SAMPLE LIS01-A2. Archived Document
|
|
|
- Olivia Parker
- 5 years ago
- Views:
Transcription
1 Archived Document This archived document is no longer being reviewed through the CLSI Consensus Document Development Process. However, this document is technically valid as of January Because of its value to the laboratory community, it is being retained in CLSI s library. Specification for Low-Level Protocol to Transfer Messages Between Clinical Laboratory Instruments and Computer Systems; Approved Standard Second Edition This document describes the electronic transmission of digital information between clinical laboratory instruments and computer systems. April 2008 A standard for global application developed through the Clinical and Laboratory Standards Institute consensus process.
2 Clinical and Laboratory Standards Institute Setting the standard for quality in medical laboratory testing around the world. The Clinical and Laboratory Standards Institute (CLSI) is a not-for-profit membership organization that brings together the varied perspectives and expertise of the worldwide laboratory community for the advancement of a common cause: to foster excellence in laboratory medicine by developing and implementing medical laboratory standards and guidelines that help laboratories fulfill their responsibilities with efficiency, effectiveness, and global applicability. Consensus Process Consensus the substantial agreement by materially affected, competent, and interested parties is core to the development of all CLSI documents. It does not always connote unanimous agreement, but does mean that the participants in the development of a consensus document have considered and resolved all relevant objections and accept the resulting agreement. Commenting on Documents CLSI documents undergo periodic evaluation and modification to keep pace with advancements in technologies, procedures, methods, and protocols affecting the laboratory or health care. CLSI s consensus process depends on experts who volunteer to serve as contributing authors and/or as participants in the reviewing and commenting process. At the end of each comment period, the committee that developed the document is obligated to review all comments, respond in writing to all substantive comments, and revise the draft document as appropriate. Comments on published CLSI documents are equally essential, and may be submitted by anyone, at any time, on any document. All comments are managed according to the consensus process by a committee of experts. Appeals Process When it is believed that an objection has not been adequately considered and responded to, the process for appeals, documented in the CLSI Standards Development Policies and Processes, is followed. All comments and responses submitted on draft and published documents are retained on file at CLSI and are available upon request. Get Involved Volunteer! Do you use CLSI documents in your workplace? Do you see room for improvement? Would you like to get involved in the revision process? Or maybe you see a need to develop a new document for an emerging technology? CLSI wants to hear from you. We are always looking for volunteers. By donating your time and talents to improve the standards that affect your own work, you will play an active role in improving public health across the globe. For additional information on committee participation or to submit comments, contact CLSI. Clinical and Laboratory Standards Institute 950 West Valley Road, Suite 2500 Wayne, PA USA P: F: standard@clsi.org
3 ISBN Vol. 28 No. 13 Replaces LIS1-A ISSN Vol. 23 No. 7 Specification for Low-Level Protocol to Transfer Messages Between Clinical Laboratory Instruments and Computer Systems; Approved Standard Second Edition Volume 28 Number 13 David Chou, MD Andrzej J. Knafel, PhD Charles D. Hawker, PhD, MBA, FACB Ed Heierman, PhD David A. Lacher, MD, MEd William Neeley, MD, FACP Eugene T. Reilly Richard S. Seaberg, MT(ASCP) Abstract CLSI document Specification for Low-Level Protocol to Transfer Messages Between Clinical Laboratory Instruments and Computer Systems; Approved Standard Second Edition describes the electronic transmission of digital information between clinical laboratory instruments (those that measure one or more parameters from one or multiple samples) and computer systems (those that are configured to accept instrument results for further processing, storage, reporting, or manipulation). Clinical and Laboratory Standards Institute (CLSI). Specification for Low-Level Protocol to Transfer Messages Between Clinical Laboratory Instruments and Computer Systems; Approved Standard Second Edition. CLSI document (ISBN ). Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania USA, The Clinical and Laboratory Standards Institute consensus process, which is the mechanism for moving a document through two or more levels of review by the health care community, is an ongoing process. Users should expect revised editions of any given document. Because rapid changes in technology may affect the procedures, methods, and protocols in a standard or guideline, users should replace outdated editions with the current editions of CLSI documents. Current editions are listed in the CLSI catalog and posted on our website at If your organization is not a member and would like to become one, and to request a copy of the catalog, contact us at: Telephone: ; Fax: ; customerservice@clsi.org; Website:
4 Number 13 Copyright 2008 Clinical and Laboratory Standards Institute. Except as stated below, any reproduction of content from a CLSI copyrighted standard, guideline, companion product, or other material requires express written consent from CLSI. All rights reserved. Interested parties may send permission requests to permissions@clsi.org. CLSI hereby grants permission to each individual member or purchaser to make a single reproduction of this publication for use in its laboratory procedure manual at a single site. To request permission to use this publication in any other manner, permissions@clsi.org. Suggested Citation CLSI. Specification for Low-Level Protocol to Transfer Messages Between Clinical Laboratory Instruments and Computer Systems; Approved Standard Second Edition. CLSI document. Wayne, PA: Clinical and Laboratory Standards Institute; Previous Edition: April 2003 Reaffirmed: September 2011 Archived: January 2017 ISBN ISSN ii
5 Volume 28 Contents Abstract... i Committee Membership... iii Foreword... vii 1 Scope Introduction Terminology Significance and Use Physical Layer for Serial Binary Data Exchange Overview Electrical Characteristics Mechanical Characteristics Data Link Layer for Serial Binary Data Exchange Overview Establishment Phase (Link Connection) Transfer Phase Termination Phase (Link Release) Error Recovery Restricted Message Characters Physical Layer for TCP/IP Data Exchange Overview Electrical Characteristics Mechanical Characteristics Data Link Layer for TCP/IP Data Exchange Overview Establishment Phase (Link Connection) Transfer Phase Termination Phase (Link Release) Error Recovery Restricted Message Characters References Appendix A. Mandatory Information Appendix B. Nonmandatory Information Summary of Delegate Comments and Area Committee Responses The Quality Management System Approach Related CLSI Reference Materials v
6 Volume 28 Foreword In 2001, ASTM Committee E31 decided to restructure its operations, with the intent of focusing on standards-development issues such as security, privacy, and the electronic health record. Part of the reorganization plan was to transfer responsibility for E31.13 standards to CLSI, then known as NCCLS. Following this transfer, nine standards (formerly ASTM E792; E1029; E1238; E1246; E1381; E1394; E1466; E1639; and E2118) were redesignated as CLSI/NCCLS standards LIS1 through LIS This collection of standards provides a wide variety of information relating to clinical laboratory computer systems. Some included documents are of general interest as reference sources; others represent specifications of primary importance to instrument manufacturers. LIS2 is a revision of the former ASTM E The Area Committee on Automation and Informatics has assumed responsibility for maintaining the documents and will revise or update each document in accord with the CLSI Administrative Procedures. The area committee prioritized LIS1-A as the second standard from this collection to be updated, incorporated into the CLSI document template, and advanced through the CLSI consensus process. The area committee will revise other documents in the series in a similar manner. With the transfer of the former ASTM standards, the Area Committee on Automation and Informatics has expanded its mission statement to include laboratory information systems. In the future, the area committee will develop additional standards addressing informatics issues, as well as issues related to the integration of patient clinical data. This document replaces the first edition of the approved guideline, LIS1-A, which was published in Several changes were made in this edition; among them, TCP/IP communication is now included (Sections 4.4 and 4.5) and the state diagram was replaced (see Appendix A) so it is consistent with the text of the document. The revisions in this edition of the LIS01 standard are also intended to delineate this document from its former ASTM edition. The title and text have been revised throughout to indicate that this standard applies to clinical laboratory instruments. The term computer has been replaced with the term information to better reflect the current terminology (ie, LIS) and the headings of Sections 6 and 8 have been changed to make them more specific. Key Words data link layer, physical layer, serial binary data exchange, TCP/IP data exchange vii
7 Volume 28 Specification for Low-Level Protocol to Transfer Messages Between Clinical Laboratory Instruments and Computer Systems; Approved Standard Second Edition 1 Scope This specification describes the electronic transmission of digital information between clinical laboratory instruments and computer systems. This specification addresses the low-level protocol used for both serial binary data exchange and TCP/IP data exchange. For message content in the interface between clinical instruments and computer systems, reference CLSI/NCCLS document LIS Introduction The clinical laboratory instruments under consideration are those that measure one or more parameters from one or more patient samples. Often they will be automated instruments that measure many parameters from many patient samples. The computer systems considered here are those that are configured to accept instrument results for further processing, storage, reporting, or manipulation. This instrument output may include patient results, quality control results, and other related information. Typically, the computer system will be a clinical laboratory information management system (CLIMS). The terminology of the International Organization for Standardization (ISO) Reference Model for Open Systems Interconnection (OSI) is generally followed in describing the communications protocol and services. 9 The electrical and mechanical connection between instrument and computer is described in the Physical Layer sections (see Sections 5 and 7). The methods for establishing communication, error detection, error recovery, and sending and receiving of messages are described in the Data Link Layer sections (see Sections 6 and 8). The data link layer interacts with higher layers in terms of sending and receiving messages, handles data link connection and release requests, and reports the data link status. 3 Terminology 3.1 receiver the device that responds to the sender and accepts the message. 3.2 sender the device that has a message to send and initiates the transmission process. 3.3 The parts of a communication between instrument and computer are identified by the following terms. The parts are hierarchical and are listed in order of most encompassing first session a total unit of communication activity, used in this standard to indicate the events starting with the establishment phase and ending with the termination phase, as described in subsequent sections message a collection of related information on a single topic, used here to mean all the identity, tests, and comments sent at one time; NOTE: When used with CLSI/NCCLS document LIS2, 1 this term means a record as defined by CLSI/NCCLS document LIS frame a subdivision of a message, used to allow periodic communication housekeeping, such as error checks and acknowledgments. Clinical and Laboratory Standards Institute. All rights reserved. 1
8 Volume 28 Related CLSI Reference Materials AUTO3-A LIS2-A2 Laboratory Automation: Communications with Automated Clinical Laboratory Systems, Instruments, Devices, and Information Systems; Approved Standard (2000). This document provides standards to facilitate accurate and timely electronic exchange of data and information between the automated laboratory elements. Specification for Transferring Information Between Clinical Laboratory Instruments and Information Systems; Approved Standard Second Edition (2004). This document covers the two-way digital transmission of remote requests and results between clinical laboratory instruments and information systems. Proposed-level documents are being advanced through the Clinical and Laboratory Standards Institute consensus process; therefore, readers should refer to the most current editions. Clinical and Laboratory Standards Institute. All rights reserved. 25
9 E PL Explore the Latest Offerings From CLSI! As we continue to set the global standard for quality in laboratory testing, we are adding products and programs to bring even more value to our members and customers. M By becoming a CLSI member, your laboratory will join 1,600+ other influential organizations all working together to further CLSI s efforts to improve health care outcomes. You can play an active role in raising global laboratory testing standards in your laboratory, and around the world. SA Find out which membership option is best for you at Find what your laboratory needs to succeed! CLSI U provides convenient, cost-effective continuing education and training resources to help you advance your professional development. We have a variety of easy-to-use, online educational resources that make elearning stress-free and convenient for you and your staff. See our current educational offerings at When laboratory testing quality is critical, standards are needed and there is no time to waste. eclipse Ultimate Access, our cloud-based online portal of the complete library of CLSI standards, makes it easy to quickly find the CLSI resources you need. Learn more and purchase eclipse at clsi.org/eclipse. For more information, visit today.
10 950 West Valley Road, Suite 2500, Wayne, PA USA P: Toll Free (US): F: ISBN E:
Computing Accreditation Commission Version 2.0 CRITERIA FOR ACCREDITING COMPUTING PROGRAMS
 Computing Accreditation Commission Version 2.0 CRITERIA FOR ACCREDITING COMPUTING PROGRAMS Optional for Reviews During the 2018-2019 Accreditation Cycle Mandatory for Reviews During the 2019-2020 Accreditation
Computing Accreditation Commission Version 2.0 CRITERIA FOR ACCREDITING COMPUTING PROGRAMS Optional for Reviews During the 2018-2019 Accreditation Cycle Mandatory for Reviews During the 2019-2020 Accreditation
CLSI Documents Referenced to The Joint Commission Laboratory Accreditation Standards Chapters July 2011
 CLSI Documents Referenced to The Joint Commission Laboratory Accreditation Standards Chapters July 2011 940 West Valley Road Suite 1400 Wayne, PA 19087 610.688.0100 F: 610.688.0700 customerservice@clsi.org
CLSI Documents Referenced to The Joint Commission Laboratory Accreditation Standards Chapters July 2011 940 West Valley Road Suite 1400 Wayne, PA 19087 610.688.0100 F: 610.688.0700 customerservice@clsi.org
January 16, Re: Request for Comment: Data Access and Data Sharing Policy. Dear Dr. Selby:
 Dr. Joe V. Selby, MD, MPH Executive Director Patient-Centered Outcomes Research Institute 1828 L Street, NW, Suite 900 Washington, DC 20036 Submitted electronically at: http://www.pcori.org/webform/data-access-and-data-sharing-policypublic-comment
Dr. Joe V. Selby, MD, MPH Executive Director Patient-Centered Outcomes Research Institute 1828 L Street, NW, Suite 900 Washington, DC 20036 Submitted electronically at: http://www.pcori.org/webform/data-access-and-data-sharing-policypublic-comment
Electronic Service Provider Standard
 Electronic Service Provider Standard Version: 1.6 Document ID: 3538 Copyright Notice Copyright 2018, ehealth Ontario All rights reserved No part of this document may be reproduced in any form, including
Electronic Service Provider Standard Version: 1.6 Document ID: 3538 Copyright Notice Copyright 2018, ehealth Ontario All rights reserved No part of this document may be reproduced in any form, including
CDISC Operating Procedure COP-001 Standards Development
 CDISC Operating Procedure COP-001 Revision History Date Revision Description Author 4 February 2002 1.0 Revisions/approval Shirley Williams/ Board of Directors 1 January 2006 1.1 Revisions required to
CDISC Operating Procedure COP-001 Revision History Date Revision Description Author 4 February 2002 1.0 Revisions/approval Shirley Williams/ Board of Directors 1 January 2006 1.1 Revisions required to
Document Number: HITSP 08 N 378 Date: December 17, 2008 Report from the HITSP Education, Communication and Outreach (HITSP-ECO) Committee
 0 Document Number: HITSP 08 N 378 Date: December 17, 2008 Report from the HITSP Education, Communication and Outreach (HITSP-ECO) Committee Co-Chairs: Walter G. Suarez, MD, Institute for HIPAA/HIT Education
0 Document Number: HITSP 08 N 378 Date: December 17, 2008 Report from the HITSP Education, Communication and Outreach (HITSP-ECO) Committee Co-Chairs: Walter G. Suarez, MD, Institute for HIPAA/HIT Education
For Facilitator Use Only. B3. Conformity Assessment: Roles and Responsibilities (Perfect score: 30 points)
 For Facilitator Use Only B3. Conformity Assessment: Roles and Responsibilities (Perfect score: 30 points) 1. a. Council on Standards and Certification b. Board on Conformity Assessment c. Standards Committees
For Facilitator Use Only B3. Conformity Assessment: Roles and Responsibilities (Perfect score: 30 points) 1. a. Council on Standards and Certification b. Board on Conformity Assessment c. Standards Committees
Overview of conformity assessment programs and ASTM International s related activities
 Overview of conformity assessment programs and ASTM International s related activities African Organization for Standardization 21 June 2016 Tim Brooke Vice President Certification Training, and Proficiency
Overview of conformity assessment programs and ASTM International s related activities African Organization for Standardization 21 June 2016 Tim Brooke Vice President Certification Training, and Proficiency
OpenChain Specification Version 1.3 (DRAFT)
 OpenChain Specification Version 1.3 (DRAFT) 2018.10.14 DRAFT: This is the draft of the next version 1.3 of the OpenChain specification. Recommended changes to be made over the current released version
OpenChain Specification Version 1.3 (DRAFT) 2018.10.14 DRAFT: This is the draft of the next version 1.3 of the OpenChain specification. Recommended changes to be made over the current released version
University of Pennsylvania Institutional Review Board
 Reliance Agreement Guidance Creating Consent Templates This document provides step by step guidance on how to convert the Penn IRB approved consent form into a consent form template that can be shared
Reliance Agreement Guidance Creating Consent Templates This document provides step by step guidance on how to convert the Penn IRB approved consent form into a consent form template that can be shared
Australian Standard. Records Management. Part 1: General AS ISO ISO
 AS ISO 15489.1 2002 ISO 15489-1 AS ISO 15489.1 Australian Standard Records Management Part 1: General [ISO title: Information and documentation Records management Part 1: General] This Australian Standard
AS ISO 15489.1 2002 ISO 15489-1 AS ISO 15489.1 Australian Standard Records Management Part 1: General [ISO title: Information and documentation Records management Part 1: General] This Australian Standard
SLAS Special Interest Group Charter Application
 SLAS Special Interest Group Charter Application SLAS is an international community of more than 15,000 individual scientists, engineers, researchers, technologists and others from academic, government
SLAS Special Interest Group Charter Application SLAS is an international community of more than 15,000 individual scientists, engineers, researchers, technologists and others from academic, government
Therapy Provider Portal. User Guide
 Therapy Provider Portal User Guide Page 2 of 16 UCare User Guide V1.7 Table of Contents I. Introduction...3 About HSM Therapy Management... 4 Terms of Use... 4 Contact Information... 6 II. Using the Therapy
Therapy Provider Portal User Guide Page 2 of 16 UCare User Guide V1.7 Table of Contents I. Introduction...3 About HSM Therapy Management... 4 Terms of Use... 4 Contact Information... 6 II. Using the Therapy
Frequently Asked Questions
 Frequently Asked Questions ISO 15189 Accreditation Program cap.org Contents ISO and the International Organization for Standardization What does ISO stand for? (page 3) What is the International Organization
Frequently Asked Questions ISO 15189 Accreditation Program cap.org Contents ISO and the International Organization for Standardization What does ISO stand for? (page 3) What is the International Organization
Australian Standard. Records Management. Part 2: Guidelines AS ISO ISO TR
 AS ISO 15489.2 2002 ISO TR 15489-2 AS ISO 15489.2 Australian Standard Records Management Part 2: Guidelines [ISO title: Information and documentation Records management Part 2: Guidelines] This Australian
AS ISO 15489.2 2002 ISO TR 15489-2 AS ISO 15489.2 Australian Standard Records Management Part 2: Guidelines [ISO title: Information and documentation Records management Part 2: Guidelines] This Australian
The IDN Variant TLD Program: Updated Program Plan 23 August 2012
 The IDN Variant TLD Program: Updated Program Plan 23 August 2012 Table of Contents Project Background... 2 The IDN Variant TLD Program... 2 Revised Program Plan, Projects and Timeline:... 3 Communication
The IDN Variant TLD Program: Updated Program Plan 23 August 2012 Table of Contents Project Background... 2 The IDN Variant TLD Program... 2 Revised Program Plan, Projects and Timeline:... 3 Communication
STRATEGIC PLAN
 STRATEGIC PLAN 2013-2018 In an era of growing demand for IT services, it is imperative that strong guiding principles are followed that will allow for the fulfillment of the Division of Information Technology
STRATEGIC PLAN 2013-2018 In an era of growing demand for IT services, it is imperative that strong guiding principles are followed that will allow for the fulfillment of the Division of Information Technology
Systems and software engineering Requirements for managers of information for users of systems, software, and services
 This is a preview - click here to buy the full publication INTERNATIONAL STANDARD ISO/IEC/ IEEE 26511 Second edition 2018-12 Systems and software engineering Requirements for managers of information for
This is a preview - click here to buy the full publication INTERNATIONAL STANDARD ISO/IEC/ IEEE 26511 Second edition 2018-12 Systems and software engineering Requirements for managers of information for
OIX DDP. Open-IX Document Development Process draft July 2017
 OIX DDP Open-IX Document Development Process draft 04 11 July 2017 Table 1 - Version History Version Date Author Description d01 7 May 2017 Chris Grundemann Initial Draft d02 21 May 2017 Chris Grundemann
OIX DDP Open-IX Document Development Process draft 04 11 July 2017 Table 1 - Version History Version Date Author Description d01 7 May 2017 Chris Grundemann Initial Draft d02 21 May 2017 Chris Grundemann
SAMPLE REPORT. Business Continuity Gap Analysis Report. Prepared for XYZ Business by CSC Business Continuity Services Date: xx/xx/xxxx
 SAMPLE REPORT Business Continuity Gap Analysis Report Prepared for XYZ Business by CSC Business Continuity Services Date: xx/xx/xxxx COMMERCIAL-IN-CONFIDENCE PAGE 1 OF 11 Contact Details CSC Contacts CSC
SAMPLE REPORT Business Continuity Gap Analysis Report Prepared for XYZ Business by CSC Business Continuity Services Date: xx/xx/xxxx COMMERCIAL-IN-CONFIDENCE PAGE 1 OF 11 Contact Details CSC Contacts CSC
WASHINGTON UNIVERSITY HIPAA Privacy Policy # 7. Appropriate Methods of Communicating Protected Health Information
 WASHINGTON UNIVERSITY HIPAA Privacy Policy # 7 Appropriate Methods of Communicating Protected Health Information Statement of Policy Washington University and its member organizations (collectively, Washington
WASHINGTON UNIVERSITY HIPAA Privacy Policy # 7 Appropriate Methods of Communicating Protected Health Information Statement of Policy Washington University and its member organizations (collectively, Washington
Architecture and Standards Development Lifecycle
 Architecture and Standards Development Lifecycle Architecture and Standards Branch Author: Architecture and Standards Branch Date Created: April 2, 2008 Last Update: July 22, 2008 Version: 1.0 ~ This Page
Architecture and Standards Development Lifecycle Architecture and Standards Branch Author: Architecture and Standards Branch Date Created: April 2, 2008 Last Update: July 22, 2008 Version: 1.0 ~ This Page
ENCePP Code of Conduct Implementation Guidance for Sharing of ENCePP Study Data
 EMA/409316/2010 Revision 2, dated 14 July 2016 European Network of Centres for Pharmacoepidemiology and Pharmacovigilance ENCePP Code of Conduct Implementation Guidance for Sharing of ENCePP Study Data
EMA/409316/2010 Revision 2, dated 14 July 2016 European Network of Centres for Pharmacoepidemiology and Pharmacovigilance ENCePP Code of Conduct Implementation Guidance for Sharing of ENCePP Study Data
A S ISO Records Management Part 1: General
 AS ISO 15489.1 2002 ISO 15489-1 AS ISO 15489.1 Australian Standard Records Management Part 1: General [ISO title: Information and documentation Records management Part 1: General] This Australian Standard
AS ISO 15489.1 2002 ISO 15489-1 AS ISO 15489.1 Australian Standard Records Management Part 1: General [ISO title: Information and documentation Records management Part 1: General] This Australian Standard
Standards Designation and Organization Manual
 Standards Designation and Organization Manual InfoComm International Standards Program Ver. 2014-1 April 28, 2014 Issued by: Joseph Bocchiaro III, Ph.D., CStd., CTS-D, CTS-I, ISF-C Director of Standards
Standards Designation and Organization Manual InfoComm International Standards Program Ver. 2014-1 April 28, 2014 Issued by: Joseph Bocchiaro III, Ph.D., CStd., CTS-D, CTS-I, ISF-C Director of Standards
Survey on Patient Safety Culture Database Data Use Agreement
 Survey on Patient Safety Culture Database Data Use Agreement Instructions 1. Westat has pre-signed this Data Use Agreement (DUA) in its current form. Any changes or modifications to the DUA other than
Survey on Patient Safety Culture Database Data Use Agreement Instructions 1. Westat has pre-signed this Data Use Agreement (DUA) in its current form. Any changes or modifications to the DUA other than
Privacy Policy- ADDO Worldwide LLC LAST UPDATED: September 9, 2014
 Privacy Policy- ADDO Worldwide LLC LAST UPDATED: September 9, 2014 1. Introduction Chick-fil-A Leader Academy is owned and operated by ADDO Worldwide, LLC ( ADDO Worldwide, LLC, we, us, our ). We at ADDO
Privacy Policy- ADDO Worldwide LLC LAST UPDATED: September 9, 2014 1. Introduction Chick-fil-A Leader Academy is owned and operated by ADDO Worldwide, LLC ( ADDO Worldwide, LLC, we, us, our ). We at ADDO
Security and Privacy Governance Program Guidelines
 Security and Privacy Governance Program Guidelines Effective Security and Privacy Programs start with attention to Governance. Governance refers to the roles and responsibilities that are established by
Security and Privacy Governance Program Guidelines Effective Security and Privacy Programs start with attention to Governance. Governance refers to the roles and responsibilities that are established by
EHR SECURITY POLICIES & SECURITY SITE ASSESSMENT OVERVIEW WEBINAR. For Viewer Sites
 EHR SECURITY POLICIES & SECURITY SITE ASSESSMENT OVERVIEW WEBINAR For Viewer Sites Agenda 1 Introduction and EHR Security Policies Background 2 EHR Security Policy Overview 3 EHR Security Policy Assessment
EHR SECURITY POLICIES & SECURITY SITE ASSESSMENT OVERVIEW WEBINAR For Viewer Sites Agenda 1 Introduction and EHR Security Policies Background 2 EHR Security Policy Overview 3 EHR Security Policy Assessment
The World s Leading Organization for Electrical Power Systems Professionals and Industry Leaders
 The World s Leading Organization for Electrical Power Systems Professionals and Industry Leaders IMPROVING THE POWER SYSTEMS OF TODAY AND TOMORROW THROUGH INTERNATIONAL COLLABORATION CIGRÉ, the International
The World s Leading Organization for Electrical Power Systems Professionals and Industry Leaders IMPROVING THE POWER SYSTEMS OF TODAY AND TOMORROW THROUGH INTERNATIONAL COLLABORATION CIGRÉ, the International
Frequently Asked Questions
 HFMA s 2015 Realigned CHFP Program Frequently Asked Questions New CHFP Q: I have heard that the certification program is being revised. What are the changes? How will the exam(s) be structured? A: The
HFMA s 2015 Realigned CHFP Program Frequently Asked Questions New CHFP Q: I have heard that the certification program is being revised. What are the changes? How will the exam(s) be structured? A: The
Invitation to Participate
 Invitation to Participate 2009 For the lead agency participation in New York State Quality Rating and Improvement System -- QUALITYstarsNY field test Page 1 of 7 101 West 31 st Street, 7th Floor New York,
Invitation to Participate 2009 For the lead agency participation in New York State Quality Rating and Improvement System -- QUALITYstarsNY field test Page 1 of 7 101 West 31 st Street, 7th Floor New York,
This is a preview - click here to buy the full publication PUBLICLY AVAILABLE SPECIFICATION. Pre-Standard
 PUBLICLY AVAILABLE SPECIFICATION Pre-Standard IEC PAS 61512-3 First edition 2004-11 Batch control Part 3: General and site recipe models and representation Reference number IEC/PAS 61512-3:2004(E) AMERICAN
PUBLICLY AVAILABLE SPECIFICATION Pre-Standard IEC PAS 61512-3 First edition 2004-11 Batch control Part 3: General and site recipe models and representation Reference number IEC/PAS 61512-3:2004(E) AMERICAN
CRITERIA FOR ACCREDITING COMPUTING PROGRAMS
 CRITERIA FOR ACCREDITING COMPUTING PROGRAMS Effective for Reviews During the 2014-2015 Accreditation Cycle Incorporates all changes approved by the ABET Board of Directors as of October 26, 2013 Computing
CRITERIA FOR ACCREDITING COMPUTING PROGRAMS Effective for Reviews During the 2014-2015 Accreditation Cycle Incorporates all changes approved by the ABET Board of Directors as of October 26, 2013 Computing
INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD IEC 60870-5-6 First edition 2006-03 Telecontrol equipment and systems Part 5-6: Guidelines for conformance testing for the IEC 60870-5 companion standards IEC 2006 Copyright - all
INTERNATIONAL STANDARD IEC 60870-5-6 First edition 2006-03 Telecontrol equipment and systems Part 5-6: Guidelines for conformance testing for the IEC 60870-5 companion standards IEC 2006 Copyright - all
ACCREDITATION COMMISSION FOR CONFORMITY ASSESSMENT BODIES
 ACCREDITATION COMMISSION FOR CONFORMITY ASSESSMENT BODIES ACCREDITATION SCHEME MANUAL Document Title: Document Number: Various Accreditation Schemes ACCAB-ASM-7.0 CONTROLLED COPY Revision Number Revision
ACCREDITATION COMMISSION FOR CONFORMITY ASSESSMENT BODIES ACCREDITATION SCHEME MANUAL Document Title: Document Number: Various Accreditation Schemes ACCAB-ASM-7.0 CONTROLLED COPY Revision Number Revision
The 2016 TNI Laboratory Accreditation Standard. August 4, 2016
 The 2016 TNI Laboratory Accreditation Standard August 4, 2016 TNI s Consensus Standards Development Program has released a new consensus standard for the accreditation of environmental laboratories, Management
The 2016 TNI Laboratory Accreditation Standard August 4, 2016 TNI s Consensus Standards Development Program has released a new consensus standard for the accreditation of environmental laboratories, Management
Information Technology (CCHIT): Report on Activities and Progress
 Certification Commission for Healthcare Information Technology Certification Commission for Healthcare Information Technology (CCHIT): Report on Activities and Progress Mark Leavitt, MD, PhD Chair, CCHIT
Certification Commission for Healthcare Information Technology Certification Commission for Healthcare Information Technology (CCHIT): Report on Activities and Progress Mark Leavitt, MD, PhD Chair, CCHIT
GENERAL OPERATIONS MANUAL FOR ASTM PERSONNEL CERTIFICATE PROGRAMS
 GENERAL OPERATIONS MANUAL FOR ASTM PERSONNEL CERTIFICATE PROGRAMS INTERNATIONAL Standards Worldwide Issued September 19 th, 2012 Updates Approved by CCP on March 23 rd, 2012 Version 5.0 BY AMERICAN SOCIETY
GENERAL OPERATIONS MANUAL FOR ASTM PERSONNEL CERTIFICATE PROGRAMS INTERNATIONAL Standards Worldwide Issued September 19 th, 2012 Updates Approved by CCP on March 23 rd, 2012 Version 5.0 BY AMERICAN SOCIETY
Ensuring Privacy and Security of Health Information Exchange in Pennsylvania
 Ensuring Privacy and Security of Health Information Exchange in Pennsylvania The Pennsylvania ehealth Initiative in collaboration with the Pennsylvania ehealth Partnership Authority Introduction The Pennsylvania
Ensuring Privacy and Security of Health Information Exchange in Pennsylvania The Pennsylvania ehealth Initiative in collaboration with the Pennsylvania ehealth Partnership Authority Introduction The Pennsylvania
Manager, Infrastructure Services. Position Number Community Division/Region Yellowknife Technology Service Centre
 IDENTIFICATION Department Position Title Infrastructure Manager, Infrastructure Services Position Number Community Division/Region 32-11488 Yellowknife Technology Service Centre PURPOSE OF THE POSITION
IDENTIFICATION Department Position Title Infrastructure Manager, Infrastructure Services Position Number Community Division/Region 32-11488 Yellowknife Technology Service Centre PURPOSE OF THE POSITION
Scientific Working Group on Digital Evidence
 SWGDE Requirements for Report Writing in Digital and Multimedia Forensics Disclaimer: As a condition to the use of this document and the information contained therein, the SWGDE requests notification by
SWGDE Requirements for Report Writing in Digital and Multimedia Forensics Disclaimer: As a condition to the use of this document and the information contained therein, the SWGDE requests notification by
ACCREDITATION COMMISSION FOR CONFORMITY ASSESSMENT BODIES
 ACCREDITATION COMMISSION FOR CONFORMITY ASSESSMENT BODIES ACCREDITATION SCHEME MANUAL Document Title: Document Number: Various Accreditation Schemes ACCAB-ASM-7.0 CONTROLLED COPY Revision Number Revision
ACCREDITATION COMMISSION FOR CONFORMITY ASSESSMENT BODIES ACCREDITATION SCHEME MANUAL Document Title: Document Number: Various Accreditation Schemes ACCAB-ASM-7.0 CONTROLLED COPY Revision Number Revision
Russ Housley 21 June 2015
 Introduction to the Internet Engineering Task Force Russ Housley 21 June 2015 Internet Engineering Task Force We make the net work The mission of the IETF is to produce high quality, relevant technical
Introduction to the Internet Engineering Task Force Russ Housley 21 June 2015 Internet Engineering Task Force We make the net work The mission of the IETF is to produce high quality, relevant technical
Financial Planning Institute of Southern Africa SETTING THE STANDARD. Continuous Professional Development (Cpd) Policy
 FPI FPI Financial Planning Institute of Southern Africa SETTING THE STANDARD Continuous Professional Development (Cpd) Policy Table of Contents Definitions 3-4 Introduction 4 Primary Responsibility 5 Mandatory
FPI FPI Financial Planning Institute of Southern Africa SETTING THE STANDARD Continuous Professional Development (Cpd) Policy Table of Contents Definitions 3-4 Introduction 4 Primary Responsibility 5 Mandatory
March 20, Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852
 701 Pennsylvania Avenue, NW Suite 800 Washington, D.C. 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org March 20, 2017 Division of Dockets Management (HFA-305) Food and Drug Administration
701 Pennsylvania Avenue, NW Suite 800 Washington, D.C. 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org March 20, 2017 Division of Dockets Management (HFA-305) Food and Drug Administration
Views on the Framework for Improving Critical Infrastructure Cybersecurity
 This document is scheduled to be published in the Federal Register on 12/11/2015 and available online at http://federalregister.gov/a/2015-31217, and on FDsys.gov Billing Code: 3510-13 DEPARTMENT OF COMMERCE
This document is scheduled to be published in the Federal Register on 12/11/2015 and available online at http://federalregister.gov/a/2015-31217, and on FDsys.gov Billing Code: 3510-13 DEPARTMENT OF COMMERCE
Pre-Standard PUBLICLY AVAILABLE SPECIFICATION IEC PAS Batch control. Part 3: General and site recipe models and representation
 PUBLICLY AVAILABLE SPECIFICATION Pre-Standard IEC PAS 61512-3 First edition 2004-11 Batch control Part 3: General and site recipe models and representation Reference number IEC/PAS 61512-3:2004(E) Publication
PUBLICLY AVAILABLE SPECIFICATION Pre-Standard IEC PAS 61512-3 First edition 2004-11 Batch control Part 3: General and site recipe models and representation Reference number IEC/PAS 61512-3:2004(E) Publication
American National Standard for Electric Lamps Light-Emitting Diode Package Specification Sheet for General Illumination Applications
 American National Standard for Electric Lamps Light-Emitting Diode Package Specification Sheet for General Illumination Applications Secretariat: National Electrical Manufacturers Association Approved
American National Standard for Electric Lamps Light-Emitting Diode Package Specification Sheet for General Illumination Applications Secretariat: National Electrical Manufacturers Association Approved
Traditional Certification Application (Please type online and print finished copy) Applying for:
 International Association of Eating Disorders Professionals PO Box 1295 / Pekin, IL 61555-1295 Tel. (800) 800-8126 / Fax (800) 800-8126 Email: info@iaedp.com / Website: www.iaedp.com Traditional Certification
International Association of Eating Disorders Professionals PO Box 1295 / Pekin, IL 61555-1295 Tel. (800) 800-8126 / Fax (800) 800-8126 Email: info@iaedp.com / Website: www.iaedp.com Traditional Certification
FSC STANDARD. Standard for Multi-site Certification of Chain of Custody Operations. FSC-STD (Version 1-0) EN
 FOREST STEWARDSHIP COUNCIL INTERNATIONAL CENTER FSC STANDARD Standard for Multi-site Certification of Chain of Custody Operations FSC-STD-40-003 (Version 1-0) EN 2007 Forest Stewardship Council A.C. All
FOREST STEWARDSHIP COUNCIL INTERNATIONAL CENTER FSC STANDARD Standard for Multi-site Certification of Chain of Custody Operations FSC-STD-40-003 (Version 1-0) EN 2007 Forest Stewardship Council A.C. All
Velos ecompliance. Researcher User Manual
 Velos ecompliance Researcher User Manual Table of Contents Disclaimer... 4 ecompliance Overview... 5 Contacts... 6 Administrator... 6 Super Users... 6 IT/Technical Support... 6 Quick Help... 7 Getting
Velos ecompliance Researcher User Manual Table of Contents Disclaimer... 4 ecompliance Overview... 5 Contacts... 6 Administrator... 6 Super Users... 6 IT/Technical Support... 6 Quick Help... 7 Getting
Last updated January 29, Approved by ECC March 3, 2015
 Last updated January 29, 2015 Approved by ECC March 3, 2015 ASHRAE Electronic Communications Strategic Plan Electronic Communications Committee (ECC) January 2015 Electronic Communications Committee Strategic
Last updated January 29, 2015 Approved by ECC March 3, 2015 ASHRAE Electronic Communications Strategic Plan Electronic Communications Committee (ECC) January 2015 Electronic Communications Committee Strategic
INTERNATIONAL STANDARD
 IEC 61158-3-11 INTERNATIONAL STANDARD Edition 1.0 2007-12 Industrial communication networks Fieldbus specifications Part 3-11: Data-link layer service definition Type 11 elements INTERNATIONAL ELECTROTECHNICAL
IEC 61158-3-11 INTERNATIONAL STANDARD Edition 1.0 2007-12 Industrial communication networks Fieldbus specifications Part 3-11: Data-link layer service definition Type 11 elements INTERNATIONAL ELECTROTECHNICAL
CONTINUING PROFESSIONAL DEVELOPMENT RULES
 Independent Objective Authoritative The home for property professionals in Australia Australian Property Institute Limited CONTINUING PROFESSIONAL DEVELOPMENT RULES Reference Continuing Professional Development
Independent Objective Authoritative The home for property professionals in Australia Australian Property Institute Limited CONTINUING PROFESSIONAL DEVELOPMENT RULES Reference Continuing Professional Development
IVOA Document Standards Version 0.1
 International Virtual Observatory Alliance IVOA Document Standards Version 0.1 IVOA Working Draft 09 July 2003 This version: http://www.ivoa.net/documents/wd/docstandard/documentstandards-20030709.html
International Virtual Observatory Alliance IVOA Document Standards Version 0.1 IVOA Working Draft 09 July 2003 This version: http://www.ivoa.net/documents/wd/docstandard/documentstandards-20030709.html
Client Services Procedure Manual
 Procedure: 85.00 Subject: Administration and Promotion of the Health and Safety Learning Series The Health and Safety Learning Series is a program designed and delivered by staff at WorkplaceNL to increase
Procedure: 85.00 Subject: Administration and Promotion of the Health and Safety Learning Series The Health and Safety Learning Series is a program designed and delivered by staff at WorkplaceNL to increase
FOUNDED GOAL of New ORGANIZATION. CLEAR Annual Educational Conference Getting the Most Out of CLEAR. St. Louis October 3-5, 2013
 Deanna Williams FOUNDED 1980 In 1980, a group of professional and occupational regulators and private sector representatives met in New Orleans to discuss their need to share information. GOAL of New ORGANIZATION
Deanna Williams FOUNDED 1980 In 1980, a group of professional and occupational regulators and private sector representatives met in New Orleans to discuss their need to share information. GOAL of New ORGANIZATION
Data Quality Project Open Industry Meeting. Steven Wilson, BSI. What can we do to find lost customers and why do we want to do it?
 Steven Wilson, BSI What can we do to find lost customers and why do we want to do it? PAS standard overview for TISA 5 th September 2013 Steven Wilson BSI Standards Solutions 020 8996 6358 steven.wilson@bsigroup.com
Steven Wilson, BSI What can we do to find lost customers and why do we want to do it? PAS standard overview for TISA 5 th September 2013 Steven Wilson BSI Standards Solutions 020 8996 6358 steven.wilson@bsigroup.com
Image Exchange Portal
 Image Exchange Portal Approved By: Date Approved: 25 th January 2011 Trust Reference: C132/2016 Version: Supersedes: Author / Originator(s): Name of Responsible Committee/Individual: Imaging Clinical Business
Image Exchange Portal Approved By: Date Approved: 25 th January 2011 Trust Reference: C132/2016 Version: Supersedes: Author / Originator(s): Name of Responsible Committee/Individual: Imaging Clinical Business
Agenda and General Information
 Agenda and General Information IEEE P802.3bt 4-Pair Power over Ethernet (4PPoE) Chad Jones Cisco Systems Geneva, CH January 24, 2018 Page 1 Agenda Welcome and Introductions (Secretary) Approve Agenda Approve
Agenda and General Information IEEE P802.3bt 4-Pair Power over Ethernet (4PPoE) Chad Jones Cisco Systems Geneva, CH January 24, 2018 Page 1 Agenda Welcome and Introductions (Secretary) Approve Agenda Approve
NOTICE. (Formulated under the cognizance of the CTA R7 Home Networks Committee.)
 ANSI/CTA Standard Control Networking Protocol Specification Part 5: Implementation Application Layer Guidelines ANSI/CTA-709.5 (Formerly ANSI/CEA-709.5) September 2015 NOTICE Consumer Technology Association
ANSI/CTA Standard Control Networking Protocol Specification Part 5: Implementation Application Layer Guidelines ANSI/CTA-709.5 (Formerly ANSI/CEA-709.5) September 2015 NOTICE Consumer Technology Association
PROPOSED DRAFT FOR TRIAL USE AND DISCUSSION ONLY secretariat PROPOSED DRAFT AES24-2-TU 99/02/2818:41
 STANDARDS The AES Standards Committee is the organization responsible for the standards program of the Audio Engineering Society. It publishes technical standards, information documents and technical reports.
STANDARDS The AES Standards Committee is the organization responsible for the standards program of the Audio Engineering Society. It publishes technical standards, information documents and technical reports.
NEMA Standards Publication AB Establishing Levels of Selective Coordination for Low Voltage Circuit Breakers
 NEMA Standards Publication AB 5-2011 Establishing Levels of Selective Coordination for Low Voltage Circuit Breakers Published by: National Electrical Manufacturers Association 1300 North 17th Street, Suite
NEMA Standards Publication AB 5-2011 Establishing Levels of Selective Coordination for Low Voltage Circuit Breakers Published by: National Electrical Manufacturers Association 1300 North 17th Street, Suite
Government of Ontario IT Standard (GO ITS)
 Government of Ontario IT Standard (GO ITS) GO-ITS Number 56.3 Information Modeling Standard Version # : 1.5 Status: Approved Prepared under the delegated authority of the Management Board of Cabinet Queen's
Government of Ontario IT Standard (GO ITS) GO-ITS Number 56.3 Information Modeling Standard Version # : 1.5 Status: Approved Prepared under the delegated authority of the Management Board of Cabinet Queen's
CONTINUING PROFESSIONAL DEVELOPMENT SCHEME (CPD) FOR AATSL MEMBERS
 CONTINUING PROFESSIONAL DEVELOPMENT SCHEME (CPD) FOR AATSL MEMBERS The members of the Association of Accounting Technicians of Sri Lanka (AATSL) are hereby informed that Continuing Professional Development
CONTINUING PROFESSIONAL DEVELOPMENT SCHEME (CPD) FOR AATSL MEMBERS The members of the Association of Accounting Technicians of Sri Lanka (AATSL) are hereby informed that Continuing Professional Development
ANSI-Accredited U.S. TAGs to ISO 2019 Procedural Compliance Form & 2018 TAG Annual Report 2019 ANSI International Procedures
 ANSI-Accredited U.S. TAGs to ISO 2019 Procedural Compliance Form & 2018 TAG Annual Report 2019 ANSI International Procedures (www.ansi.org/internationalprocedures) Agenda ANSI Staff available to help:
ANSI-Accredited U.S. TAGs to ISO 2019 Procedural Compliance Form & 2018 TAG Annual Report 2019 ANSI International Procedures (www.ansi.org/internationalprocedures) Agenda ANSI Staff available to help:
The Open Group Standards Process Part 1 - Overview. Copyright 2015 The Open Group
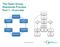 The Open Group Standards Process Part 1 - Overview Agenda q This presentation provides an overview of The Open Group Standards Process Purpose Definition: Open Group Standard Principles Processes Supporting
The Open Group Standards Process Part 1 - Overview Agenda q This presentation provides an overview of The Open Group Standards Process Purpose Definition: Open Group Standard Principles Processes Supporting
Chapter 10: Regulatory Documentation
 Table of Contents Chapter 10: Regulatory Documentation... 10-1 10.1 Regulatory Requirements:... 10-1 10.2 Ongoing Regulatory Documents:... 10-4 10.3 After study completion or termination of the trial...
Table of Contents Chapter 10: Regulatory Documentation... 10-1 10.1 Regulatory Requirements:... 10-1 10.2 Ongoing Regulatory Documents:... 10-4 10.3 After study completion or termination of the trial...
Subject: University Information Technology Resource Security Policy: OUTDATED
 Policy 1-18 Rev. 2 Date: September 7, 2006 Back to Index Subject: University Information Technology Resource Security Policy: I. PURPOSE II. University Information Technology Resources are at risk from
Policy 1-18 Rev. 2 Date: September 7, 2006 Back to Index Subject: University Information Technology Resource Security Policy: I. PURPOSE II. University Information Technology Resources are at risk from
CONTENTS Program Description... 1 Scope... 1 Course Requirements... 1 Fees... 1 Program Resources... 1 Assessment
 TRUE Advisor Certificate Program Handbook January 2018 CONTENTS Program Description... 1 Scope... 1 Course Requirements... 1 Fees... 1 Program Resources... 1 Assessment... 1 Passing Score... 2 Assessment
TRUE Advisor Certificate Program Handbook January 2018 CONTENTS Program Description... 1 Scope... 1 Course Requirements... 1 Fees... 1 Program Resources... 1 Assessment... 1 Passing Score... 2 Assessment
Implementing the EMS Education Agenda for the Future: A Systems Approach. A Priority for the NASEMSO
 Implementing the EMS Education Agenda for the Future: A Systems Approach A Priority for the NASEMSO Objectives Review the development of the EMS Education Agenda for the Future (EAoF) Identify the elements
Implementing the EMS Education Agenda for the Future: A Systems Approach A Priority for the NASEMSO Objectives Review the development of the EMS Education Agenda for the Future (EAoF) Identify the elements
INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD IEC 61850-10 First edition 2005-05 Communication networks and systems in substations Part 10: Conformance testing Reference number IEC 61850-10:2005(E) Publication numbering As from
INTERNATIONAL STANDARD IEC 61850-10 First edition 2005-05 Communication networks and systems in substations Part 10: Conformance testing Reference number IEC 61850-10:2005(E) Publication numbering As from
WORLD TELECOMMUNICATION STANDARDIZATION ASSEMBLY Hammamet, 25 October 3 November 2016
 I n t e r n a t i o n a l T e l e c o m m u n i c a t i o n U n i o n ITU-T TELECOMMUNICATION STANDARDIZATION SECTOR OF ITU WORLD TELECOMMUNICATION STANDARDIZATION ASSEMBLY Hammamet, 25 October 3 November
I n t e r n a t i o n a l T e l e c o m m u n i c a t i o n U n i o n ITU-T TELECOMMUNICATION STANDARDIZATION SECTOR OF ITU WORLD TELECOMMUNICATION STANDARDIZATION ASSEMBLY Hammamet, 25 October 3 November
Pennsylvania s HIE Journey
 Pennsylvania s HIE Journey Alix Goss, Executive Director Pennsylvania ehealth Partnership Authority William Buddy Gillespie Director Healthcare Solutions DSS What is HIE? Health Information Exchange puts
Pennsylvania s HIE Journey Alix Goss, Executive Director Pennsylvania ehealth Partnership Authority William Buddy Gillespie Director Healthcare Solutions DSS What is HIE? Health Information Exchange puts
Certification program PCWU-3
 The certification program of utility products type 3 of the certification program according to PN-EN ISO/IEC 17067 Number: Page: 1 z 8 MS-0013527 Is valid from: 01.03.2016 Prepared: Tomasz Marcinek Approved:
The certification program of utility products type 3 of the certification program according to PN-EN ISO/IEC 17067 Number: Page: 1 z 8 MS-0013527 Is valid from: 01.03.2016 Prepared: Tomasz Marcinek Approved:
Guide to Adjunct and Part Time Clinical Re- Appointments
 Guide to Adjunct and Part Time Clinical Re- Appointments Using Web Forms and LaserFiche Prepared by: Discovery Commons February 2018 Version 2.0 Table of Contents Guide to Adjunct and Part Time Clinical
Guide to Adjunct and Part Time Clinical Re- Appointments Using Web Forms and LaserFiche Prepared by: Discovery Commons February 2018 Version 2.0 Table of Contents Guide to Adjunct and Part Time Clinical
Report (CCR) Rule Retrospective Review: Electronic Delivery
 Consumer Confidence Report (CCR) Rule Retrospective Review: Electronic Delivery U.S. EPA Office of Ground Water and Drinking Water January 17, 2013 Audio The call in number is located within the Audio
Consumer Confidence Report (CCR) Rule Retrospective Review: Electronic Delivery U.S. EPA Office of Ground Water and Drinking Water January 17, 2013 Audio The call in number is located within the Audio
Make sure you delete all red type explanations. NOTE: Anticipate no action on applications that do not meet the timeline as posted on the IRB web site
 APPLICATION FOR REVIEW OF HUMAN PARTICIPANTS RESEARCH The following is designed to briefly introduce you to important considerations when answering each item. There is no one correct answer for each item,
APPLICATION FOR REVIEW OF HUMAN PARTICIPANTS RESEARCH The following is designed to briefly introduce you to important considerations when answering each item. There is no one correct answer for each item,
Request For Proposal ONWAA Website & E-Learn Portal
 Request For Proposal ONWAA Website & E-Learn Portal ONWAA 880 17 E, Garden River, Ontario P6A 6Z5 Table Of Contents General information Project Overview Statement of Needs Proposal Format Proposal Preparation
Request For Proposal ONWAA Website & E-Learn Portal ONWAA 880 17 E, Garden River, Ontario P6A 6Z5 Table Of Contents General information Project Overview Statement of Needs Proposal Format Proposal Preparation
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES
 SOP details SOP title: Protocol development SOP number: TM 010 SOP category: Trial Management Version number: 03 Version date: 16 December 2016 Effective date: 16 January 2017 Revision due date: 16 January
SOP details SOP title: Protocol development SOP number: TM 010 SOP category: Trial Management Version number: 03 Version date: 16 December 2016 Effective date: 16 January 2017 Revision due date: 16 January
SIF Data Model Specification Development, Review, Approval and Versioning Processes
 SIF Data Model Specification Development, Review, Approval and Versioning Processes www.a4l.org Version 1.0, Feb 2017 Table of Contents Specification Process Overview... 2 I. The SIF Specification... 2
SIF Data Model Specification Development, Review, Approval and Versioning Processes www.a4l.org Version 1.0, Feb 2017 Table of Contents Specification Process Overview... 2 I. The SIF Specification... 2
2. Goals of IRWM Plan Water management goals within the Tahoe Sierra IRWM Plan include but are not limited to:
 DRAFT Memorandum of Understanding (MOU) Regarding the Tahoe Sierra IRWM Plan (Integrated Regional Water Management Plan) Revised January 28, 2014, 1. Purpose of MOU The signatories of this Memorandum of
DRAFT Memorandum of Understanding (MOU) Regarding the Tahoe Sierra IRWM Plan (Integrated Regional Water Management Plan) Revised January 28, 2014, 1. Purpose of MOU The signatories of this Memorandum of
HPE Data Center Operations Consulting Service
 Data sheet HPE Data Center Operations Consulting Service HPE Packaged Consulting Services Data centers are large-scale capital investments that may not meet their capacity, availability, and operational
Data sheet HPE Data Center Operations Consulting Service HPE Packaged Consulting Services Data centers are large-scale capital investments that may not meet their capacity, availability, and operational
INTERNATIONAL STANDARD
 IEC 61158-3-13 INTERNATIONAL STANDARD Edition 1.0 2007-12 Industrial communication networks Fieldbus specifications Part 3-13: Data-link layer service definition Type 13 elements INTERNATIONAL ELECTROTECHNICAL
IEC 61158-3-13 INTERNATIONAL STANDARD Edition 1.0 2007-12 Industrial communication networks Fieldbus specifications Part 3-13: Data-link layer service definition Type 13 elements INTERNATIONAL ELECTROTECHNICAL
Statement on Continuing Professional Education 2003*
 01/2018 * The statement was issued in January, 2003 and amended from time to time. 1.1 The Institute of Chartered Accountants of India (ICAI), set up under the Chartered Accountants Act, 1949 (the Act),
01/2018 * The statement was issued in January, 2003 and amended from time to time. 1.1 The Institute of Chartered Accountants of India (ICAI), set up under the Chartered Accountants Act, 1949 (the Act),
TDWI Data Governance Fundamentals: Managing Data as an Asset
 TDWI Data Governance Fundamentals: Managing Data as an Asset Training Details Training Time : 1 Day Capacity : 10 Prerequisites : There are no prerequisites for this course. About Training About Training
TDWI Data Governance Fundamentals: Managing Data as an Asset Training Details Training Time : 1 Day Capacity : 10 Prerequisites : There are no prerequisites for this course. About Training About Training
Green Chemistry and Commerce Council Roundtable
 NSF/ANSI Standards Green Chemistry and Commerce Council Roundtable Portland, Oregon July 10, 2008 Clif McLellan Director of Toxicology Services Overview of Presentation Introduction to NSF International
NSF/ANSI Standards Green Chemistry and Commerce Council Roundtable Portland, Oregon July 10, 2008 Clif McLellan Director of Toxicology Services Overview of Presentation Introduction to NSF International
CEN and CENELEC Position Paper on the draft regulation ''Cybersecurity Act''
 CEN Identification number in the EC register: 63623305522-13 CENELEC Identification number in the EC register: 58258552517-56 CEN and CENELEC Position Paper on the draft regulation ''Cybersecurity Act''
CEN Identification number in the EC register: 63623305522-13 CENELEC Identification number in the EC register: 58258552517-56 CEN and CENELEC Position Paper on the draft regulation ''Cybersecurity Act''
ONC Health IT Certification Program
 ONC Health IT Certification Program Certification Requirements Update March 17, 2016 ICSA Labs Health IT Program Agenda Introduction Mandatory Product Disclosures and Transparency Requirements Certified
ONC Health IT Certification Program Certification Requirements Update March 17, 2016 ICSA Labs Health IT Program Agenda Introduction Mandatory Product Disclosures and Transparency Requirements Certified
Quality Data Model (QDM)
 Quality Data Model (QDM) Overview Document National Quality Forum 4/20/2011 QUALITY DATA MODEL (QDM): OVERVIEW TABLE OF CONTENTS Quality Data Model (QDM): Overview... 2 National Quality Forum: Overview
Quality Data Model (QDM) Overview Document National Quality Forum 4/20/2011 QUALITY DATA MODEL (QDM): OVERVIEW TABLE OF CONTENTS Quality Data Model (QDM): Overview... 2 National Quality Forum: Overview
OIML-CS PD-08 Edition 1
 PROCEDURAL DOCUMENT OIML-CS PD-08 Edition 1 Signing the OIML-CS Declaration OIML-CS PD-08 Edition 1 ORGANISATION INTERNATIONALE DE METROLOGIE LEGALE INTERNATIONAL ORGANIZATION OF LEGAL METROLOGY Contents
PROCEDURAL DOCUMENT OIML-CS PD-08 Edition 1 Signing the OIML-CS Declaration OIML-CS PD-08 Edition 1 ORGANISATION INTERNATIONALE DE METROLOGIE LEGALE INTERNATIONAL ORGANIZATION OF LEGAL METROLOGY Contents
EUROPEAN MEDICINES AGENCY (EMA) CONSULTATION
 EUROPEAN MEDICINES AGENCY (EMA) CONSULTATION Guideline on GCP compliance in relation to trial master file (paper and/or electronic) for content, management, archiving, audit and inspection of clinical
EUROPEAN MEDICINES AGENCY (EMA) CONSULTATION Guideline on GCP compliance in relation to trial master file (paper and/or electronic) for content, management, archiving, audit and inspection of clinical
INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD IEC 61158-5-20 Edition 2.0 2010-08 Industrial communication networks Fieldbus specifications Part 5-20: Application layer service definition Type 20 elements INTERNATIONAL ELECTROTECHNICAL
INTERNATIONAL STANDARD IEC 61158-5-20 Edition 2.0 2010-08 Industrial communication networks Fieldbus specifications Part 5-20: Application layer service definition Type 20 elements INTERNATIONAL ELECTROTECHNICAL
An Overview of ISO/IEC family of Information Security Management System Standards
 What is ISO/IEC 27001? The ISO/IEC 27001 standard, published by the International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC), is known as Information
What is ISO/IEC 27001? The ISO/IEC 27001 standard, published by the International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC), is known as Information
Developing a Cross Part Client Level Data System
 Developing a Cross Part Client Level Data System The Minnesota Experience Ryan White All Grantee Meeting August 25, 2010 SheilaMurphy RN CPHQ Hennepin County Human Services and Public Health Department
Developing a Cross Part Client Level Data System The Minnesota Experience Ryan White All Grantee Meeting August 25, 2010 SheilaMurphy RN CPHQ Hennepin County Human Services and Public Health Department
ISO/IEC/ IEEE INTERNATIONAL STANDARD. Systems and software engineering Requirements for acquirers and suppliers of user documentation
 INTERNATIONAL STANDARD ISO/IEC/ IEEE 26512 First edition 2011-06-01 Systems and software engineering Requirements for acquirers and suppliers of user documentation Ingénierie du logiciel et des systèmes
INTERNATIONAL STANDARD ISO/IEC/ IEEE 26512 First edition 2011-06-01 Systems and software engineering Requirements for acquirers and suppliers of user documentation Ingénierie du logiciel et des systèmes
June 30, Phyllis Schneider, AAP, Director, Network Rules ᅳ Rules Development & Technical Support
 June 30, 2010 TO: FROM: ACH Rulebook Subscribers Phyllis Schneider, AAP, Director, Network Rules ᅳ Rules Development & Technical Support RE: 2010 ACH Rulebook ᅳ Supplement #1-2010 Rules Simplification
June 30, 2010 TO: FROM: ACH Rulebook Subscribers Phyllis Schneider, AAP, Director, Network Rules ᅳ Rules Development & Technical Support RE: 2010 ACH Rulebook ᅳ Supplement #1-2010 Rules Simplification
Government of Ontario IT Standard (GO ITS) GO-ITS Number 56.3 Information Modeling Standard
 Government of Ontario IT Standard (GO ITS) GO-ITS Number 56.3 Information Modeling Standard Version # : 1.6 Status: Approved Prepared under the delegated authority of the Management Board of Cabinet Queen's
Government of Ontario IT Standard (GO ITS) GO-ITS Number 56.3 Information Modeling Standard Version # : 1.6 Status: Approved Prepared under the delegated authority of the Management Board of Cabinet Queen's
2014 ISACA Academic Scholarship Competition DUE DATE EXTENDED TO MAY 1, 2014
 2014 ISACA Academic Scholarship Competition DUE DATE EXTENDED TO MAY 1, 2014 ISACA is a pace-setting global organization for IT professionals focusing on information governance, security and audit. IT
2014 ISACA Academic Scholarship Competition DUE DATE EXTENDED TO MAY 1, 2014 ISACA is a pace-setting global organization for IT professionals focusing on information governance, security and audit. IT
