, imaging devices like
Object Identification in Ultrasound Scans Wits University Dec 05, 2012 Roadmap Introduction to the problem Motivation Related Work Our approach Expected Results Introduction Nowadays, imaging devices like
A Comprehensive Method for Geometrically Correct 3-D Reconstruction of Coronary Arteries by Fusion of Intravascular Ultrasound and Biplane Angiography
 Computer-Aided Diagnosis in Medical Imaging, September 20-23, 1998, Chicago IL Elsevier ICS 1182 A Comprehensive Method for Geometrically Correct 3-D Reconstruction of Coronary Arteries by Fusion of Intravascular
Computer-Aided Diagnosis in Medical Imaging, September 20-23, 1998, Chicago IL Elsevier ICS 1182 A Comprehensive Method for Geometrically Correct 3-D Reconstruction of Coronary Arteries by Fusion of Intravascular
Computational Radiology Lab, Children s Hospital, Harvard Medical School, Boston, MA.
 Shape prior integration in discrete optimization segmentation algorithms M. Freiman Computational Radiology Lab, Children s Hospital, Harvard Medical School, Boston, MA. Email: moti.freiman@childrens.harvard.edu
Shape prior integration in discrete optimization segmentation algorithms M. Freiman Computational Radiology Lab, Children s Hospital, Harvard Medical School, Boston, MA. Email: moti.freiman@childrens.harvard.edu
Guide wire tracking in interventional radiology
 Guide wire tracking in interventional radiology S.A.M. Baert,W.J. Niessen, E.H.W. Meijering, A.F. Frangi, M.A. Viergever Image Sciences Institute, University Medical Center Utrecht, rm E 01.334, P.O.Box
Guide wire tracking in interventional radiology S.A.M. Baert,W.J. Niessen, E.H.W. Meijering, A.F. Frangi, M.A. Viergever Image Sciences Institute, University Medical Center Utrecht, rm E 01.334, P.O.Box
Automated segmentation methods for liver analysis in oncology applications
 University of Szeged Department of Image Processing and Computer Graphics Automated segmentation methods for liver analysis in oncology applications Ph. D. Thesis László Ruskó Thesis Advisor Dr. Antal
University of Szeged Department of Image Processing and Computer Graphics Automated segmentation methods for liver analysis in oncology applications Ph. D. Thesis László Ruskó Thesis Advisor Dr. Antal
A Non-Linear Image Registration Scheme for Real-Time Liver Ultrasound Tracking using Normalized Gradient Fields
 A Non-Linear Image Registration Scheme for Real-Time Liver Ultrasound Tracking using Normalized Gradient Fields Lars König, Till Kipshagen and Jan Rühaak Fraunhofer MEVIS Project Group Image Registration,
A Non-Linear Image Registration Scheme for Real-Time Liver Ultrasound Tracking using Normalized Gradient Fields Lars König, Till Kipshagen and Jan Rühaak Fraunhofer MEVIS Project Group Image Registration,
Vessel Explorer: a tool for quantitative measurements in CT and MR angiography
 Clinical applications Vessel Explorer: a tool for quantitative measurements in CT and MR angiography J. Oliván Bescós J. Sonnemans R. Habets J. Peters H. van den Bosch T. Leiner Healthcare Informatics/Patient
Clinical applications Vessel Explorer: a tool for quantitative measurements in CT and MR angiography J. Oliván Bescós J. Sonnemans R. Habets J. Peters H. van den Bosch T. Leiner Healthcare Informatics/Patient
Blood Vessel Visualization on CT Data
 WDS'12 Proceedings of Contributed Papers, Part I, 88 93, 2012. ISBN 978-80-7378-224-5 MATFYZPRESS Blood Vessel Visualization on CT Data J. Dupej Charles University Prague, Faculty of Mathematics and Physics,
WDS'12 Proceedings of Contributed Papers, Part I, 88 93, 2012. ISBN 978-80-7378-224-5 MATFYZPRESS Blood Vessel Visualization on CT Data J. Dupej Charles University Prague, Faculty of Mathematics and Physics,
CHAPTER 3 RETINAL OPTIC DISC SEGMENTATION
 60 CHAPTER 3 RETINAL OPTIC DISC SEGMENTATION 3.1 IMPORTANCE OF OPTIC DISC Ocular fundus images provide information about ophthalmic, retinal and even systemic diseases such as hypertension, diabetes, macular
60 CHAPTER 3 RETINAL OPTIC DISC SEGMENTATION 3.1 IMPORTANCE OF OPTIC DISC Ocular fundus images provide information about ophthalmic, retinal and even systemic diseases such as hypertension, diabetes, macular
Accurate Quantification of Small-Diameter Tubular Structures in Isotropic CT Volume Data Based on Multiscale Line Filter Responses
 Accurate Quantification of Small-Diameter Tubular Structures in Isotropic CT Volume Data Based on Multiscale Line Filter Responses Yoshinobu Sato 1, Shuji Yamamoto 2, and Shinichi Tamura 1 1 Division of
Accurate Quantification of Small-Diameter Tubular Structures in Isotropic CT Volume Data Based on Multiscale Line Filter Responses Yoshinobu Sato 1, Shuji Yamamoto 2, and Shinichi Tamura 1 1 Division of
Edge and local feature detection - 2. Importance of edge detection in computer vision
 Edge and local feature detection Gradient based edge detection Edge detection by function fitting Second derivative edge detectors Edge linking and the construction of the chain graph Edge and local feature
Edge and local feature detection Gradient based edge detection Edge detection by function fitting Second derivative edge detectors Edge linking and the construction of the chain graph Edge and local feature
RSNA, /rg
 RSNA, 2015 10.1148/rg.2015140320 Appendix As noted in the main article, DICOM image files cannot be used directly for 3D printing; further steps are necessary to make them readable by 3D printers. The
RSNA, 2015 10.1148/rg.2015140320 Appendix As noted in the main article, DICOM image files cannot be used directly for 3D printing; further steps are necessary to make them readable by 3D printers. The
Application of level set based method for segmentation of blood vessels in angiography images
 Lodz University of Technology Faculty of Electrical, Electronic, Computer and Control Engineering Institute of Electronics PhD Thesis Application of level set based method for segmentation of blood vessels
Lodz University of Technology Faculty of Electrical, Electronic, Computer and Control Engineering Institute of Electronics PhD Thesis Application of level set based method for segmentation of blood vessels
Coronary artery extraction from CT images
 Coronary artery extraction from CT images Richard Nordenskjöld April 2, 2009 Master s Thesis in Computing Science, 30 ECTS credits Supervisor at CS-UmU: Fredrik Georgsson Examiner: Per Lindström Umeå University
Coronary artery extraction from CT images Richard Nordenskjöld April 2, 2009 Master s Thesis in Computing Science, 30 ECTS credits Supervisor at CS-UmU: Fredrik Georgsson Examiner: Per Lindström Umeå University
Classification of Subject Motion for Improved Reconstruction of Dynamic Magnetic Resonance Imaging
 1 CS 9 Final Project Classification of Subject Motion for Improved Reconstruction of Dynamic Magnetic Resonance Imaging Feiyu Chen Department of Electrical Engineering ABSTRACT Subject motion is a significant
1 CS 9 Final Project Classification of Subject Motion for Improved Reconstruction of Dynamic Magnetic Resonance Imaging Feiyu Chen Department of Electrical Engineering ABSTRACT Subject motion is a significant
Fast 3D Mean Shift Filter for CT Images
 Fast 3D Mean Shift Filter for CT Images Gustavo Fernández Domínguez, Horst Bischof, and Reinhard Beichel Institute for Computer Graphics and Vision, Graz University of Technology Inffeldgasse 16/2, A-8010,
Fast 3D Mean Shift Filter for CT Images Gustavo Fernández Domínguez, Horst Bischof, and Reinhard Beichel Institute for Computer Graphics and Vision, Graz University of Technology Inffeldgasse 16/2, A-8010,
Interactive Boundary Detection for Automatic Definition of 2D Opacity Transfer Function
 Interactive Boundary Detection for Automatic Definition of 2D Opacity Transfer Function Martin Rauberger, Heinrich Martin Overhoff Medical Engineering Laboratory, University of Applied Sciences Gelsenkirchen,
Interactive Boundary Detection for Automatic Definition of 2D Opacity Transfer Function Martin Rauberger, Heinrich Martin Overhoff Medical Engineering Laboratory, University of Applied Sciences Gelsenkirchen,
Automated Lesion Detection Methods for 2D and 3D Chest X-Ray Images
 Automated Lesion Detection Methods for 2D and 3D Chest X-Ray Images Takeshi Hara, Hiroshi Fujita,Yongbum Lee, Hitoshi Yoshimura* and Shoji Kido** Department of Information Science, Gifu University Yanagido
Automated Lesion Detection Methods for 2D and 3D Chest X-Ray Images Takeshi Hara, Hiroshi Fujita,Yongbum Lee, Hitoshi Yoshimura* and Shoji Kido** Department of Information Science, Gifu University Yanagido
Interactive Differential Segmentation of the Prostate using Graph-Cuts with a Feature Detector-based Boundary Term
 MOSCHIDIS, GRAHAM: GRAPH-CUTS WITH FEATURE DETECTORS 1 Interactive Differential Segmentation of the Prostate using Graph-Cuts with a Feature Detector-based Boundary Term Emmanouil Moschidis emmanouil.moschidis@postgrad.manchester.ac.uk
MOSCHIDIS, GRAHAM: GRAPH-CUTS WITH FEATURE DETECTORS 1 Interactive Differential Segmentation of the Prostate using Graph-Cuts with a Feature Detector-based Boundary Term Emmanouil Moschidis emmanouil.moschidis@postgrad.manchester.ac.uk
Blood Particle Trajectories in Phase-Contrast-MRI as Minimal Paths Computed with Anisotropic Fast Marching
 Blood Particle Trajectories in Phase-Contrast-MRI as Minimal Paths Computed with Anisotropic Fast Marching Michael Schwenke 1, Anja Hennemuth 1, Bernd Fischer 2, Ola Friman 1 1 Fraunhofer MEVIS, Institute
Blood Particle Trajectories in Phase-Contrast-MRI as Minimal Paths Computed with Anisotropic Fast Marching Michael Schwenke 1, Anja Hennemuth 1, Bernd Fischer 2, Ola Friman 1 1 Fraunhofer MEVIS, Institute
Registration-Based Segmentation of Medical Images
 School of Computing National University of Singapore Graduate Research Paper Registration-Based Segmentation of Medical Images by Li Hao under guidance of A/Prof. Leow Wee Kheng July, 2006 Abstract Medical
School of Computing National University of Singapore Graduate Research Paper Registration-Based Segmentation of Medical Images by Li Hao under guidance of A/Prof. Leow Wee Kheng July, 2006 Abstract Medical
CoE4TN4 Image Processing
 CoE4TN4 Image Processing Chapter 11 Image Representation & Description Image Representation & Description After an image is segmented into regions, the regions are represented and described in a form suitable
CoE4TN4 Image Processing Chapter 11 Image Representation & Description Image Representation & Description After an image is segmented into regions, the regions are represented and described in a form suitable
An Automated Image-based Method for Multi-Leaf Collimator Positioning Verification in Intensity Modulated Radiation Therapy
 An Automated Image-based Method for Multi-Leaf Collimator Positioning Verification in Intensity Modulated Radiation Therapy Chenyang Xu 1, Siemens Corporate Research, Inc., Princeton, NJ, USA Xiaolei Huang,
An Automated Image-based Method for Multi-Leaf Collimator Positioning Verification in Intensity Modulated Radiation Therapy Chenyang Xu 1, Siemens Corporate Research, Inc., Princeton, NJ, USA Xiaolei Huang,
SEGMENTATION AND RECONSTRUCTION OF 3D ARTERY MODELS FOR SURGICAL PLANNING
 SEGMENTATION AND RECONSTRUCTION OF 3D ARTERY MODELS FOR SURGICAL PLANNING QI YINGYI (B.Sc., FUDAN UNIVERSITY, 2005 ) A THESIS SUBMITTED FOR THE DEGREE OF MASTER OF SCIENCE DEPARTMENT OF COMPUTER SCIENCE
SEGMENTATION AND RECONSTRUCTION OF 3D ARTERY MODELS FOR SURGICAL PLANNING QI YINGYI (B.Sc., FUDAN UNIVERSITY, 2005 ) A THESIS SUBMITTED FOR THE DEGREE OF MASTER OF SCIENCE DEPARTMENT OF COMPUTER SCIENCE
MEDICAL IMAGE NOISE REDUCTION AND REGION CONTRAST ENHANCEMENT USING PARTIAL DIFFERENTIAL EQUATIONS
 MEDICAL IMAGE NOISE REDUCTION AND REGION CONTRAST ENHANCEMENT USING PARTIAL DIFFERENTIAL EQUATIONS Miguel Alemán-Flores, Luis Álvarez-León Departamento de Informática y Sistemas, Universidad de Las Palmas
MEDICAL IMAGE NOISE REDUCTION AND REGION CONTRAST ENHANCEMENT USING PARTIAL DIFFERENTIAL EQUATIONS Miguel Alemán-Flores, Luis Álvarez-León Departamento de Informática y Sistemas, Universidad de Las Palmas
Medicale Image Analysis
 Medicale Image Analysis Registration Validation Prof. Dr. Philippe Cattin MIAC, University of Basel Prof. Dr. Philippe Cattin: Registration Validation Contents 1 Validation 1.1 Validation of Registration
Medicale Image Analysis Registration Validation Prof. Dr. Philippe Cattin MIAC, University of Basel Prof. Dr. Philippe Cattin: Registration Validation Contents 1 Validation 1.1 Validation of Registration
Introduction to Medical Image Processing
 Introduction to Medical Image Processing Δ Essential environments of a medical imaging system Subject Image Analysis Energy Imaging System Images Image Processing Feature Images Image processing may be
Introduction to Medical Image Processing Δ Essential environments of a medical imaging system Subject Image Analysis Energy Imaging System Images Image Processing Feature Images Image processing may be
TUMOR DETECTION IN MRI IMAGES
 TUMOR DETECTION IN MRI IMAGES Prof. Pravin P. Adivarekar, 2 Priyanka P. Khatate, 3 Punam N. Pawar Prof. Pravin P. Adivarekar, 2 Priyanka P. Khatate, 3 Punam N. Pawar Asst. Professor, 2,3 BE Student,,2,3
TUMOR DETECTION IN MRI IMAGES Prof. Pravin P. Adivarekar, 2 Priyanka P. Khatate, 3 Punam N. Pawar Prof. Pravin P. Adivarekar, 2 Priyanka P. Khatate, 3 Punam N. Pawar Asst. Professor, 2,3 BE Student,,2,3
4D Magnetic Resonance Analysis. MR 4D Flow. Visualization and Quantification of Aortic Blood Flow
 4D Magnetic Resonance Analysis MR 4D Flow Visualization and Quantification of Aortic Blood Flow 4D Magnetic Resonance Analysis Complete assesment of your MR 4D Flow data Time-efficient and intuitive analysis
4D Magnetic Resonance Analysis MR 4D Flow Visualization and Quantification of Aortic Blood Flow 4D Magnetic Resonance Analysis Complete assesment of your MR 4D Flow data Time-efficient and intuitive analysis
Respiratory Motion Estimation using a 3D Diaphragm Model
 Respiratory Motion Estimation using a 3D Diaphragm Model Marco Bögel 1,2, Christian Riess 1,2, Andreas Maier 1, Joachim Hornegger 1, Rebecca Fahrig 2 1 Pattern Recognition Lab, FAU Erlangen-Nürnberg 2
Respiratory Motion Estimation using a 3D Diaphragm Model Marco Bögel 1,2, Christian Riess 1,2, Andreas Maier 1, Joachim Hornegger 1, Rebecca Fahrig 2 1 Pattern Recognition Lab, FAU Erlangen-Nürnberg 2
Generalizing vesselness with respect to dimensionality and shape Release 1.00
 Generalizing vesselness with respect to dimensionality and shape Release 1.00 Luca Antiga 1 August 3, 2007 1 Medical Imaging Unit Mario Negri Institute, Bergamo, Italy email: antiga at marionegri.it Abstract
Generalizing vesselness with respect to dimensionality and shape Release 1.00 Luca Antiga 1 August 3, 2007 1 Medical Imaging Unit Mario Negri Institute, Bergamo, Italy email: antiga at marionegri.it Abstract
Automatic segmentation of the cortical grey and white matter in MRI using a Region Growing approach based on anatomical knowledge
 Automatic segmentation of the cortical grey and white matter in MRI using a Region Growing approach based on anatomical knowledge Christian Wasserthal 1, Karin Engel 1, Karsten Rink 1 und André Brechmann
Automatic segmentation of the cortical grey and white matter in MRI using a Region Growing approach based on anatomical knowledge Christian Wasserthal 1, Karin Engel 1, Karsten Rink 1 und André Brechmann
Segmentation Using a Region Growing Thresholding
 Segmentation Using a Region Growing Thresholding Matei MANCAS 1, Bernard GOSSELIN 1, Benoît MACQ 2 1 Faculté Polytechnique de Mons, Circuit Theory and Signal Processing Laboratory Bâtiment MULTITEL/TCTS
Segmentation Using a Region Growing Thresholding Matei MANCAS 1, Bernard GOSSELIN 1, Benoît MACQ 2 1 Faculté Polytechnique de Mons, Circuit Theory and Signal Processing Laboratory Bâtiment MULTITEL/TCTS
Respiratory Motion Compensation for C-arm CT Liver Imaging
 Respiratory Motion Compensation for C-arm CT Liver Imaging Aline Sindel 1, Marco Bögel 1,2, Andreas Maier 1,2, Rebecca Fahrig 3, Joachim Hornegger 1,2, Arnd Dörfler 4 1 Pattern Recognition Lab, FAU Erlangen-Nürnberg
Respiratory Motion Compensation for C-arm CT Liver Imaging Aline Sindel 1, Marco Bögel 1,2, Andreas Maier 1,2, Rebecca Fahrig 3, Joachim Hornegger 1,2, Arnd Dörfler 4 1 Pattern Recognition Lab, FAU Erlangen-Nürnberg
Motion artifact detection in four-dimensional computed tomography images
 Motion artifact detection in four-dimensional computed tomography images G Bouilhol 1,, M Ayadi, R Pinho, S Rit 1, and D Sarrut 1, 1 University of Lyon, CREATIS; CNRS UMR 5; Inserm U144; INSA-Lyon; University
Motion artifact detection in four-dimensional computed tomography images G Bouilhol 1,, M Ayadi, R Pinho, S Rit 1, and D Sarrut 1, 1 University of Lyon, CREATIS; CNRS UMR 5; Inserm U144; INSA-Lyon; University
Network Snakes for the Segmentation of Adjacent Cells in Confocal Images
 Network Snakes for the Segmentation of Adjacent Cells in Confocal Images Matthias Butenuth 1 and Fritz Jetzek 2 1 Institut für Photogrammetrie und GeoInformation, Leibniz Universität Hannover, 30167 Hannover
Network Snakes for the Segmentation of Adjacent Cells in Confocal Images Matthias Butenuth 1 and Fritz Jetzek 2 1 Institut für Photogrammetrie und GeoInformation, Leibniz Universität Hannover, 30167 Hannover
Simultaneous Model-based Segmentation of Multiple Objects
 Simultaneous Model-based Segmentation of Multiple Objects Astrid Franz 1, Robin Wolz 1, Tobias Klinder 1,2, Cristian Lorenz 1, Hans Barschdorf 1, Thomas Blaffert 1, Sebastian P. M. Dries 1, Steffen Renisch
Simultaneous Model-based Segmentation of Multiple Objects Astrid Franz 1, Robin Wolz 1, Tobias Klinder 1,2, Cristian Lorenz 1, Hans Barschdorf 1, Thomas Blaffert 1, Sebastian P. M. Dries 1, Steffen Renisch
Evaluation of Hessian-based filters to enhance the axis of coronary arteries in CT images
 International Congress Series 1256 (2003) 1191 1196 Evaluation of Hessian-based filters to enhance the axis of coronary arteries in CT images S.D. Olabarriaga a, *, M. Breeuwer b, W.J. Niessen a a University
International Congress Series 1256 (2003) 1191 1196 Evaluation of Hessian-based filters to enhance the axis of coronary arteries in CT images S.D. Olabarriaga a, *, M. Breeuwer b, W.J. Niessen a a University
HOUGH TRANSFORM CS 6350 C V
 HOUGH TRANSFORM CS 6350 C V HOUGH TRANSFORM The problem: Given a set of points in 2-D, find if a sub-set of these points, fall on a LINE. Hough Transform One powerful global method for detecting edges
HOUGH TRANSFORM CS 6350 C V HOUGH TRANSFORM The problem: Given a set of points in 2-D, find if a sub-set of these points, fall on a LINE. Hough Transform One powerful global method for detecting edges
INDUSTRIAL SYSTEM DEVELOPMENT FOR VOLUMETRIC INTEGRITY
 INDUSTRIAL SYSTEM DEVELOPMENT FOR VOLUMETRIC INTEGRITY VERIFICATION AND ANALYSIS M. L. Hsiao and J. W. Eberhard CR&D General Electric Company Schenectady, NY 12301 J. B. Ross Aircraft Engine - QTC General
INDUSTRIAL SYSTEM DEVELOPMENT FOR VOLUMETRIC INTEGRITY VERIFICATION AND ANALYSIS M. L. Hsiao and J. W. Eberhard CR&D General Electric Company Schenectady, NY 12301 J. B. Ross Aircraft Engine - QTC General
MEDICAL IMAGE COMPUTING (CAP 5937) LECTURE 19: Machine Learning in Medical Imaging (A Brief Introduction)
 SPRING 2016 1 MEDICAL IMAGE COMPUTING (CAP 5937) LECTURE 19: Machine Learning in Medical Imaging (A Brief Introduction) Dr. Ulas Bagci HEC 221, Center for Research in Computer Vision (CRCV), University
SPRING 2016 1 MEDICAL IMAGE COMPUTING (CAP 5937) LECTURE 19: Machine Learning in Medical Imaging (A Brief Introduction) Dr. Ulas Bagci HEC 221, Center for Research in Computer Vision (CRCV), University
DETECTING and extracting blood vessels in magnetic resonance
 1224 IEEE TRANSACTIONS ON MEDICAL IMAGING, VOL. 26, NO. 9, SEPTEMBER 2007 Weighted Local Variance-Based Edge Detection and Its Application to Vascular Segmentation in Magnetic Resonance Angiography Max
1224 IEEE TRANSACTIONS ON MEDICAL IMAGING, VOL. 26, NO. 9, SEPTEMBER 2007 Weighted Local Variance-Based Edge Detection and Its Application to Vascular Segmentation in Magnetic Resonance Angiography Max
A Hybrid Method for Coronary Artery Stenoses Detection and Quantification in CTA Images
 A Hybrid Method for Coronary Artery Stenoses Detection and Quantification in CTA Images İlkay Öksüz 1, Devrim Ünay 2, Kamuran Kadıpaşaoğlu 2 1 Electrical and Electronics Engineering, Bahçeşehir University,
A Hybrid Method for Coronary Artery Stenoses Detection and Quantification in CTA Images İlkay Öksüz 1, Devrim Ünay 2, Kamuran Kadıpaşaoğlu 2 1 Electrical and Electronics Engineering, Bahçeşehir University,
Vessel Centerline Tracking in CTA and MRA Images Using Hough Transform
 Vessel Centerline Tracking in CTA and MRA Images Using Hough Transform Maysa M.G. Macedo 1, Choukri Mekkaoui 2, and Marcel P. Jackowski 1 1 University of São Paulo, Department of Computer Science, Rua
Vessel Centerline Tracking in CTA and MRA Images Using Hough Transform Maysa M.G. Macedo 1, Choukri Mekkaoui 2, and Marcel P. Jackowski 1 1 University of São Paulo, Department of Computer Science, Rua
3D Guide Wire Navigation from Single Plane Fluoroscopic Images in Abdominal Catheterizations
 3D Guide Wire Navigation from Single Plane Fluoroscopic Images in Abdominal Catheterizations Martin Groher 2, Frederik Bender 1, Ali Khamene 3, Wolfgang Wein 3, Tim Hauke Heibel 2, Nassir Navab 2 1 Siemens
3D Guide Wire Navigation from Single Plane Fluoroscopic Images in Abdominal Catheterizations Martin Groher 2, Frederik Bender 1, Ali Khamene 3, Wolfgang Wein 3, Tim Hauke Heibel 2, Nassir Navab 2 1 Siemens
Edge-Preserving Denoising for Segmentation in CT-Images
 Edge-Preserving Denoising for Segmentation in CT-Images Eva Eibenberger, Anja Borsdorf, Andreas Wimmer, Joachim Hornegger Lehrstuhl für Mustererkennung, Friedrich-Alexander-Universität Erlangen-Nürnberg
Edge-Preserving Denoising for Segmentation in CT-Images Eva Eibenberger, Anja Borsdorf, Andreas Wimmer, Joachim Hornegger Lehrstuhl für Mustererkennung, Friedrich-Alexander-Universität Erlangen-Nürnberg
Learning-based Neuroimage Registration
 Learning-based Neuroimage Registration Leonid Teverovskiy and Yanxi Liu 1 October 2004 CMU-CALD-04-108, CMU-RI-TR-04-59 School of Computer Science Carnegie Mellon University Pittsburgh, PA 15213 Abstract
Learning-based Neuroimage Registration Leonid Teverovskiy and Yanxi Liu 1 October 2004 CMU-CALD-04-108, CMU-RI-TR-04-59 School of Computer Science Carnegie Mellon University Pittsburgh, PA 15213 Abstract
Image Acquisition Systems
 Image Acquisition Systems Goals and Terminology Conventional Radiography Axial Tomography Computer Axial Tomography (CAT) Magnetic Resonance Imaging (MRI) PET, SPECT Ultrasound Microscopy Imaging ITCS
Image Acquisition Systems Goals and Terminology Conventional Radiography Axial Tomography Computer Axial Tomography (CAT) Magnetic Resonance Imaging (MRI) PET, SPECT Ultrasound Microscopy Imaging ITCS
Hybrid Spline-based Multimodal Registration using a Local Measure for Mutual Information
 Hybrid Spline-based Multimodal Registration using a Local Measure for Mutual Information Andreas Biesdorf 1, Stefan Wörz 1, Hans-Jürgen Kaiser 2, Karl Rohr 1 1 University of Heidelberg, BIOQUANT, IPMB,
Hybrid Spline-based Multimodal Registration using a Local Measure for Mutual Information Andreas Biesdorf 1, Stefan Wörz 1, Hans-Jürgen Kaiser 2, Karl Rohr 1 1 University of Heidelberg, BIOQUANT, IPMB,
Chapter 11 Arc Extraction and Segmentation
 Chapter 11 Arc Extraction and Segmentation 11.1 Introduction edge detection: labels each pixel as edge or no edge additional properties of edge: direction, gradient magnitude, contrast edge grouping: edge
Chapter 11 Arc Extraction and Segmentation 11.1 Introduction edge detection: labels each pixel as edge or no edge additional properties of edge: direction, gradient magnitude, contrast edge grouping: edge
Biometrics Technology: Image Processing & Pattern Recognition (by Dr. Dickson Tong)
 Biometrics Technology: Image Processing & Pattern Recognition (by Dr. Dickson Tong) References: [1] http://homepages.inf.ed.ac.uk/rbf/hipr2/index.htm [2] http://www.cs.wisc.edu/~dyer/cs540/notes/vision.html
Biometrics Technology: Image Processing & Pattern Recognition (by Dr. Dickson Tong) References: [1] http://homepages.inf.ed.ac.uk/rbf/hipr2/index.htm [2] http://www.cs.wisc.edu/~dyer/cs540/notes/vision.html
Biomedical Image Analysis. Point, Edge and Line Detection
 Biomedical Image Analysis Point, Edge and Line Detection Contents: Point and line detection Advanced edge detection: Canny Local/regional edge processing Global processing: Hough transform BMIA 15 V. Roth
Biomedical Image Analysis Point, Edge and Line Detection Contents: Point and line detection Advanced edge detection: Canny Local/regional edge processing Global processing: Hough transform BMIA 15 V. Roth
Convolution-Based Truncation Correction for C-Arm CT using Scattered Radiation
 Convolution-Based Truncation Correction for C-Arm CT using Scattered Radiation Bastian Bier 1, Chris Schwemmer 1,2, Andreas Maier 1,3, Hannes G. Hofmann 1, Yan Xia 1, Joachim Hornegger 1,2, Tobias Struffert
Convolution-Based Truncation Correction for C-Arm CT using Scattered Radiation Bastian Bier 1, Chris Schwemmer 1,2, Andreas Maier 1,3, Hannes G. Hofmann 1, Yan Xia 1, Joachim Hornegger 1,2, Tobias Struffert
Announcements. Edges. Last Lecture. Gradients: Numerical Derivatives f(x) Edge Detection, Lines. Intro Computer Vision. CSE 152 Lecture 10
 Announcements Assignment 2 due Tuesday, May 4. Edge Detection, Lines Midterm: Thursday, May 6. Introduction to Computer Vision CSE 152 Lecture 10 Edges Last Lecture 1. Object boundaries 2. Surface normal
Announcements Assignment 2 due Tuesday, May 4. Edge Detection, Lines Midterm: Thursday, May 6. Introduction to Computer Vision CSE 152 Lecture 10 Edges Last Lecture 1. Object boundaries 2. Surface normal
A Registration-Based Atlas Propagation Framework for Automatic Whole Heart Segmentation
 A Registration-Based Atlas Propagation Framework for Automatic Whole Heart Segmentation Xiahai Zhuang (PhD) Centre for Medical Image Computing University College London Fields-MITACS Conference on Mathematics
A Registration-Based Atlas Propagation Framework for Automatic Whole Heart Segmentation Xiahai Zhuang (PhD) Centre for Medical Image Computing University College London Fields-MITACS Conference on Mathematics
Pearling: Medical Image Segmentation with Pearl Strings
 Pearling: Medical Image Segmentation with Pearl Strings Jarek Rossignac 1, Brian Whited 1, Greg Slabaugh 2, Tong Fang 2, Gozde Unal 2 1 Georgia Institute of Technology Graphics, Visualization, and Usability
Pearling: Medical Image Segmentation with Pearl Strings Jarek Rossignac 1, Brian Whited 1, Greg Slabaugh 2, Tong Fang 2, Gozde Unal 2 1 Georgia Institute of Technology Graphics, Visualization, and Usability
Towards Projector-based Visualization for Computer-assisted CABG at the Open Heart
 Towards Projector-based Visualization for Computer-assisted CABG at the Open Heart Christine Hartung 1, Claudia Gnahm 1, Stefan Sailer 1, Marcel Schenderlein 1, Reinhard Friedl 2, Martin Hoffmann 3, Klaus
Towards Projector-based Visualization for Computer-assisted CABG at the Open Heart Christine Hartung 1, Claudia Gnahm 1, Stefan Sailer 1, Marcel Schenderlein 1, Reinhard Friedl 2, Martin Hoffmann 3, Klaus
SUMMARY: DISTINCTIVE IMAGE FEATURES FROM SCALE- INVARIANT KEYPOINTS
 SUMMARY: DISTINCTIVE IMAGE FEATURES FROM SCALE- INVARIANT KEYPOINTS Cognitive Robotics Original: David G. Lowe, 004 Summary: Coen van Leeuwen, s1460919 Abstract: This article presents a method to extract
SUMMARY: DISTINCTIVE IMAGE FEATURES FROM SCALE- INVARIANT KEYPOINTS Cognitive Robotics Original: David G. Lowe, 004 Summary: Coen van Leeuwen, s1460919 Abstract: This article presents a method to extract
Robotics Programming Laboratory
 Chair of Software Engineering Robotics Programming Laboratory Bertrand Meyer Jiwon Shin Lecture 8: Robot Perception Perception http://pascallin.ecs.soton.ac.uk/challenges/voc/databases.html#caltech car
Chair of Software Engineering Robotics Programming Laboratory Bertrand Meyer Jiwon Shin Lecture 8: Robot Perception Perception http://pascallin.ecs.soton.ac.uk/challenges/voc/databases.html#caltech car
A client-server architecture for semi-automatic segmentation of peripheral vessels in CTA data
 A client-server architecture for semi-automatic segmentation of peripheral vessels in CTA data Poster No.: C-2174 Congress: ECR 2013 Type: Authors: Keywords: DOI: Scientific Exhibit A. Grünauer, E. Vuçini,
A client-server architecture for semi-automatic segmentation of peripheral vessels in CTA data Poster No.: C-2174 Congress: ECR 2013 Type: Authors: Keywords: DOI: Scientific Exhibit A. Grünauer, E. Vuçini,
Multi-slice CT Image Reconstruction Jiang Hsieh, Ph.D.
 Multi-slice CT Image Reconstruction Jiang Hsieh, Ph.D. Applied Science Laboratory, GE Healthcare Technologies 1 Image Generation Reconstruction of images from projections. textbook reconstruction advanced
Multi-slice CT Image Reconstruction Jiang Hsieh, Ph.D. Applied Science Laboratory, GE Healthcare Technologies 1 Image Generation Reconstruction of images from projections. textbook reconstruction advanced
Problem Solving Assignment 1
 CS6240 Problem Solving Assignment 1 p. 1/20 Problem Solving Assignment 1 CS6240 Multimedia Analysis Daniel Dahlmeier National University of Singapore CS6240 Problem Solving Assignment 1 p. 2/20 Introduction
CS6240 Problem Solving Assignment 1 p. 1/20 Problem Solving Assignment 1 CS6240 Multimedia Analysis Daniel Dahlmeier National University of Singapore CS6240 Problem Solving Assignment 1 p. 2/20 Introduction
Computer-aided Diagnosis of Retinopathy of Prematurity
 Computer-aided Diagnosis of Retinopathy of Prematurity Rangaraj M. Rangayyan, Faraz Oloumi, and Anna L. Ells Department of Electrical and Computer Engineering, University of Calgary Alberta Children's
Computer-aided Diagnosis of Retinopathy of Prematurity Rangaraj M. Rangayyan, Faraz Oloumi, and Anna L. Ells Department of Electrical and Computer Engineering, University of Calgary Alberta Children's
Frequency split metal artifact reduction (FSMAR) in computed tomography
 The Johns Hopkins University Advanced Computer Integrated Surgery Group 4 Metal Artifact Removal in C-arm Cone-Beam CT Paper Seminar Critical Review of Frequency split metal artifact reduction (FSMAR)
The Johns Hopkins University Advanced Computer Integrated Surgery Group 4 Metal Artifact Removal in C-arm Cone-Beam CT Paper Seminar Critical Review of Frequency split metal artifact reduction (FSMAR)
coding of various parts showing different features, the possibility of rotation or of hiding covering parts of the object's surface to gain an insight
 Three-Dimensional Object Reconstruction from Layered Spatial Data Michael Dangl and Robert Sablatnig Vienna University of Technology, Institute of Computer Aided Automation, Pattern Recognition and Image
Three-Dimensional Object Reconstruction from Layered Spatial Data Michael Dangl and Robert Sablatnig Vienna University of Technology, Institute of Computer Aided Automation, Pattern Recognition and Image
Norbert Schuff VA Medical Center and UCSF
 Norbert Schuff Medical Center and UCSF Norbert.schuff@ucsf.edu Medical Imaging Informatics N.Schuff Course # 170.03 Slide 1/67 Objective Learn the principle segmentation techniques Understand the role
Norbert Schuff Medical Center and UCSF Norbert.schuff@ucsf.edu Medical Imaging Informatics N.Schuff Course # 170.03 Slide 1/67 Objective Learn the principle segmentation techniques Understand the role
Landmark-based 3D Elastic Registration of Pre- and Postoperative Liver CT Data
 Landmark-based 3D Elastic Registration of Pre- and Postoperative Liver CT Data An Experimental Comparison Thomas Lange 1, Stefan Wörz 2, Karl Rohr 2, Peter M. Schlag 3 1 Experimental and Clinical Research
Landmark-based 3D Elastic Registration of Pre- and Postoperative Liver CT Data An Experimental Comparison Thomas Lange 1, Stefan Wörz 2, Karl Rohr 2, Peter M. Schlag 3 1 Experimental and Clinical Research
Automatic Extraction of Femur Contours from Hip X-ray Images
 Automatic Extraction of Femur Contours from Hip X-ray Images Ying Chen 1, Xianhe Ee 1, Wee Kheng Leow 1, and Tet Sen Howe 2 1 Dept of Computer Science, National University of Singapore, 3 Science Drive
Automatic Extraction of Femur Contours from Hip X-ray Images Ying Chen 1, Xianhe Ee 1, Wee Kheng Leow 1, and Tet Sen Howe 2 1 Dept of Computer Science, National University of Singapore, 3 Science Drive
Computer-Aided Diagnosis in Abdominal and Cardiac Radiology Using Neural Networks
 Computer-Aided Diagnosis in Abdominal and Cardiac Radiology Using Neural Networks Du-Yih Tsai, Masaru Sekiya and Yongbum Lee Department of Radiological Technology, School of Health Sciences, Faculty of
Computer-Aided Diagnosis in Abdominal and Cardiac Radiology Using Neural Networks Du-Yih Tsai, Masaru Sekiya and Yongbum Lee Department of Radiological Technology, School of Health Sciences, Faculty of
GPU Based Region Growth and Vessel Tracking. Supratik Moulik M.D. Jason Walsh
 GPU Based Region Growth and Vessel Tracking Supratik Moulik M.D. (supratik@moulik.com) Jason Walsh Conflict of Interest Dr. Supratik Moulik does not have a significant financial stake in any company, nor
GPU Based Region Growth and Vessel Tracking Supratik Moulik M.D. (supratik@moulik.com) Jason Walsh Conflict of Interest Dr. Supratik Moulik does not have a significant financial stake in any company, nor
Generation of Triangle Meshes from Time-of-Flight Data for Surface Registration
 Generation of Triangle Meshes from Time-of-Flight Data for Surface Registration Thomas Kilgus, Thiago R. dos Santos, Alexander Seitel, Kwong Yung, Alfred M. Franz, Anja Groch, Ivo Wolf, Hans-Peter Meinzer,
Generation of Triangle Meshes from Time-of-Flight Data for Surface Registration Thomas Kilgus, Thiago R. dos Santos, Alexander Seitel, Kwong Yung, Alfred M. Franz, Anja Groch, Ivo Wolf, Hans-Peter Meinzer,
Calypso Construction Features. Construction Features 1
 Calypso 1 The Construction dropdown menu contains several useful construction features that can be used to compare two other features or perform special calculations. Construction features will show up
Calypso 1 The Construction dropdown menu contains several useful construction features that can be used to compare two other features or perform special calculations. Construction features will show up
9 length of contour = no. of horizontal and vertical components + ( 2 no. of diagonal components) diameter of boundary B
 8. Boundary Descriptor 8.. Some Simple Descriptors length of contour : simplest descriptor - chain-coded curve 9 length of contour no. of horiontal and vertical components ( no. of diagonal components
8. Boundary Descriptor 8.. Some Simple Descriptors length of contour : simplest descriptor - chain-coded curve 9 length of contour no. of horiontal and vertical components ( no. of diagonal components
An Automatic Seeding Method For Coronary Artery Segmentation and Skeletonization in CTA Release 0.00
 An Automatic Seeding Method For Coronary Artery Segmentation and Skeletonization in CTA Release 0.00 Chunliang Wang 1 and Örjan Smedby 1 July 8, 2008 1 Linkping University, CMIV Abstract An automatic seeding
An Automatic Seeding Method For Coronary Artery Segmentation and Skeletonization in CTA Release 0.00 Chunliang Wang 1 and Örjan Smedby 1 July 8, 2008 1 Linkping University, CMIV Abstract An automatic seeding
Methodological progress in image registration for ventilation estimation, segmentation propagation and multi-modal fusion
 Methodological progress in image registration for ventilation estimation, segmentation propagation and multi-modal fusion Mattias P. Heinrich Julia A. Schnabel, Mark Jenkinson, Sir Michael Brady 2 Clinical
Methodological progress in image registration for ventilation estimation, segmentation propagation and multi-modal fusion Mattias P. Heinrich Julia A. Schnabel, Mark Jenkinson, Sir Michael Brady 2 Clinical
The Near Future in Cardiac CT Image Reconstruction
 SCCT 2010 The Near Future in Cardiac CT Image Reconstruction Marc Kachelrieß Institute of Medical Physics (IMP) Friedrich-Alexander Alexander-University Erlangen-Nürnberg rnberg www.imp.uni-erlangen.de
SCCT 2010 The Near Future in Cardiac CT Image Reconstruction Marc Kachelrieß Institute of Medical Physics (IMP) Friedrich-Alexander Alexander-University Erlangen-Nürnberg rnberg www.imp.uni-erlangen.de
GPU-Based Airway Segmentation and Centerline Extraction for Image Guided Bronchoscopy
 GPU-Based Airway Segmentation and Centerline Extraction for Image Guided Bronchoscopy Erik Smistad 1, Anne C. Elster 1, Frank Lindseth 1,2 1) Norwegian University of Science and Technology 2) SINTEF Medical
GPU-Based Airway Segmentation and Centerline Extraction for Image Guided Bronchoscopy Erik Smistad 1, Anne C. Elster 1, Frank Lindseth 1,2 1) Norwegian University of Science and Technology 2) SINTEF Medical
Quantitative IntraVascular UltraSound (QCU)
 Quantitative IntraVascular UltraSound (QCU) Authors: Jouke Dijkstra, Ph.D. and Johan H.C. Reiber, Ph.D., Leiden University Medical Center, Dept of Radiology, Leiden, The Netherlands Introduction: For decades,
Quantitative IntraVascular UltraSound (QCU) Authors: Jouke Dijkstra, Ph.D. and Johan H.C. Reiber, Ph.D., Leiden University Medical Center, Dept of Radiology, Leiden, The Netherlands Introduction: For decades,
Rigid and Deformable Vasculature-to-Image Registration : a Hierarchical Approach
 Rigid and Deformable Vasculature-to-Image Registration : a Hierarchical Approach Julien Jomier and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab The University of North Carolina at Chapel
Rigid and Deformable Vasculature-to-Image Registration : a Hierarchical Approach Julien Jomier and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab The University of North Carolina at Chapel
