Harmonic Skeleton Guided Evaluation of Stenoses in Human Coronary Arteries
|
|
|
- Lucas Wilkinson
- 5 years ago
- Views:
Transcription
1 Harmonic Skeleton Guided Evaluation of Stenoses in Human Coronary Arteries Yan Yang 1,LeiZhu 1,StevenHaker 2, Allen R. Tannenbaum 1, and Don P. Giddens 1 1 Department of Biomedical Engineering, Georgia Institute of Technology, Atlanta, GA, 30332, USA gtg929i@mail.gatech.edu, zlzl@ece.gatech.edu, tannenba@ece.gatech.edu, don.giddens@coe.gatech.edu 2 Surgical Planning Lab, Brigham and Women s Hospital, Harvard Medical School, Boston, MA 02115, USA haker@bwh.harvard.edu Abstract. This paper presents a novel approach that three-dimensionally visualizes and evaluates stenoses in human coronary arteries by using harmonic skeletons. A harmonic skeleton is the center line of a multi-branched tubular surface extracted based on a harmonic function, which is the solution of the Laplace equation. This skeletonization method guarantees smoothness and connectivity and provides a fast and straightforward way to calculate local cross-sectional areas of the arteries, and thus provides the possibility to localize and evaluate coronary artery stenosis, which is a commonly seen pathology in coronary artery disease. 1 Introduction Stenosis is a commonly seen pathology resulting from atherosclerosis, which is a systemic disease of the vessel wall that occurs in the aorta, carotid, coronary, and peripheral arteries [1]. Atherosclerotic plaques that develop in vessel walls can intrude into the vessel lumen and result in a narrowing (stenosis) of the lumen. When stenosis occurs in coronary arteries, it will cause insufficient supply of blood to the heart tissue, and sometimes leads to serious results such as heart attacks. The detection of stenoses in vivo can greatly assist the diagnoses and treatments of coronary heart diseases. The conventional way of identifying stenoses is by using coronary angiography imaging, but this method is invasive, and it only provides 2D projections of the vessel lumen, which may yield biased or inaccurate evaluation of the stenosis. Thanks to the development of multislice- CT (MS-CT), we are now able to acquire 3D volumetric data of the entire coronary artery tree, and obtain the complete information of its geometry so that we can perform more accurate measurements based on these data. Numerous methods have been proposed to extract skeletons of tubular structuresinthehumanbodysuchasbloodvessels,airwaysandcolons.mostmethods in literature fall into the following two classes [4]: boundary peeling (also called thinning, erosion, etc.) [5], and distance coding (distance transform) [2, 3]. Although both classes of methods have yielded promising results, these methods J. Duncan and G. Gerig (Eds.): MICCAI 2005, LNCS 3749, pp , c Springer-Verlag Berlin Heidelberg 2005
2 Harmonic Skeleton Guided Evaluation of Stenoses in Human Coronary Arteries 491 usually have problems of preserving connectivity and being smooth. Also, direct extension of these methods to 3D is usually difficult and may not guarantee a unique solution for a single structure. In this paper, we explore a skeletonization method based on the solution of the Laplace equation with boundary conditions of the first kind (Dirichlet problem) on the surface of a tubular structure. Since the solution of the Laplace equation is a harmonic function, the skeleton extracted using this method is called the harmonic skeleton [8]. The harmonic skeleton is easy to compute, is guaranteed to be smooth, and can automatically give a viewing vector when used as a guidance for fly-throughs for virtual endoscopy. Moreover the space of harmonic functions is linear (as opposed to that of the distance transform on which the ordinary skeleton is founded). The distance transform is commonly used in endoscopy; see [9] and the references therein. Further, we develop an effective way to determine the boundary conditions for solving the appropriate Laplace equation, so that the points that form the final skeleton are evenly distributed, without being too dense or too sparse, thus guaranteeing the connectivity of the skeleton. Most importantly for our applications, the cross-sectional area at a certain location on the artery can be conveniently measured using the least squares plane that fits all the points on the iso-value contour of the solution of the Laplace equation. This means that we can acquire the cross-sectional areas throughout the artery in order to identify stenoses. We tested our method on CT data sets of both normal and diseased coronary arteries and developed different ways to visualize and analyze stenoses of the arteries. The method is described in detail in Section 2, and we show the results in Section 3. 2 Methodology 2.1 Harmonic Skeleton The harmonic skeleton can be obtained by solving the Laplace equation on the surface of the artery. Let Σ denote the surface, which is topologically a branched tube. Each branch has a boundary σ i (i =0,...,N 1), which is a closed contour, as shown in Figure 1. Here N denotes the total number of boundaries including the root σ 0, which can be selected arbitrarily or selected based on physiological considerations. We have chosen the root σ 0 to be at the proximal section of the left main (LM) coronary artery. Note that we are working on a triangulated representation of Σ, which makes it convenient for performing numerical computations. The surface can be obtained by segmenting the MS- CT images. In this case, we have used a segmentation approach that combines Bayesian classification and active contours [6, 7] to get the required surface for the skeletonization algorithm. Next we can calculate the harmonic function u by solving the Laplace equation on Σ\σ i (i =0,...,N 1): u =0 (1)
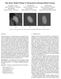
3 492 Y. Yang et al. Fig. 1. The segmented and openended surface of left coronary arteries with boundary contours at each end; the surface is colorpainted with solved harmonic function u, where dark color means lower u, and light color means higher u with proper boundary conditions. To find these boundary conditions, we assign the values at the boundaries in the following manner: 1. Set the boundary value of u at the root σ 0 to be zero, and set StepNumber =0; 2. Label all the points on σ 0 as visited ; 3. Label all the neighbors of the visited points as visited ; 4. StepNumber ++; 5. Repeat steps 3. and 4. until visited points reached another boundary σ i ; 6. Set the value at the current boundary σ i to be the current value of Step- Number. In other words, we set the boundary value of σ i to be the number of triangle strips between σ i and the root boundary σ 0. In coronary arteries, there are several branches, some of which are long and some of which are short. Note that if we used standard Dirichlet boundary conditions in which u(σ 0 )=0and u(σ i ) = 1 for all i s, as for example, is done in [13], the resulting points that form the skeleton will be denser for the short branches, and sparser for the longer ones. Hence we employ the boundary conditions stated above to get more evenly distributed skeleton points. Having obtained the boundary conditions for the Dirichlet problem, we can solve the Laplace equation u = 0 on the surface using finite element techniques [6, 12]. From the standard theory [10], u exists and is unique, with the maximum and minimum values on the boundaries. In Figure 1, the surface of the artery is painted with the solved u, where brighter color indicates higher u, and darker color indicates lower u. Using the harmonic function u, we can now build the harmonic skeleton. We first find the level sets of u on the surface, i.e., sets {x u(x) =ν} for values of ν ranging from 0 to max(u). Each level set corresponds to a group of points on the surfacethathavethevalueofacertainν. These points are not necessarily located on the vertices of our original triangulation of the surface, so we obtain these level points by linearly interpolating the coordinates of the vertices according to the values of u.
4 Harmonic Skeleton Guided Evaluation of Stenoses in Human Coronary Arteries 493 We also check the change in the topology of the level contours as we proceed, and further partition these contours into sub-contours if not all the points are connected to form a single contour. This means there is a bifurcation and one contour splits into two at the current level of u. We assume one contour cannot split into more than two contours, which is usually true for coronary arteries. 1 The centroid of each contour or sub-contour is then calculated: M ΣM i=1 x i,where x i =(x i,y i,z i ) is a point on the contour, and M is the total number of points on the current contour. Each centroid contributes a point to the final harmonic skeleton. By increasing ν from 0 to the maximum value of u, we can find a series of centroids that can finally be connected to build a structured tree, which is the harmonic skeleton. For the sake of accuracy, an additional step can be performed to refine the harmonic skeleton by replacing the previous boundary conditions with new values obtained as the distance along the skeleton from the root boundary to the other boundaries. We then solve the Dirichlet problem with the new boundary conditions and further obtain a refined harmonic skeleton. 2.2 Cross-Sectional Area Measurement The harmonic skeleton obtained using the above method can be used as a guide in calculating cross-sectional areas at specific locations along the vessels, and the cross-sectional areas can in turn be used to precisely evaluate stenoses in the coronaries. Cross-sectional areas are better than diameters because atherosclerotic plaques are usually eccentric and asymmetric, and the cross-sections of the narrowed vessel lumen may not be perfectly circular. The stenosis may be underestimated if one uses only diameters for stenosis evaluation. Cross-sectional areas, on the other hand, provide more information about the stenosis, and are directly related to the flow rate of blood through the cross-section. One straightforward way of obtaining the cross-sections of the artery is by looking for a plane that is perpendicular to the skeleton to cut the tube at a certain location. But this strongly depends on the local properties of the skeleton, and a plane that is perpendicular to the skeleton locally may not be perpendicular to the vessel itself. What we do here is to start with the contours or sub-contours obtained in Section 2.1, and measure the cross-sectional area at the same time as we calculate the centroid. The first thing to do is to find the cross-sectional plane. We have many more points (about 50 points, somewhat more for wider contours and somewhat less for narrower contours) on a level contour than what is necessary (3 points) for determining a plane. Hence we need to use least squares to solve this overdetermined problem. Suppose we have a set of points X =(x 1, x 2,...x N ),where x i =(x i,y i,z i ),i=1,...,m is a point on the level contour, and we build a matrix A = X x 0,wherex 0 (x 0,y 0,z 0 ) is the centroid of all the points on the contour. Calculating the Singular Value Decomposition (SVD)[14] of matrix A, wegeta = USV T,whereU and V are orthogonal matrices and S is a diagonal matrix with non-negative diagonal entries in decreasing order. The singular vector in V that corresponds to the smallest singular value in S is the
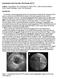
5 494 Y. Yang et al. cosine of the direction of the normal to the best-fit plane. The centroid and the normal vector can now be used to uniquely determine the least squares plane. We now use this plane to cut the surface of the vessel and get a series of points on the surface that also belong to the least squares plane. Again, these points may not coincide with the vertices of the triangulated surface, so we find the points on the triangle edges, and connect them to form a polygon. We then calculate the area of the polygon, and the result is the required cross-sectional area at the current location on the vessel. 3 Results 3.1 Clinical Data We tested our method on a healthy volunteer without any clinical symptoms and a patient with plaques in left anterior descending (LAD) coronary artery. The healthy volunteer was imaged using a GE LightSpeed16 CT scanner with a slice spacing of 0.625mm and an in-plane resolution of 0.60mm, while the patient with LAD plaques was imaged using a Siemens Sensation64 CT scanner with a slice spacing of 0.75mm and a in-plane resolution of 0.39mm. Contrast agents were used in both cases. The coronary arteries were extracted by image segmentation using the techniques of [6, 7], and the triangulated surfaces were generated using the Visualization Toolkit (VTK) [15]. We then performed our skeletonization and cross-sectional area measurement methods on the triangulated surfaces. 3.2 Generation of Harmonic Skeletons The harmonic skeletons of both cases were generated using the method described in Section 2.1. Figure 2 (Left) and (Middle) show the surfaces in half-transparent mode with the skeletons inside the surfaces, and Figure 2 (Right) shows the skeleton of the diseased (Middle) coronary arteries. The skeletons provide the Fig. 2. Left: Coronaries and the skeleton of a healthy volunteer. Middle: Coronaries and the skeleton of a patient with plaques in LAD. Right: The skeleton of the middle coronaries.
6 Harmonic Skeleton Guided Evaluation of Stenoses in Human Coronary Arteries 495 basic information of the arteries, such as bifurcation sites, bifurcation angles and curvatures along the vessels, but they themselves alone are not able to provide much information about the stenosis. At the bifurcation sites, some post-processing was performed to keep the skeleton smooth and natural. Increasing the value of u and tracking the topology change of the level contours, one contour will split into two at a certain u level, and there will be a jump between the centroid of the single parent contour and the centroids of the two daughter contours. If we connect the centroids directly, the resulting skeleton may not be smooth where it bifurcates. To solve this problem, we start from the daughter branches, and trace back until they reach the bifurcation site, and then extend the branches further until they meet the parent branch. After the post-processing, the skeleton is visually more natural at the bifurcation sites. 3.3 Stenosis Evaluation We calculated cross-sectional areas along the vessel with a resolution of δu =0.5, which means we measure the cross-sectional area once we increase the harmonic function u by 0.5. For each coronary artery, there are roughly 400 levels of crosssectional areas, and at each level, there are one or several cross-sectional areas measured according to the number of branches at this level. We can visualize the quantitative measurementsin two different ways. The first way is to specify a u value, and show the cross-sectional areas at this u level, as illustrated in Figure 3 (Left). This is equivalent to showing the areas by specifying a location on the vessel. The second way is to create a color map of the crosssectional area values, and paint the surface with the color of these values. This provides an intuitive way to localize the stenosis, by showing different colors where the cross-sectional area has an apparent change along the vessels, as indicated in Figure 3 (Right). In this figure, lighter color indicates normal sized vessel lumen, and darker color means the lumen is narrowed, thus implying stenosis. Fig. 3. Cross-sectional area visualization. Left: The cross-sectional area of each branch of the coronaries for a certain u. Right: Color-painted LAD indicates a stenosis site in the vessel.
7 496 Y. Yang et al Cross sectional area (mm 2 ) u value along LAD Stenosis percentage (%) Fig. 4. Cross-sectional area along the LAD shown in Fig. 3 (Right). The left y-axis shows the cross-sectional area in mm 2, and the right y-axis shows the stenosis percentage. Figure 4 is a plot of u versus cross-sectional area along the LAD of Figure 3 (Right). The left y-axis shows the cross-sectional areas in mm 2,andtheright y-axis shows the stenosis percentage, which is an important measure for the evaluation of stenosis. We can see from this plot that this LAD has two stenosis sites, one mild (a little less than 40% blockage) and another more severe (60% blockage). This is in consistent with what is shown in Figure 3 (Right). 4 Discussion and Conclusions In this note, we have proposed an approach to extract harmonic skeletons of tubular structures and applied it to both healthy and diseased coronary arteries. The skeleton generated can be used as a guide for cross-sectional area measurements in order to localize and evaluate stenosis of the arteries. The harmonic skeleton is smooth and unique, and can also serve as a guide in other applications such as virtual endoscopy. Our algorithm is reasonably fast, taking 2-3 minutes in Matlab to generate the harmonic skeleton from a surface consisting of about 30,000 triangles and 12,000 vertices, on a 1G Hz personal computer running Linux. If the skeleton extraction and cross-sectional area measurement are performed at the same time, it takes 6-7 minutes for the same sized data. This makes it possible to use this algorithm in a real clinical setting. We should also point out that this skeletonization and cross-sectional area measuring approach is performed on a segmented surface of a tubular structure, so the measurement results highly rely on the correctness of the image segmentation. On the other hand, the cross-sectional area can also serve a measure for the validation of segmentation to compare the values of a ground truth model (such as a phantom or a vessel cast) and the segmentation result. Acknowledgements This work is part of the National Alliance for Medical Image Computing (NAMIC), funded by the National Institutes of Health through the NIH Roadmap for Medical Research, Grant U54 EB This work was also supported by a grant from NIH (NAC P41 RR through Brigham and Women s Hospital). S. Haker s
8 Harmonic Skeleton Guided Evaluation of Stenoses in Human Coronary Arteries 497 work is supported by NIH grants R01CA109246, R01LM007861, R01CA and P41RR References 1. V. Fuster, Z.A. Fayad, J. J. Badimon, Acute coronary syndromes: biology, Lancet 1999; 353 (supp1 2): SII5-SII9 2. M. Maddah, H. Soltanian-Zadeh, and A. Afzali-Kusha, Snake modeling and distance transform approach to vascular centerline extraction and quantification, Computerized Medical Imaging and Graphics, 27, 2003, pp M. Wan, Z. Liang, Q. Ke, L. Hong, I. Bitter, and A. Kaufman, Automatic Centerline Extraction for Virtual Colonoscopy, IEEE Trans. on Med. Imag. 21, 2002, pp Y. Zhou, and A. W. Toga Efficient Skeletonization of Volumetric Objects, IEEE Tran. on Visualization and Computer Graphics, Vol. 5, NO. 3, 1999, pp L. Hong, A. Kaufman, Y. Wei, A. Viswambharan, M. Wax and Z. Liang, 3D Virtual Colonoscopy, Proc. of IEEE Biomedical Visualization, 1995, pp S. Haker, G. Sapiro, A. Tannenbaum, Knowledge-Based Segmentation of SAR Data with learned Priors, IEEE Tran. on Image Processing, Vol. 9, No. 2, Feb. 2000, pp Y. Yang, A. Tannenbaum, and D. Giddens. Knowledge-Based 3D Segmentation and Reconstruction of Coronary Arteries Using CT Images, In Proceedings of the 26th Annual International Conference of the IEEE EMBS, pages , L. Zhu, S. Haker, and A. Tannenbaum. Flattening Maps for the Visualization of Multi-branched Vessels, IEEE Trans. on Med. Imag., 24(2): , S. Bouix, K. Siddiqi, A. Tannenbaum, Flux Driven Automatic Centerline Extraction, Medical Image Analysis 9, 2005, pp Stanley J. Farlow Partial Differential Equations for Scientists and Engineers, Dover Publications, Inc. New York, S. Haker, S. Angenent, A. Tannenbaum, and R.Kikinis, Nondistorting flattening maps and the 3D visualization of colon CT images, IEEE Trans. Med. Imag., Vol. 19, 2000, pp T. Hughes, The Finite Element Method, Upper Saddle River, NJ: Prentice-Hall, A. J. Yezzi, Jr., J. L. Prince, An Eulerian PDE Approach for Computing Tissure Thickness, IEEE Trans. Med. Imag., Vol. 22, No. 10, 2003, pp W. Press, S. Teukolsky, W. Vetterling, and B. Flannery, Numerical Recipes in C, 2nd ed. Cambridge: Cambridge Univ. Press, Kitware Inc.
Conformal flattening maps for the visualization of vessels
 Conformal flattening maps for the visualization of vessels Lei Zhua, Steven Hakerb, and Allen Tannenbauma adept of Biomedical Engineering, Georgia Tech, Atlanta bdept of Radiology, Brigham and Women's
Conformal flattening maps for the visualization of vessels Lei Zhua, Steven Hakerb, and Allen Tannenbauma adept of Biomedical Engineering, Georgia Tech, Atlanta bdept of Radiology, Brigham and Women's
THE techniques of surface deformation and mapping can
 IEEE TRANSACTIONS ON MEDICAL IMAGING, VOL. 24, NO. 2, FEBRUARY 2005 191 Flattening Maps for the Visualization of Multibranched Vessels Lei Zhu, Steven Haker, and Allen Tannenbaum*, Member, IEEE Abstract
IEEE TRANSACTIONS ON MEDICAL IMAGING, VOL. 24, NO. 2, FEBRUARY 2005 191 Flattening Maps for the Visualization of Multibranched Vessels Lei Zhu, Steven Haker, and Allen Tannenbaum*, Member, IEEE Abstract
Area-Preserving Mappings for the Visualization of Medical Structures
 Area-Preserving Mappings for the Visualization of Medical Structures Lei Zhu 1, Steven Haker 2, and Allen Tannenbaum 1 1 Department of Biomedical Engineering, Georgia Institute of Technology, Atlanta,
Area-Preserving Mappings for the Visualization of Medical Structures Lei Zhu 1, Steven Haker 2, and Allen Tannenbaum 1 1 Department of Biomedical Engineering, Georgia Institute of Technology, Atlanta,
Probabilistic Tracking and Model-based Segmentation of 3D Tubular Structures
 Probabilistic Tracking and Model-based Segmentation of 3D Tubular Structures Stefan Wörz, William J. Godinez, Karl Rohr University of Heidelberg, BIOQUANT, IPMB, and DKFZ Heidelberg, Dept. Bioinformatics
Probabilistic Tracking and Model-based Segmentation of 3D Tubular Structures Stefan Wörz, William J. Godinez, Karl Rohr University of Heidelberg, BIOQUANT, IPMB, and DKFZ Heidelberg, Dept. Bioinformatics
An ITK Filter for Bayesian Segmentation: itkbayesianclassifierimagefilter
 An ITK Filter for Bayesian Segmentation: itkbayesianclassifierimagefilter John Melonakos 1, Karthik Krishnan 2 and Allen Tannenbaum 1 1 Georgia Institute of Technology, Atlanta GA 30332, USA {jmelonak,
An ITK Filter for Bayesian Segmentation: itkbayesianclassifierimagefilter John Melonakos 1, Karthik Krishnan 2 and Allen Tannenbaum 1 1 Georgia Institute of Technology, Atlanta GA 30332, USA {jmelonak,
NIH Public Access Author Manuscript Conf Proc IEEE Eng Med Biol Soc. Author manuscript; available in PMC 2009 December 11.
 NIH Public Access Author Manuscript Published in final edited form as: Conf Proc IEEE Eng Med Biol Soc. 2004 ; 2: 1521 1524. doi:10.1109/iembs.2004.1403466. An Elliptic PDE Approach for Shape Characterization
NIH Public Access Author Manuscript Published in final edited form as: Conf Proc IEEE Eng Med Biol Soc. 2004 ; 2: 1521 1524. doi:10.1109/iembs.2004.1403466. An Elliptic PDE Approach for Shape Characterization
Intra-Patient Prone to Supine Colon Registration for Synchronized Virtual Colonoscopy
 Intra-Patient Prone to Supine Colon Registration for Synchronized Virtual Colonoscopy Delphine Nain 1, Steven Haker 2, W. Eric L. Grimson 1, Eric Cosman Jr 1, William W. Wells 1,2, Hoon Ji 2, Ron Kikinis
Intra-Patient Prone to Supine Colon Registration for Synchronized Virtual Colonoscopy Delphine Nain 1, Steven Haker 2, W. Eric L. Grimson 1, Eric Cosman Jr 1, William W. Wells 1,2, Hoon Ji 2, Ron Kikinis
Comparison of Vessel Segmentations Using STAPLE
 Comparison of Vessel Segmentations Using STAPLE Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab, The University of North Carolina at Chapel Hill, Department
Comparison of Vessel Segmentations Using STAPLE Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab, The University of North Carolina at Chapel Hill, Department
Knowledge-Based Segmentation of Brain MRI Scans Using the Insight Toolkit
 Knowledge-Based Segmentation of Brain MRI Scans Using the Insight Toolkit John Melonakos 1, Ramsey Al-Hakim 1, James Fallon 2 and Allen Tannenbaum 1 1 Georgia Institute of Technology, Atlanta GA 30332,
Knowledge-Based Segmentation of Brain MRI Scans Using the Insight Toolkit John Melonakos 1, Ramsey Al-Hakim 1, James Fallon 2 and Allen Tannenbaum 1 1 Georgia Institute of Technology, Atlanta GA 30332,
Conformal Flattening ITK Filter
 =1 Conformal Flattening ITK Filter Release 0.00 Yi Gao 1, John Melonakos 1, and Allen Tannenbaum 1 July 10, 2006 1 Georgia Institute of Technology, Atlanta, GA Abstract This paper describes the Insight
=1 Conformal Flattening ITK Filter Release 0.00 Yi Gao 1, John Melonakos 1, and Allen Tannenbaum 1 July 10, 2006 1 Georgia Institute of Technology, Atlanta, GA Abstract This paper describes the Insight
Human Heart Coronary Arteries Segmentation
 Human Heart Coronary Arteries Segmentation Qian Huang Wright State University, Computer Science Department Abstract The volume information extracted from computed tomography angiogram (CTA) datasets makes
Human Heart Coronary Arteries Segmentation Qian Huang Wright State University, Computer Science Department Abstract The volume information extracted from computed tomography angiogram (CTA) datasets makes
Conformal Flattening ITK Filter
 =1 Conformal Flattening ITK Filter Release 0.00 Yi Gao 1, John Melonakos 1, and Allen Tannenbaum 1 July 22, 2006 1 Georgia Institute of Technology, Atlanta, GA Abstract This paper describes the Insight
=1 Conformal Flattening ITK Filter Release 0.00 Yi Gao 1, John Melonakos 1, and Allen Tannenbaum 1 July 22, 2006 1 Georgia Institute of Technology, Atlanta, GA Abstract This paper describes the Insight
Mimics Innovation Suite Mimics Centerline Extraction: Quantitative Validation
 Mimics Innovation Suite Mimics Centerline Extraction: Quantitative Validation Verhoelst Eefje, Shtankevych Oleksii, Schepers Jan, Maes Jan, Sindhwani Nikhil, Lysogor Andriy, Veeckmans Bart 1 / Introduction
Mimics Innovation Suite Mimics Centerline Extraction: Quantitative Validation Verhoelst Eefje, Shtankevych Oleksii, Schepers Jan, Maes Jan, Sindhwani Nikhil, Lysogor Andriy, Veeckmans Bart 1 / Introduction
Vessel Centerline Tracking in CTA and MRA Images Using Hough Transform
 Vessel Centerline Tracking in CTA and MRA Images Using Hough Transform Maysa M.G. Macedo 1, Choukri Mekkaoui 2, and Marcel P. Jackowski 1 1 University of São Paulo, Department of Computer Science, Rua
Vessel Centerline Tracking in CTA and MRA Images Using Hough Transform Maysa M.G. Macedo 1, Choukri Mekkaoui 2, and Marcel P. Jackowski 1 1 University of São Paulo, Department of Computer Science, Rua
Accurate Lumen Segmentation and Stenosis Detection and Quantification in Coronary CTA
 Accurate Lumen Segmentation and Stenosis Detection and Quantification in Coronary CTA Brian Mohr, Saad Masood, Costas Plakas Toshiba Medical Visualization Systems, Edinburgh, UK Abstract. Accurate detection
Accurate Lumen Segmentation and Stenosis Detection and Quantification in Coronary CTA Brian Mohr, Saad Masood, Costas Plakas Toshiba Medical Visualization Systems, Edinburgh, UK Abstract. Accurate detection
Vessel Explorer: a tool for quantitative measurements in CT and MR angiography
 Clinical applications Vessel Explorer: a tool for quantitative measurements in CT and MR angiography J. Oliván Bescós J. Sonnemans R. Habets J. Peters H. van den Bosch T. Leiner Healthcare Informatics/Patient
Clinical applications Vessel Explorer: a tool for quantitative measurements in CT and MR angiography J. Oliván Bescós J. Sonnemans R. Habets J. Peters H. van den Bosch T. Leiner Healthcare Informatics/Patient
A Hybrid Method for Coronary Artery Stenoses Detection and Quantification in CTA Images
 A Hybrid Method for Coronary Artery Stenoses Detection and Quantification in CTA Images İlkay Öksüz 1, Devrim Ünay 2, Kamuran Kadıpaşaoğlu 2 1 Electrical and Electronics Engineering, Bahçeşehir University,
A Hybrid Method for Coronary Artery Stenoses Detection and Quantification in CTA Images İlkay Öksüz 1, Devrim Ünay 2, Kamuran Kadıpaşaoğlu 2 1 Electrical and Electronics Engineering, Bahçeşehir University,
Estimating Arterial Wall Shear Stress 1
 DEPARTMENT OF STATISTICS University of Wisconsin 1210 West Dayton St. Madison, WI 53706 TECHNICAL REPORT NO. 1088 December 12, 2003 Estimating Arterial Wall Shear Stress 1 John D. Carew 2 Departments of
DEPARTMENT OF STATISTICS University of Wisconsin 1210 West Dayton St. Madison, WI 53706 TECHNICAL REPORT NO. 1088 December 12, 2003 Estimating Arterial Wall Shear Stress 1 John D. Carew 2 Departments of
Knowledge-Based Segmentation of Brain MRI Scans Using the Insight Toolkit
 Knowledge-Based Segmentation of Brain MRI Scans Using the Insight Toolkit John Melonakos 1, Ramsey Al-Hakim 1, James Fallon 2 and Allen Tannenbaum 1 1 Georgia Institute of Technology, Atlanta GA 30332,
Knowledge-Based Segmentation of Brain MRI Scans Using the Insight Toolkit John Melonakos 1, Ramsey Al-Hakim 1, James Fallon 2 and Allen Tannenbaum 1 1 Georgia Institute of Technology, Atlanta GA 30332,
Automatic Vascular Tree Formation Using the Mahalanobis Distance
 Automatic Vascular Tree Formation Using the Mahalanobis Distance Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab, Department of Radiology The University
Automatic Vascular Tree Formation Using the Mahalanobis Distance Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab, Department of Radiology The University
Robust and Accurate Coronary Artery Centerline Extraction in CTA by Combining Model-Driven and Data-Driven Approaches
 Robust and Accurate Coronary Artery Centerline Extraction in CTA by Combining Model-Driven and Data-Driven Approaches Yefeng Zheng, Huseyin Tek, and Gareth Funka-Lea Imaging and Computer Vision, Siemens
Robust and Accurate Coronary Artery Centerline Extraction in CTA by Combining Model-Driven and Data-Driven Approaches Yefeng Zheng, Huseyin Tek, and Gareth Funka-Lea Imaging and Computer Vision, Siemens
Phantom-based evaluation of a semi-automatic segmentation algorithm for cerebral vascular structures in 3D ultrasound angiography (3D USA)
 Phantom-based evaluation of a semi-automatic segmentation algorithm for cerebral vascular structures in 3D ultrasound angiography (3D USA) C. Chalopin¹, K. Krissian², A. Müns 3, F. Arlt 3, J. Meixensberger³,
Phantom-based evaluation of a semi-automatic segmentation algorithm for cerebral vascular structures in 3D ultrasound angiography (3D USA) C. Chalopin¹, K. Krissian², A. Müns 3, F. Arlt 3, J. Meixensberger³,
NIH Public Access Author Manuscript Proc Soc Photo Opt Instrum Eng. Author manuscript; available in PMC 2014 October 07.
 NIH Public Access Author Manuscript Published in final edited form as: Proc Soc Photo Opt Instrum Eng. 2014 March 21; 9034: 903442. doi:10.1117/12.2042915. MRI Brain Tumor Segmentation and Necrosis Detection
NIH Public Access Author Manuscript Published in final edited form as: Proc Soc Photo Opt Instrum Eng. 2014 March 21; 9034: 903442. doi:10.1117/12.2042915. MRI Brain Tumor Segmentation and Necrosis Detection
Vessel Segmentation with Automatic Centerline Extraction Using Tubular Tree Segmentation
 Vessel Segmentation with Automatic Centerline Extraction Using Tubular Tree Segmentation Vandana Mohan, Ganesh Sundaramoorthi, Arthur Stillman, Allen Tannenbaum To cite this version: Vandana Mohan, Ganesh
Vessel Segmentation with Automatic Centerline Extraction Using Tubular Tree Segmentation Vandana Mohan, Ganesh Sundaramoorthi, Arthur Stillman, Allen Tannenbaum To cite this version: Vandana Mohan, Ganesh
Application of level set based method for segmentation of blood vessels in angiography images
 Lodz University of Technology Faculty of Electrical, Electronic, Computer and Control Engineering Institute of Electronics PhD Thesis Application of level set based method for segmentation of blood vessels
Lodz University of Technology Faculty of Electrical, Electronic, Computer and Control Engineering Institute of Electronics PhD Thesis Application of level set based method for segmentation of blood vessels
Tissue Tracking: Applications for Brain MRI Classification
 Tissue Tracking: Applications for Brain MRI Classification John Melonakos a and Yi Gao a and Allen Tannenbaum a a Georgia Institute of Technology, 414 Ferst Dr, Atlanta, GA, USA; ABSTRACT Bayesian classification
Tissue Tracking: Applications for Brain MRI Classification John Melonakos a and Yi Gao a and Allen Tannenbaum a a Georgia Institute of Technology, 414 Ferst Dr, Atlanta, GA, USA; ABSTRACT Bayesian classification
NIH Public Access Author Manuscript Med Image Comput Comput Assist Interv. Author manuscript; available in PMC 2013 May 07.
 NIH Public Access Author Manuscript Published in final edited form as: Med Image Comput Comput Assist Interv. 2005 ; 8(0 2): 459 467. Multiscale 3D Shape Analysis using Spherical Wavelets Delphine Nain
NIH Public Access Author Manuscript Published in final edited form as: Med Image Comput Comput Assist Interv. 2005 ; 8(0 2): 459 467. Multiscale 3D Shape Analysis using Spherical Wavelets Delphine Nain
Copyright 2008 Society of Photo Optical Instrumentation Engineers. This paper was published in Proceedings of SPIE, vol. 6915, Medical Imaging 2008:
 Copyright 2008 Society of Photo Optical Instrumentation Engineers. This paper was published in Proceedings of SPIE, vol. 6915, Medical Imaging 2008: Computer Aided Diagnosis and is made available as an
Copyright 2008 Society of Photo Optical Instrumentation Engineers. This paper was published in Proceedings of SPIE, vol. 6915, Medical Imaging 2008: Computer Aided Diagnosis and is made available as an
FINDING THE TRUE EDGE IN CTA
 FINDING THE TRUE EDGE IN CTA by: John A. Rumberger, PhD, MD, FACC Your patient has chest pain. The Cardiac CT Angiography shows plaque in the LAD. You adjust the viewing window trying to evaluate the stenosis
FINDING THE TRUE EDGE IN CTA by: John A. Rumberger, PhD, MD, FACC Your patient has chest pain. The Cardiac CT Angiography shows plaque in the LAD. You adjust the viewing window trying to evaluate the stenosis
Multiscale 3D Shape Analysis Using Spherical Wavelets
 Multiscale 3D Shape Analysis Using Spherical Wavelets Delphine Nain 1,StevenHaker 2, Aaron Bobick 1, and Allen R. Tannenbaum 3 1 College of Computing, Georgia Institute of Technology, Atlanta, GA 30332-0280
Multiscale 3D Shape Analysis Using Spherical Wavelets Delphine Nain 1,StevenHaker 2, Aaron Bobick 1, and Allen R. Tannenbaum 3 1 College of Computing, Georgia Institute of Technology, Atlanta, GA 30332-0280
Fast Centerline Extraction Method of Cardiovascular Virtual Endoscopy
 Method of Cardiovascular Virtual Endoscopy 12 Department of Automation, Shanghai Jiao Tong University No.800 Dongchuan Rd. Shanghai, China, 200240 E-mail: aanonymous@sjtu.edu.cn Xin Yang Department of
Method of Cardiovascular Virtual Endoscopy 12 Department of Automation, Shanghai Jiao Tong University No.800 Dongchuan Rd. Shanghai, China, 200240 E-mail: aanonymous@sjtu.edu.cn Xin Yang Department of
2D Vessel Segmentation Using Local Adaptive Contrast Enhancement
 2D Vessel Segmentation Using Local Adaptive Contrast Enhancement Dominik Schuldhaus 1,2, Martin Spiegel 1,2,3,4, Thomas Redel 3, Maria Polyanskaya 1,3, Tobias Struffert 2, Joachim Hornegger 1,4, Arnd Doerfler
2D Vessel Segmentation Using Local Adaptive Contrast Enhancement Dominik Schuldhaus 1,2, Martin Spiegel 1,2,3,4, Thomas Redel 3, Maria Polyanskaya 1,3, Tobias Struffert 2, Joachim Hornegger 1,4, Arnd Doerfler
Image Thickness Correction for Navigation with 3D Intra-cardiac Ultrasound Catheter
 Image Thickness Correction for Navigation with 3D Intra-cardiac Ultrasound Catheter Hua Zhong 1, Takeo Kanade 1,andDavidSchwartzman 2 1 Computer Science Department, Carnegie Mellon University, USA 2 University
Image Thickness Correction for Navigation with 3D Intra-cardiac Ultrasound Catheter Hua Zhong 1, Takeo Kanade 1,andDavidSchwartzman 2 1 Computer Science Department, Carnegie Mellon University, USA 2 University
Comparison of Vessel Segmentations using STAPLE
 Comparison of Vessel Segmentations using STAPLE Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab The University of North Carolina at Chapel Hill, Department
Comparison of Vessel Segmentations using STAPLE Julien Jomier, Vincent LeDigarcher, and Stephen R. Aylward Computer-Aided Diagnosis and Display Lab The University of North Carolina at Chapel Hill, Department
A Comprehensive Method for Geometrically Correct 3-D Reconstruction of Coronary Arteries by Fusion of Intravascular Ultrasound and Biplane Angiography
 Computer-Aided Diagnosis in Medical Imaging, September 20-23, 1998, Chicago IL Elsevier ICS 1182 A Comprehensive Method for Geometrically Correct 3-D Reconstruction of Coronary Arteries by Fusion of Intravascular
Computer-Aided Diagnosis in Medical Imaging, September 20-23, 1998, Chicago IL Elsevier ICS 1182 A Comprehensive Method for Geometrically Correct 3-D Reconstruction of Coronary Arteries by Fusion of Intravascular
2 Michael E. Leventon and Sarah F. F. Gibson a b c d Fig. 1. (a, b) Two MR scans of a person's knee. Both images have high resolution in-plane, but ha
 Model Generation from Multiple Volumes using Constrained Elastic SurfaceNets Michael E. Leventon and Sarah F. F. Gibson 1 MIT Artificial Intelligence Laboratory, Cambridge, MA 02139, USA leventon@ai.mit.edu
Model Generation from Multiple Volumes using Constrained Elastic SurfaceNets Michael E. Leventon and Sarah F. F. Gibson 1 MIT Artificial Intelligence Laboratory, Cambridge, MA 02139, USA leventon@ai.mit.edu
VASCULAR TREE CHARACTERISTIC TABLE BUILDING FROM 3D MR BRAIN ANGIOGRAPHY IMAGES
 VASCULAR TREE CHARACTERISTIC TABLE BUILDING FROM 3D MR BRAIN ANGIOGRAPHY IMAGES D.V. Sanko 1), A.V. Tuzikov 2), P.V. Vasiliev 2) 1) Department of Discrete Mathematics and Algorithmics, Belarusian State
VASCULAR TREE CHARACTERISTIC TABLE BUILDING FROM 3D MR BRAIN ANGIOGRAPHY IMAGES D.V. Sanko 1), A.V. Tuzikov 2), P.V. Vasiliev 2) 1) Department of Discrete Mathematics and Algorithmics, Belarusian State
Coronary vessel cores from 3D imagery: a topological approach
 Coronary vessel cores from 3D imagery: a topological approach Andrzej Szymczak a and Allen Tannenbaum b and Konstantin Mischaikow c a College of Computing, Georgia Tech, Atlanta, GA 30332, USA; b Department
Coronary vessel cores from 3D imagery: a topological approach Andrzej Szymczak a and Allen Tannenbaum b and Konstantin Mischaikow c a College of Computing, Georgia Tech, Atlanta, GA 30332, USA; b Department
Isogeometric Analysis of Fluid-Structure Interaction
 Isogeometric Analysis of Fluid-Structure Interaction Y. Bazilevs, V.M. Calo, T.J.R. Hughes Institute for Computational Engineering and Sciences, The University of Texas at Austin, USA e-mail: {bazily,victor,hughes}@ices.utexas.edu
Isogeometric Analysis of Fluid-Structure Interaction Y. Bazilevs, V.M. Calo, T.J.R. Hughes Institute for Computational Engineering and Sciences, The University of Texas at Austin, USA e-mail: {bazily,victor,hughes}@ices.utexas.edu
Modeling Mitral Valve Leaflets from Three-Dimensional Ultrasound
 Modeling Mitral Valve Leaflets from Three-Dimensional Ultrasound Robert J. Schneider 1, William C. Burke 1, Gerald R. Marx 3, Pedro J. del Nido 2, and Robert D. Howe 1 1 Harvard School of Engineering and
Modeling Mitral Valve Leaflets from Three-Dimensional Ultrasound Robert J. Schneider 1, William C. Burke 1, Gerald R. Marx 3, Pedro J. del Nido 2, and Robert D. Howe 1 1 Harvard School of Engineering and
Chapter 11 Representation & Description
 Chain Codes Chain codes are used to represent a boundary by a connected sequence of straight-line segments of specified length and direction. The direction of each segment is coded by using a numbering
Chain Codes Chain codes are used to represent a boundary by a connected sequence of straight-line segments of specified length and direction. The direction of each segment is coded by using a numbering
Pearling: Medical Image Segmentation with Pearl Strings
 Pearling: Medical Image Segmentation with Pearl Strings Jarek Rossignac 1, Brian Whited 1, Greg Slabaugh 2, Tong Fang 2, Gozde Unal 2 1 Georgia Institute of Technology Graphics, Visualization, and Usability
Pearling: Medical Image Segmentation with Pearl Strings Jarek Rossignac 1, Brian Whited 1, Greg Slabaugh 2, Tong Fang 2, Gozde Unal 2 1 Georgia Institute of Technology Graphics, Visualization, and Usability
Combining Classifiers Using Their Receiver Operating Characteristics and Maximum Likelihood Estimation
 Combining Classifiers Using Their Receiver Operating Characteristics and Maximum Likelihood Estimation Steven Haker, William M. Wells III, Simon K. Warfield, Ion-Florin Talos, Jui G. Bhagwat, Daniel Goldberg-Zimring,
Combining Classifiers Using Their Receiver Operating Characteristics and Maximum Likelihood Estimation Steven Haker, William M. Wells III, Simon K. Warfield, Ion-Florin Talos, Jui G. Bhagwat, Daniel Goldberg-Zimring,
Automatic Ascending Aorta Detection in CTA Datasets
 Automatic Ascending Aorta Detection in CTA Datasets Stefan C. Saur 1, Caroline Kühnel 2, Tobias Boskamp 2, Gábor Székely 1, Philippe Cattin 1,3 1 Computer Vision Laboratory, ETH Zurich, 8092 Zurich, Switzerland
Automatic Ascending Aorta Detection in CTA Datasets Stefan C. Saur 1, Caroline Kühnel 2, Tobias Boskamp 2, Gábor Székely 1, Philippe Cattin 1,3 1 Computer Vision Laboratory, ETH Zurich, 8092 Zurich, Switzerland
3D Volume Mesh Generation of Human Organs Using Surface Geometries Created from the Visible Human Data Set
 3D Volume Mesh Generation of Human Organs Using Surface Geometries Created from the Visible Human Data Set John M. Sullivan, Jr., Ziji Wu, and Anand Kulkarni Worcester Polytechnic Institute Worcester,
3D Volume Mesh Generation of Human Organs Using Surface Geometries Created from the Visible Human Data Set John M. Sullivan, Jr., Ziji Wu, and Anand Kulkarni Worcester Polytechnic Institute Worcester,
Automatic Optimization of Segmentation Algorithms Through Simultaneous Truth and Performance Level Estimation (STAPLE)
 Automatic Optimization of Segmentation Algorithms Through Simultaneous Truth and Performance Level Estimation (STAPLE) Mahnaz Maddah, Kelly H. Zou, William M. Wells, Ron Kikinis, and Simon K. Warfield
Automatic Optimization of Segmentation Algorithms Through Simultaneous Truth and Performance Level Estimation (STAPLE) Mahnaz Maddah, Kelly H. Zou, William M. Wells, Ron Kikinis, and Simon K. Warfield
Flux Driven Automatic Centerline Extraction
 Flux Driven Automatic Centerline Extraction Sylvain Bouix a,b,, Kaleem Siddiqi c Allen Tannenbaum d a Department of Psychiatry, Boston VA Healthcare System, Harvard Medical School b Surgical Planning Laboratory,
Flux Driven Automatic Centerline Extraction Sylvain Bouix a,b,, Kaleem Siddiqi c Allen Tannenbaum d a Department of Psychiatry, Boston VA Healthcare System, Harvard Medical School b Surgical Planning Laboratory,
Evaluation of Hessian-based filters to enhance the axis of coronary arteries in CT images
 International Congress Series 1256 (2003) 1191 1196 Evaluation of Hessian-based filters to enhance the axis of coronary arteries in CT images S.D. Olabarriaga a, *, M. Breeuwer b, W.J. Niessen a a University
International Congress Series 1256 (2003) 1191 1196 Evaluation of Hessian-based filters to enhance the axis of coronary arteries in CT images S.D. Olabarriaga a, *, M. Breeuwer b, W.J. Niessen a a University
Three Dimensional Segmentation of Intravascular Ultrasound Data
 Three Dimensional Segmentation of Intravascular Ultrasound Data Marc Wennogle 1 and William Hoff 2 1 Veran Medical Technologies, Nashville, Tennessee marc.wennogle@veranmedical.com 2 Colorado School of
Three Dimensional Segmentation of Intravascular Ultrasound Data Marc Wennogle 1 and William Hoff 2 1 Veran Medical Technologies, Nashville, Tennessee marc.wennogle@veranmedical.com 2 Colorado School of
NIH Public Access Author Manuscript Proc Int Conf Image Proc. Author manuscript; available in PMC 2013 May 03.
 NIH Public Access Author Manuscript Published in final edited form as: Proc Int Conf Image Proc. 2008 ; : 241 244. doi:10.1109/icip.2008.4711736. TRACKING THROUGH CHANGES IN SCALE Shawn Lankton 1, James
NIH Public Access Author Manuscript Published in final edited form as: Proc Int Conf Image Proc. 2008 ; : 241 244. doi:10.1109/icip.2008.4711736. TRACKING THROUGH CHANGES IN SCALE Shawn Lankton 1, James
Characterizing the Shape of Anatomical Structures with Poisson s Equation
 Characterizing the Shape of Anatomical Structures with Poisson s Equation Haissam Haidar 1,2, Sylvain Bouix 2,3, James Levitt 2,3, Chandley Dickey 2,3, Robert W. McCarley 2,3, Martha E. Shenton 2,3 and
Characterizing the Shape of Anatomical Structures with Poisson s Equation Haissam Haidar 1,2, Sylvain Bouix 2,3, James Levitt 2,3, Chandley Dickey 2,3, Robert W. McCarley 2,3, Martha E. Shenton 2,3 and
Computational Radiology Lab, Children s Hospital, Harvard Medical School, Boston, MA.
 Shape prior integration in discrete optimization segmentation algorithms M. Freiman Computational Radiology Lab, Children s Hospital, Harvard Medical School, Boston, MA. Email: moti.freiman@childrens.harvard.edu
Shape prior integration in discrete optimization segmentation algorithms M. Freiman Computational Radiology Lab, Children s Hospital, Harvard Medical School, Boston, MA. Email: moti.freiman@childrens.harvard.edu
Non-linear Model Fitting to Parameterize Diseased Blood Vessels
 Non-linear Model Fitting to Parameterize Diseased Blood Vessels Alexandra La Cruz Vienna University of Technology Miloš Šrámek Austrian Academy of Sciences Matúš Straka Austrian Academy of Sciences Eduard
Non-linear Model Fitting to Parameterize Diseased Blood Vessels Alexandra La Cruz Vienna University of Technology Miloš Šrámek Austrian Academy of Sciences Matúš Straka Austrian Academy of Sciences Eduard
Blood Vessel Visualization on CT Data
 WDS'12 Proceedings of Contributed Papers, Part I, 88 93, 2012. ISBN 978-80-7378-224-5 MATFYZPRESS Blood Vessel Visualization on CT Data J. Dupej Charles University Prague, Faculty of Mathematics and Physics,
WDS'12 Proceedings of Contributed Papers, Part I, 88 93, 2012. ISBN 978-80-7378-224-5 MATFYZPRESS Blood Vessel Visualization on CT Data J. Dupej Charles University Prague, Faculty of Mathematics and Physics,
Blood Particle Trajectories in Phase-Contrast-MRI as Minimal Paths Computed with Anisotropic Fast Marching
 Blood Particle Trajectories in Phase-Contrast-MRI as Minimal Paths Computed with Anisotropic Fast Marching Michael Schwenke 1, Anja Hennemuth 1, Bernd Fischer 2, Ola Friman 1 1 Fraunhofer MEVIS, Institute
Blood Particle Trajectories in Phase-Contrast-MRI as Minimal Paths Computed with Anisotropic Fast Marching Michael Schwenke 1, Anja Hennemuth 1, Bernd Fischer 2, Ola Friman 1 1 Fraunhofer MEVIS, Institute
Quantitative IntraVascular UltraSound (QCU)
 Quantitative IntraVascular UltraSound (QCU) Authors: Jouke Dijkstra, Ph.D. and Johan H.C. Reiber, Ph.D., Leiden University Medical Center, Dept of Radiology, Leiden, The Netherlands Introduction: For decades,
Quantitative IntraVascular UltraSound (QCU) Authors: Jouke Dijkstra, Ph.D. and Johan H.C. Reiber, Ph.D., Leiden University Medical Center, Dept of Radiology, Leiden, The Netherlands Introduction: For decades,
Improving Accuracy in Coronary Lumen Segmentation via Explicit Calcium Exclusion, Learning-based Ray Detection and Surface Optimization
 Improving Accuracy in Coronary Lumen Segmentation via Explicit Calcium Exclusion, Learning-based Ray Detection and Surface Optimization Felix Lugauer a,b, Jingdan Zhang c, Yefeng Zheng c, Joachim Hornegger
Improving Accuracy in Coronary Lumen Segmentation via Explicit Calcium Exclusion, Learning-based Ray Detection and Surface Optimization Felix Lugauer a,b, Jingdan Zhang c, Yefeng Zheng c, Joachim Hornegger
IEEE TRANSACTIONS ON MEDICAL IMAGING, VOL. 23, NO. 10, OCTOBER Jian Chen and Amir A. Amini*, Senior Member, IEEE
 IEEE TRANSACTIONS ON MEDICAL IMAGING, VOL. 23, NO. 10, OCTOBER 2004 1251 Quantifying 3-D Vascular Structures in MRA Images Using Hybrid PDE and Geometric Deformable Models Jian Chen and Amir A. Amini*,
IEEE TRANSACTIONS ON MEDICAL IMAGING, VOL. 23, NO. 10, OCTOBER 2004 1251 Quantifying 3-D Vascular Structures in MRA Images Using Hybrid PDE and Geometric Deformable Models Jian Chen and Amir A. Amini*,
Segmentation of Doppler Carotid Ultrasound Image using Morphological Method and Classification by Neural Network
 Segmentation of Doppler Carotid Ultrasound Image using Morphological Method and Classification by Neural Network S.Usha 1, M.Karthik 2, M.Arthi 3 1&2 Assistant Professor (Sr.G), Department of EEE, Kongu
Segmentation of Doppler Carotid Ultrasound Image using Morphological Method and Classification by Neural Network S.Usha 1, M.Karthik 2, M.Arthi 3 1&2 Assistant Professor (Sr.G), Department of EEE, Kongu
Precise Lumen Segmentation in Coronary Computed Tomography Angiography
 Precise Lumen Segmentation in Coronary Computed Tomography Angiography Felix Lugauer,, Yefeng Zheng 3, Joachim Hornegger, and B. Michael Kelm Pattern Recognition Lab, University of Erlangen-Nuremberg,
Precise Lumen Segmentation in Coronary Computed Tomography Angiography Felix Lugauer,, Yefeng Zheng 3, Joachim Hornegger, and B. Michael Kelm Pattern Recognition Lab, University of Erlangen-Nuremberg,
Linköping University Post Print. Phase Based Level Set Segmentation of Blood Vessels
 Post Print Phase Based Level Set Segmentation of Blood Vessels Gunnar Läthén, Jimmy Jonasson and Magnus Borga N.B.: When citing this work, cite the original article. 2009 IEEE. Personal use of this material
Post Print Phase Based Level Set Segmentation of Blood Vessels Gunnar Läthén, Jimmy Jonasson and Magnus Borga N.B.: When citing this work, cite the original article. 2009 IEEE. Personal use of this material
Model Fitting. CS6240 Multimedia Analysis. Leow Wee Kheng. Department of Computer Science School of Computing National University of Singapore
 Model Fitting CS6240 Multimedia Analysis Leow Wee Kheng Department of Computer Science School of Computing National University of Singapore (CS6240) Model Fitting 1 / 34 Introduction Introduction Model
Model Fitting CS6240 Multimedia Analysis Leow Wee Kheng Department of Computer Science School of Computing National University of Singapore (CS6240) Model Fitting 1 / 34 Introduction Introduction Model
TUBULAR SURFACES EXTRACTION WITH MINIMAL ACTION SURFACES
 TUBULAR SURFACES EXTRACTION WITH MINIMAL ACTION SURFACES XIANGJUN GAO Department of Computer and Information Technology, Shangqiu Normal University, Shangqiu 476000, Henan, China ABSTRACT This paper presents
TUBULAR SURFACES EXTRACTION WITH MINIMAL ACTION SURFACES XIANGJUN GAO Department of Computer and Information Technology, Shangqiu Normal University, Shangqiu 476000, Henan, China ABSTRACT This paper presents
STATISTICS AND ANALYSIS OF SHAPE
 Control and Cybernetics vol. 36 (2007) No. 2 Book review: STATISTICS AND ANALYSIS OF SHAPE by H. Krim, A. Yezzi, Jr., eds. There are numerous definitions of a notion of shape of an object. These definitions
Control and Cybernetics vol. 36 (2007) No. 2 Book review: STATISTICS AND ANALYSIS OF SHAPE by H. Krim, A. Yezzi, Jr., eds. There are numerous definitions of a notion of shape of an object. These definitions
Towards Projector-based Visualization for Computer-assisted CABG at the Open Heart
 Towards Projector-based Visualization for Computer-assisted CABG at the Open Heart Christine Hartung 1, Claudia Gnahm 1, Stefan Sailer 1, Marcel Schenderlein 1, Reinhard Friedl 2, Martin Hoffmann 3, Klaus
Towards Projector-based Visualization for Computer-assisted CABG at the Open Heart Christine Hartung 1, Claudia Gnahm 1, Stefan Sailer 1, Marcel Schenderlein 1, Reinhard Friedl 2, Martin Hoffmann 3, Klaus
3D Guide Wire Navigation from Single Plane Fluoroscopic Images in Abdominal Catheterizations
 3D Guide Wire Navigation from Single Plane Fluoroscopic Images in Abdominal Catheterizations Martin Groher 2, Frederik Bender 1, Ali Khamene 3, Wolfgang Wein 3, Tim Hauke Heibel 2, Nassir Navab 2 1 Siemens
3D Guide Wire Navigation from Single Plane Fluoroscopic Images in Abdominal Catheterizations Martin Groher 2, Frederik Bender 1, Ali Khamene 3, Wolfgang Wein 3, Tim Hauke Heibel 2, Nassir Navab 2 1 Siemens
Nearly automatic vessels segmentation using graph-based energy minimization
 Nearly automatic vessels segmentation using graph-based energy minimization Release 1.00 M. Freiman 1, J. Frank 1, L. Weizman 1 E. Nammer 2, O. Shilon 2,, L. Joskowicz 1 and J. Sosna 3 July 16, 2009 1
Nearly automatic vessels segmentation using graph-based energy minimization Release 1.00 M. Freiman 1, J. Frank 1, L. Weizman 1 E. Nammer 2, O. Shilon 2,, L. Joskowicz 1 and J. Sosna 3 July 16, 2009 1
Three-dimensional synthetic blood vessel generation using stochastic L-systems
 Three-dimensional synthetic blood vessel generation using stochastic L-systems Miguel A. Galarreta-Valverde a, Maysa M. G. Macedo a, Choukri Mekkaoui b and Marcel P. Jackowski* a a Department of Computer
Three-dimensional synthetic blood vessel generation using stochastic L-systems Miguel A. Galarreta-Valverde a, Maysa M. G. Macedo a, Choukri Mekkaoui b and Marcel P. Jackowski* a a Department of Computer
A fast breast nonlinear elastography reconstruction technique using the Veronda-Westman model
 A fast breast nonlinear elastography reconstruction technique using the Veronda-Westman model Mohammadhosein Amooshahi a and Abbas Samani abc a Department of Electrical & Computer Engineering, University
A fast breast nonlinear elastography reconstruction technique using the Veronda-Westman model Mohammadhosein Amooshahi a and Abbas Samani abc a Department of Electrical & Computer Engineering, University
Coronary Artery Calcium Quantification in Contrast-enhanced Computed Tomography Angiography
 Georgia State University ScholarWorks @ Georgia State University Computer Science Dissertations Department of Computer Science 12-18-2013 Coronary Artery Calcium Quantification in Contrast-enhanced Computed
Georgia State University ScholarWorks @ Georgia State University Computer Science Dissertations Department of Computer Science 12-18-2013 Coronary Artery Calcium Quantification in Contrast-enhanced Computed
Decomposing and Sketching 3D Objects by Curve Skeleton Processing
 Decomposing and Sketching 3D Objects by Curve Skeleton Processing Luca Serino, Carlo Arcelli, and Gabriella Sanniti di Baja Institute of Cybernetics E. Caianiello, CNR, Naples, Italy {l.serino,c.arcelli,g.sannitidibaja}@cib.na.cnr.it
Decomposing and Sketching 3D Objects by Curve Skeleton Processing Luca Serino, Carlo Arcelli, and Gabriella Sanniti di Baja Institute of Cybernetics E. Caianiello, CNR, Naples, Italy {l.serino,c.arcelli,g.sannitidibaja}@cib.na.cnr.it
MODELING MIXED BOUNDARY PROBLEMS WITH THE COMPLEX VARIABLE BOUNDARY ELEMENT METHOD (CVBEM) USING MATLAB AND MATHEMATICA
 A. N. Johnson et al., Int. J. Comp. Meth. and Exp. Meas., Vol. 3, No. 3 (2015) 269 278 MODELING MIXED BOUNDARY PROBLEMS WITH THE COMPLEX VARIABLE BOUNDARY ELEMENT METHOD (CVBEM) USING MATLAB AND MATHEMATICA
A. N. Johnson et al., Int. J. Comp. Meth. and Exp. Meas., Vol. 3, No. 3 (2015) 269 278 MODELING MIXED BOUNDARY PROBLEMS WITH THE COMPLEX VARIABLE BOUNDARY ELEMENT METHOD (CVBEM) USING MATLAB AND MATHEMATICA
Image-Based Device Tracking for the Co-registration of Angiography and Intravascular Ultrasound Images
 Image-Based Device Tracking for the Co-registration of Angiography and Intravascular Ultrasound Images Peng Wang 1, Terrence Chen 1, Olivier Ecabert 2, Simone Prummer 2, Martin Ostermeier 2, and Dorin
Image-Based Device Tracking for the Co-registration of Angiography and Intravascular Ultrasound Images Peng Wang 1, Terrence Chen 1, Olivier Ecabert 2, Simone Prummer 2, Martin Ostermeier 2, and Dorin
Research Proposal: Computational Geometry with Applications on Medical Images
 Research Proposal: Computational Geometry with Applications on Medical Images MEI-HENG YUEH yueh@nctu.edu.tw National Chiao Tung University 1 Introduction My research mainly focuses on the issues of computational
Research Proposal: Computational Geometry with Applications on Medical Images MEI-HENG YUEH yueh@nctu.edu.tw National Chiao Tung University 1 Introduction My research mainly focuses on the issues of computational
Accurate Quantification of Small-Diameter Tubular Structures in Isotropic CT Volume Data Based on Multiscale Line Filter Responses
 Accurate Quantification of Small-Diameter Tubular Structures in Isotropic CT Volume Data Based on Multiscale Line Filter Responses Yoshinobu Sato 1, Shuji Yamamoto 2, and Shinichi Tamura 1 1 Division of
Accurate Quantification of Small-Diameter Tubular Structures in Isotropic CT Volume Data Based on Multiscale Line Filter Responses Yoshinobu Sato 1, Shuji Yamamoto 2, and Shinichi Tamura 1 1 Division of
Vessel Segmentation Using A Shape Driven Flow
 Vessel Segmentation Using A Shape Driven Flow Delphine Nain, Anthony Yezzi and Greg Turk Georgia Institute of Technology, Atlanta GA 30332, USA {delfin, turk}@cc.gatech.edu, ayezzi@ece.gatech.edu Abstract.
Vessel Segmentation Using A Shape Driven Flow Delphine Nain, Anthony Yezzi and Greg Turk Georgia Institute of Technology, Atlanta GA 30332, USA {delfin, turk}@cc.gatech.edu, ayezzi@ece.gatech.edu Abstract.
Efficient 3D-3D Vascular Registration Based on Multiple Orthogonal 2D Projections
 Efficient 3D-3D Vascular Registration Based on Multiple Orthogonal 2D Projections Ho-Ming Chan and Albert C. S. Chung Dept. of Computer Science, Hong Kong University of Science and Technology, HK {hmchan,achung}@cs.ust.hk
Efficient 3D-3D Vascular Registration Based on Multiple Orthogonal 2D Projections Ho-Ming Chan and Albert C. S. Chung Dept. of Computer Science, Hong Kong University of Science and Technology, HK {hmchan,achung}@cs.ust.hk
Automatic Lung Surface Registration Using Selective Distance Measure in Temporal CT Scans
 Automatic Lung Surface Registration Using Selective Distance Measure in Temporal CT Scans Helen Hong 1, Jeongjin Lee 2, Kyung Won Lee 3, and Yeong Gil Shin 2 1 School of Electrical Engineering and Computer
Automatic Lung Surface Registration Using Selective Distance Measure in Temporal CT Scans Helen Hong 1, Jeongjin Lee 2, Kyung Won Lee 3, and Yeong Gil Shin 2 1 School of Electrical Engineering and Computer
Segmentation and Modeling of the Spinal Cord for Reality-based Surgical Simulator
 Segmentation and Modeling of the Spinal Cord for Reality-based Surgical Simulator Li X.C.,, Chui C. K.,, and Ong S. H.,* Dept. of Electrical and Computer Engineering Dept. of Mechanical Engineering, National
Segmentation and Modeling of the Spinal Cord for Reality-based Surgical Simulator Li X.C.,, Chui C. K.,, and Ong S. H.,* Dept. of Electrical and Computer Engineering Dept. of Mechanical Engineering, National
Performance Evaluation Metrics and Statistics for Positional Tracker Evaluation
 Performance Evaluation Metrics and Statistics for Positional Tracker Evaluation Chris J. Needham and Roger D. Boyle School of Computing, The University of Leeds, Leeds, LS2 9JT, UK {chrisn,roger}@comp.leeds.ac.uk
Performance Evaluation Metrics and Statistics for Positional Tracker Evaluation Chris J. Needham and Roger D. Boyle School of Computing, The University of Leeds, Leeds, LS2 9JT, UK {chrisn,roger}@comp.leeds.ac.uk
Nonrigid Motion Compensation of Free Breathing Acquired Myocardial Perfusion Data
 Nonrigid Motion Compensation of Free Breathing Acquired Myocardial Perfusion Data Gert Wollny 1, Peter Kellman 2, Andrés Santos 1,3, María-Jesus Ledesma 1,3 1 Biomedical Imaging Technologies, Department
Nonrigid Motion Compensation of Free Breathing Acquired Myocardial Perfusion Data Gert Wollny 1, Peter Kellman 2, Andrés Santos 1,3, María-Jesus Ledesma 1,3 1 Biomedical Imaging Technologies, Department
2D-3D Registration using Gradient-based MI for Image Guided Surgery Systems
 2D-3D Registration using Gradient-based MI for Image Guided Surgery Systems Yeny Yim 1*, Xuanyi Chen 1, Mike Wakid 1, Steve Bielamowicz 2, James Hahn 1 1 Department of Computer Science, The George Washington
2D-3D Registration using Gradient-based MI for Image Guided Surgery Systems Yeny Yim 1*, Xuanyi Chen 1, Mike Wakid 1, Steve Bielamowicz 2, James Hahn 1 1 Department of Computer Science, The George Washington
Contrast Enhancement with Dual Energy CT for the Assessment of Atherosclerosis
 Contrast Enhancement with Dual Energy CT for the Assessment of Atherosclerosis Stefan C. Saur 1, Hatem Alkadhi 2, Luca Regazzoni 1, Simon Eugster 1, Gábor Székely 1, Philippe Cattin 1,3 1 Computer Vision
Contrast Enhancement with Dual Energy CT for the Assessment of Atherosclerosis Stefan C. Saur 1, Hatem Alkadhi 2, Luca Regazzoni 1, Simon Eugster 1, Gábor Székely 1, Philippe Cattin 1,3 1 Computer Vision
Motion Compensation from Short-Scan Data in Cardiac CT
 Motion Compensation from Short-Scan Data in Cardiac CT Juliane Hahn 1,2, Thomas Allmendinger 1, Herbert Bruder 1, and Marc Kachelrieß 2 1 Siemens Healthcare GmbH, Forchheim, Germany 2 German Cancer Research
Motion Compensation from Short-Scan Data in Cardiac CT Juliane Hahn 1,2, Thomas Allmendinger 1, Herbert Bruder 1, and Marc Kachelrieß 2 1 Siemens Healthcare GmbH, Forchheim, Germany 2 German Cancer Research
Computer Aided Diagnosis Based on Medical Image Processing and Artificial Intelligence Methods
 International Journal of Information and Computation Technology. ISSN 0974-2239 Volume 3, Number 9 (2013), pp. 887-892 International Research Publications House http://www. irphouse.com /ijict.htm Computer
International Journal of Information and Computation Technology. ISSN 0974-2239 Volume 3, Number 9 (2013), pp. 887-892 International Research Publications House http://www. irphouse.com /ijict.htm Computer
Author Manuscript Faculty of Biology and Medicine Publication
 Serveur Académique Lausannois SERVAL serval.unil.ch Author Manuscript Faculty of Biology and Medicine Publication This paper has been peer-reviewed but dos not include the final publisher proof-corrections
Serveur Académique Lausannois SERVAL serval.unil.ch Author Manuscript Faculty of Biology and Medicine Publication This paper has been peer-reviewed but dos not include the final publisher proof-corrections
Vitrea 2. CT Coronary Artery Analysis. version 4.0. User Guide VPMC-8144B
 Vitrea 2 version 4.0 CT Coronary Artery Analysis User Guide VPMC-8144B Vital Images, Inc. 2007 Protected by U.S. Patents 5,986,662; 6,130,671; 6,219,059; 7,031,504; Other Patents Pending in the U.S. and
Vitrea 2 version 4.0 CT Coronary Artery Analysis User Guide VPMC-8144B Vital Images, Inc. 2007 Protected by U.S. Patents 5,986,662; 6,130,671; 6,219,059; 7,031,504; Other Patents Pending in the U.S. and
Research Article Automated Segmentation of Coronary Arteries Based on Statistical Region Growing and Heuristic Decision Method
 BioMed Research International Volume 2016, Article ID 3530251, 7 pages http://dx.doi.org/10.1155/2016/3530251 Research Article Automated Segmentation of Coronary Arteries Based on Statistical Region Growing
BioMed Research International Volume 2016, Article ID 3530251, 7 pages http://dx.doi.org/10.1155/2016/3530251 Research Article Automated Segmentation of Coronary Arteries Based on Statistical Region Growing
Automatic Quantification of DTI Parameters along Fiber Bundles
 Automatic Quantification of DTI Parameters along Fiber Bundles Jan Klein 1, Simon Hermann 1, Olaf Konrad 1, Horst K. Hahn 1, and Heinz-Otto Peitgen 1 1 MeVis Research, 28359 Bremen Email: klein@mevis.de
Automatic Quantification of DTI Parameters along Fiber Bundles Jan Klein 1, Simon Hermann 1, Olaf Konrad 1, Horst K. Hahn 1, and Heinz-Otto Peitgen 1 1 MeVis Research, 28359 Bremen Email: klein@mevis.de
Digital Image Processing Fundamentals
 Ioannis Pitas Digital Image Processing Fundamentals Chapter 7 Shape Description Answers to the Chapter Questions Thessaloniki 1998 Chapter 7: Shape description 7.1 Introduction 1. Why is invariance to
Ioannis Pitas Digital Image Processing Fundamentals Chapter 7 Shape Description Answers to the Chapter Questions Thessaloniki 1998 Chapter 7: Shape description 7.1 Introduction 1. Why is invariance to
City, University of London Institutional Repository
 City Research Online City, University of London Institutional Repository Citation: Wang, Yin (2011). Blood Vessel Segmentation and shape analysis for quantification of Coronary Artery Stenosis in CT Angiography.
City Research Online City, University of London Institutional Repository Citation: Wang, Yin (2011). Blood Vessel Segmentation and shape analysis for quantification of Coronary Artery Stenosis in CT Angiography.
A Modified SVD-DCT Method for Enhancement of Low Contrast Satellite Images
 A Modified SVD-DCT Method for Enhancement of Low Contrast Satellite Images G.Praveena 1, M.Venkatasrinu 2, 1 M.tech student, Department of Electronics and Communication Engineering, Madanapalle Institute
A Modified SVD-DCT Method for Enhancement of Low Contrast Satellite Images G.Praveena 1, M.Venkatasrinu 2, 1 M.tech student, Department of Electronics and Communication Engineering, Madanapalle Institute
Volumetric Analysis of the Heart from Tagged-MRI. Introduction & Background
 Volumetric Analysis of the Heart from Tagged-MRI Dimitris Metaxas Center for Computational Biomedicine, Imaging and Modeling (CBIM) Rutgers University, New Brunswick, NJ Collaboration with Dr. Leon Axel,
Volumetric Analysis of the Heart from Tagged-MRI Dimitris Metaxas Center for Computational Biomedicine, Imaging and Modeling (CBIM) Rutgers University, New Brunswick, NJ Collaboration with Dr. Leon Axel,
arxiv: v1 [cs.cv] 6 Jun 2017
![arxiv: v1 [cs.cv] 6 Jun 2017 arxiv: v1 [cs.cv] 6 Jun 2017](/thumbs/96/128307097.jpg) Volume Calculation of CT lung Lesions based on Halton Low-discrepancy Sequences Liansheng Wang a, Shusheng Li a, and Shuo Li b a Department of Computer Science, Xiamen University, Xiamen, China b Dept.
Volume Calculation of CT lung Lesions based on Halton Low-discrepancy Sequences Liansheng Wang a, Shusheng Li a, and Shuo Li b a Department of Computer Science, Xiamen University, Xiamen, China b Dept.
Segmentation of Intima Media Complex of Common Carotid Artery
 Segmentation of Intima Media Complex of Common Carotid Artery Harsha.J.Krishna 1, Kavya Bhadran 2, Krishnapriya Venugopal 3, Sruthi.P 4, Vaishnavi.V 5 U.G Scholars, Dept of Electronics and Biomedical Engineering,
Segmentation of Intima Media Complex of Common Carotid Artery Harsha.J.Krishna 1, Kavya Bhadran 2, Krishnapriya Venugopal 3, Sruthi.P 4, Vaishnavi.V 5 U.G Scholars, Dept of Electronics and Biomedical Engineering,
Performance Issues in Shape Classification
 Performance Issues in Shape Classification Samson J. Timoner 1, Pollina Golland 1, Ron Kikinis 2, Martha E. Shenton 3, W. Eric L. Grimson 1, and William M. Wells III 1,2 1 MIT AI Laboratory, Cambridge
Performance Issues in Shape Classification Samson J. Timoner 1, Pollina Golland 1, Ron Kikinis 2, Martha E. Shenton 3, W. Eric L. Grimson 1, and William M. Wells III 1,2 1 MIT AI Laboratory, Cambridge
The VesselGlyph: Focus & Context Visualization in CT-Angiography
 The VesselGlyph: Focus & Context Visualization in CT-Angiography Matúš Straka M. Šrámek, A. La Cruz E. Gröller, D. Fleischmann Contents Motivation:» Why again a new visualization method for vessel data?
The VesselGlyph: Focus & Context Visualization in CT-Angiography Matúš Straka M. Šrámek, A. La Cruz E. Gröller, D. Fleischmann Contents Motivation:» Why again a new visualization method for vessel data?
Automated segmentation methods for liver analysis in oncology applications
 University of Szeged Department of Image Processing and Computer Graphics Automated segmentation methods for liver analysis in oncology applications Ph. D. Thesis László Ruskó Thesis Advisor Dr. Antal
University of Szeged Department of Image Processing and Computer Graphics Automated segmentation methods for liver analysis in oncology applications Ph. D. Thesis László Ruskó Thesis Advisor Dr. Antal
Calculating the Distance Map for Binary Sampled Data
 MITSUBISHI ELECTRIC RESEARCH LABORATORIES http://www.merl.com Calculating the Distance Map for Binary Sampled Data Sarah F. Frisken Gibson TR99-6 December 999 Abstract High quality rendering and physics-based
MITSUBISHI ELECTRIC RESEARCH LABORATORIES http://www.merl.com Calculating the Distance Map for Binary Sampled Data Sarah F. Frisken Gibson TR99-6 December 999 Abstract High quality rendering and physics-based
Triangulation Based Volume Calculation Andrew Y.T Kudowor and George Taylor Department of Geomatics University of Newcastle
 Triangulation Based Volume Calculation Andrew Y.T Kudowor and George Taylor Department of Geomatics University of Newcastle ABSTRACT Delaunay triangulation is a commonly used method for generating surface
Triangulation Based Volume Calculation Andrew Y.T Kudowor and George Taylor Department of Geomatics University of Newcastle ABSTRACT Delaunay triangulation is a commonly used method for generating surface
