Completing the Schedule of Events Cost Attribution Template (SoECAT)
|
|
|
- Madison King
- 5 years ago
- Views:
Transcription
1 Completing the Schedule of Events Cost Attribution Template (SoECAT) Introduction Please read the following guidance before completing this template. More specific guidance is given on each tab within the template. You should seek support from your sponsor in completing the template and additionally, where appropriate and applicable (e.g. if your sponsor is non-nhs), from your lead NHS R&D Office. Where you intend to open NHS research sites in England, support is also available from your Lead Local Clinical Research Network (LCRN) and sign-off by a named AcoRD expert in your Lead LCRN is needed before submitting your funding application (your funder will advise on how and when to submit). This SoECAT is designed to help appropriately attribute the costs associated with all SITE LEVEL activities at the time that application is made for research project grant/award funding. By site level we mean the costs that will be incurred by research sites, i.e. excluding costs incurred by the sponsor in managing the study, e.g. the cost of employing study managers, coordinators, monitors, etc. The DH AcoRD guidance, which provides the basis for the transparent and consistent cost attribution on which this tool builds, should be consulted before completing this template and is available here. It is also recommended that you complete the AcoRD cost attribution training available here before completing the template. Completing this tool 1. Completing the Study Information Tab Further guidance is given within this tab in the excel template. NOTE: Key functionality within the tool is disabled until question 3 is answered. 2. Completing the Standard of Care, General Activities and Per-Participant Arm tabs Further guidance is given within each tab (guidance on the per-participant tabs is given on the Arm1 tab only but applies to all per-participant Arm tabs). Each tab is set out as a Schedule of Events. A standard list of procedures and investigations is provided, and may be selected from drop-down menus as follows: 1 of 6
2 2.1. How to select activities 1 Check to see where the activity appears on the List of Activities tab. (If it doesn t appear, follow guidance on adding activities.) 2 Take a note of the Area of Activity. 2 of 6
3 3 Go to the tab being completed, select the Area of Activity from the drop-down list do this before selecting the Specific Activity. 4 Select the Specific Activity from the drop-down list. It is also possible to add procedures and/or investigations that are not present in the drop-downs 3 of 6
4 2.2 How to add new activities 1 Go to Non Tariff Costs tab and enter the new activities. Also select relevant activity type (investigation or procedure) 2 If the new activity is an investigation enter the associated costs 4 of 6
5 5 of 6
6 Once you have selected a procedure on any tab, you should provide in the 'duration' column an estimate for the time that activity will take, in minutes. For investigations the duration column will be greyed out and should not be completed, (procedure costs are calculated on the basis of staff time, whereas investigations have fixed costs). You should then choose a staff-type from the dropdown menu in the 'undertaken by' column. This should be completed both for procedures and investigations. Next you should indicate when each activity will occur. For the standard of care tab, you may either manually enter an 'x' or select 'x' from the drop-down menus. In the General Activity and Per Participant tabs, you should indicate that an activity is to occur by ascribing to it a cost attribution from the drop-down menu in the appropriate cell/s. An activity might occur once, or more than once, and should appear in each column where it is planned to occur. Cells in columns where the activity will not occur should be left blank. Cost attributions should be as per the AcoRD guidance: 3.Completing the Summary tab Further guidance is given within the summary tab in the template. END OF GUIDANCE 6 of 6
NAU New Project Submission. Guidance for creating a new project
 NAU New Project Submission Guidance for creating a new project Go to IRBNet Website address: https://www.irbnet.org/release/index.html Login to your account Enter your Username and Password and select
NAU New Project Submission Guidance for creating a new project Go to IRBNet Website address: https://www.irbnet.org/release/index.html Login to your account Enter your Username and Password and select
Date Revision Author Description /04/2017 SM Document creation /06/2017 SM Search by site code
 CPMS How to Guide Adding sites to the CPMS study record V1.1 Assumptions & Prerequisites Adding a New site to the Study Record ADD SITES LINKED TO TRUSTS/CCGS Option ADD OTHER SITES Option ADD SITES BY
CPMS How to Guide Adding sites to the CPMS study record V1.1 Assumptions & Prerequisites Adding a New site to the Study Record ADD SITES LINKED TO TRUSTS/CCGS Option ADD OTHER SITES Option ADD SITES BY
Date Revision Author Description /02/2016 CW Document creation /02/2106 CW Addition of Appendix 3 & 4
 CPMS How to Guide Recruitment Data Upload Version 1.6 Appendix 5: How to upload Non UK research activity Assumptions & Prerequisites Logging in Prefix: How to find a study you own Using the main search
CPMS How to Guide Recruitment Data Upload Version 1.6 Appendix 5: How to upload Non UK research activity Assumptions & Prerequisites Logging in Prefix: How to find a study you own Using the main search
Introduction to. Sponsored by the Pediatric Research Office (PRO)
 Introduction to Sponsored by the Pediatric Research Office (PRO) Agenda Overview of REDCap Basic project work flow Creating a project with REDCap Interactive demonstration Questions and Answers Overview
Introduction to Sponsored by the Pediatric Research Office (PRO) Agenda Overview of REDCap Basic project work flow Creating a project with REDCap Interactive demonstration Questions and Answers Overview
Submitting an application
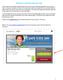
 Submitting an application Procedure 1. If you would like to submit an application via the DAAD Portal, please start by selecting your chosen programme in our database for scholarship holders or academic
Submitting an application Procedure 1. If you would like to submit an application via the DAAD Portal, please start by selecting your chosen programme in our database for scholarship holders or academic
NETSCC MIS Brief Supporting Role Signatory
 NETSCC MIS Brief Supporting Role Signatory NETSCC MIS Supporting Role Signatory Process Logging In to MIS Accept Full Application Participation (Role)/Accept Revised Application Participation (Role) Provide
NETSCC MIS Brief Supporting Role Signatory NETSCC MIS Supporting Role Signatory Process Logging In to MIS Accept Full Application Participation (Role)/Accept Revised Application Participation (Role) Provide
CTP SUBMISSION PLATFORM
 CTP SUBMISSION PLATFORM INSTRUCTION DOCUMENT Version Control Two notes of clarification added regarding data cut off and completion requirements The Submission Dashboard, p 10 Using the Spreadsheet Download,
CTP SUBMISSION PLATFORM INSTRUCTION DOCUMENT Version Control Two notes of clarification added regarding data cut off and completion requirements The Submission Dashboard, p 10 Using the Spreadsheet Download,
Claims Reporting Suite. Fact Sheet Nil reserve claims
 Claims Reporting Suite Fact Sheet Nil reserve claims This document contains information regarding the ECF Nil Reserve app, and guidance as to how it may be used. If you require any further information,
Claims Reporting Suite Fact Sheet Nil reserve claims This document contains information regarding the ECF Nil Reserve app, and guidance as to how it may be used. If you require any further information,
pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation
 pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation Avelumab (Bavencio) for metastatic Merkel Cell Carcinoma March 21, 2018 3 Feedback on perc
pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation Avelumab (Bavencio) for metastatic Merkel Cell Carcinoma March 21, 2018 3 Feedback on perc
WISER. Wake Integrated Solution for Enterprise Research. Protocol Creation, Activation, and Management. For Non-Oncology Studies.
 WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Non-Oncology Studies Version 6/11/2018 TRAINING MANUAL WELCOME to WISER! Navigation and Home Page
WISER Wake Integrated Solution for Enterprise Research Protocol Creation, Activation, and Management For Non-Oncology Studies Version 6/11/2018 TRAINING MANUAL WELCOME to WISER! Navigation and Home Page
Getting started with Moodle Student guide
 Getting started with Moodle Student guide This guide will help you to get started using Moodle and generally familiarise yourself with the new interface. Use the guide to learn how to: 1. Log in to Moodle
Getting started with Moodle Student guide This guide will help you to get started using Moodle and generally familiarise yourself with the new interface. Use the guide to learn how to: 1. Log in to Moodle
Daily Position Reporting System (DPRS) User Guide
 Daily Position Reporting System (DPRS) User Guide Please respond to: Market Surveillance +44 (0) 207 113 8203 THE LONDON METAL EXCHANGE 10 Finsbury Square, London EC2A 1AJ Tel +44 (0)20 7113 8888 Registered
Daily Position Reporting System (DPRS) User Guide Please respond to: Market Surveillance +44 (0) 207 113 8203 THE LONDON METAL EXCHANGE 10 Finsbury Square, London EC2A 1AJ Tel +44 (0)20 7113 8888 Registered
State and District User Guide for PearsonAccess next State and District Testing Overview
 In this Guide This document contains information about PearsonAccess next for state and district users. It contains information about: viewing and editing school test coordinators creating and deleting
In this Guide This document contains information about PearsonAccess next for state and district users. It contains information about: viewing and editing school test coordinators creating and deleting
LOUGHBOROUGH UNIVERSITY RESEARCH OFFICE STANDARD OPERATING PROCEDURE. Loughborough University (LU) Research Office SOP 1027 LU
 LOUGHBOROUGH UNIVERSITY RESEARCH OFFICE STANDARD OPERATING PROCEDURE Loughborough University (LU) Research Office SOP 1027 LU Process for Writing Study Protocols for NHS Research Sponsored by Loughborough
LOUGHBOROUGH UNIVERSITY RESEARCH OFFICE STANDARD OPERATING PROCEDURE Loughborough University (LU) Research Office SOP 1027 LU Process for Writing Study Protocols for NHS Research Sponsored by Loughborough
Work Instruction Study Startup
 THE UNIVERSITY OF TEXAS HEALTH SCIENCE CENTER AT SAN ANTONIO Work Instruction Study Startup Velos - eresearch 9.2 Version: 2.0, 04/29/2015 Version 2.0, 04/29/2015 Revision History Version/Amendment #:
THE UNIVERSITY OF TEXAS HEALTH SCIENCE CENTER AT SAN ANTONIO Work Instruction Study Startup Velos - eresearch 9.2 Version: 2.0, 04/29/2015 Version 2.0, 04/29/2015 Revision History Version/Amendment #:
1. Access the Journal Entry Form via University Forms and Documents.
 Creating and Submitting a Journal Entry Purpose: Journal Entries may be completed to make corrections to posted expenses to include payroll, p-card, expense reports, purchase requisitions, and local funding
Creating and Submitting a Journal Entry Purpose: Journal Entries may be completed to make corrections to posted expenses to include payroll, p-card, expense reports, purchase requisitions, and local funding
LATE COST TRANSFER EXCEPTION REQUEST WEB FORM GUIDE
 LATE COST TRANSFER EXCEPTION REQUEST WEB FORM GUIDE The Office of Research Administration (ORA) has updated their Late Cost Transfer Exception Request Form to a web form. This web form is accessible under
LATE COST TRANSFER EXCEPTION REQUEST WEB FORM GUIDE The Office of Research Administration (ORA) has updated their Late Cost Transfer Exception Request Form to a web form. This web form is accessible under
copying a protocol in Kc irb training guide
 copying a protocol in Kc irb training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 about this guide 4 copying a protocol 5 protocol actions
copying a protocol in Kc irb training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 about this guide 4 copying a protocol 5 protocol actions
Dental Contract Reform: Prototypes Prototype expression of interest form: non-pilot NHS dental contracts Completion notes
 Dental Contract Reform: Prototypes Prototype expression of interest form: non-pilot NHS dental contracts Completion notes This document sets out the completion notes for non-pilot NHS dental contracts
Dental Contract Reform: Prototypes Prototype expression of interest form: non-pilot NHS dental contracts Completion notes This document sets out the completion notes for non-pilot NHS dental contracts
BIC Innovation R & D Logger Mobile App
 BIC Innovation R & D Logger Mobile App This app is to help you as a producer or manager of a business to record research and development activity. Why? The government rightly supports R&D activity and
BIC Innovation R & D Logger Mobile App This app is to help you as a producer or manager of a business to record research and development activity. Why? The government rightly supports R&D activity and
Feedback on application for candidacy part II and consultant report templates Excel application
 Feedback on application for candidacy part II and consultant report templates Excel application Instructions for readers, consultants and IB staff July 2011 Page 1 International Baccalaureate Organization
Feedback on application for candidacy part II and consultant report templates Excel application Instructions for readers, consultants and IB staff July 2011 Page 1 International Baccalaureate Organization
Quick Start Guide To Mobility Tool+ For Key Action 1 School Staff Mobility Projects Version 1
 Quick Start Guide To Mobility Tool+ For Key Action 1 School Staff Mobility Projects Introduction This step by step guide has been produced by the UK National Agency to help beneficiaries of Key Action
Quick Start Guide To Mobility Tool+ For Key Action 1 School Staff Mobility Projects Introduction This step by step guide has been produced by the UK National Agency to help beneficiaries of Key Action
pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation (Provincial Advisory Group [PAG])
![pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation (Provincial Advisory Group [PAG]) pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation (Provincial Advisory Group [PAG])](/thumbs/90/101387747.jpg) pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation (Provincial Advisory Group [PAG]) Nivolumab-Ipilimumab for Renal Cell Carcinoma November
pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation (Provincial Advisory Group [PAG]) Nivolumab-Ipilimumab for Renal Cell Carcinoma November
UNDERSTANDING THE UG RESEARCH GRANTS ONLINE APPLICATION SYSTEM (User Guide)
 UNIVERSITY OF GHANA OFFICE OF RESEARCH, INNOVATION AND DEVELOPMENT (ORID) UNDERSTANDING THE UG RESEARCH GRANTS ONLINE APPLICATION SYSTEM (User Guide) September 2016 1. ABOUT THE ONLINE APPLICATION SYSTEM
UNIVERSITY OF GHANA OFFICE OF RESEARCH, INNOVATION AND DEVELOPMENT (ORID) UNDERSTANDING THE UG RESEARCH GRANTS ONLINE APPLICATION SYSTEM (User Guide) September 2016 1. ABOUT THE ONLINE APPLICATION SYSTEM
https://s.qualtrics.com/controlpanel/ajax.php?action=getsurveyprintpre... IASLC Conflict of Interest (COI) Disclosure Form for Individuals
 1 of 6 10/5/2015 12:58 PM Introduction IASLC Conflict of Interest (COI) Disclosure Form for Individuals - 2015-2017 This form and policy applies to all persons who: are members, employees, or staff of
1 of 6 10/5/2015 12:58 PM Introduction IASLC Conflict of Interest (COI) Disclosure Form for Individuals - 2015-2017 This form and policy applies to all persons who: are members, employees, or staff of
Date Revision Author Description /6/2016 SM Update to Adding a New site to the Study Record. Published.
 CPMS How to Guide Locating the study you want and adding new sites. V1.2 Assumptions & Prerequisites: Logging in The Home screen Using the main search box Using the filters Exporting your filtered search
CPMS How to Guide Locating the study you want and adding new sites. V1.2 Assumptions & Prerequisites: Logging in The Home screen Using the main search box Using the filters Exporting your filtered search
Understanding the Main Screen and Dashboard The How Do I Check or Wiring Instructions Distribution and Customer Files upload links
 Understanding the Main Screen and Dashboard The PenChecks Pay Portal (P3) main screen and dashboard is shown below, with all the resources you need at a click of a button. As shown below, the left hand
Understanding the Main Screen and Dashboard The PenChecks Pay Portal (P3) main screen and dashboard is shown below, with all the resources you need at a click of a button. As shown below, the left hand
Pharmacy - Frequently Asked Questions
 Pharmacy - Frequently Asked Questions Published October 2017 Version 4 Copyright 2017Health and Social Care Information Centre. The Health and Social Care Information Centre is a non-departmental body
Pharmacy - Frequently Asked Questions Published October 2017 Version 4 Copyright 2017Health and Social Care Information Centre. The Health and Social Care Information Centre is a non-departmental body
User Guide Data Entry User
 User Guide Data Entry User Version 1.6 Page 1 of 22 Document control Version Description Release date Reason for change Reviewer 1.1 Data Entry User Guide 1.2 Data Entry User Guide 1.3 Data Entry User
User Guide Data Entry User Version 1.6 Page 1 of 22 Document control Version Description Release date Reason for change Reviewer 1.1 Data Entry User Guide 1.2 Data Entry User Guide 1.3 Data Entry User
Application form guidance
 Efficiency Research Programme 2018 This guidance document is to be read alongside the application form on AIMS in order to help applicants complete the form correctly. Please ensure that you have read
Efficiency Research Programme 2018 This guidance document is to be read alongside the application form on AIMS in order to help applicants complete the form correctly. Please ensure that you have read
SEVIS Real-Time Interface. Release 5.1 & Release 5.2 Training
 United States Department of State SEVIS Real-Time Interface Release 5.1 & Release 5.2 Training December 2005 Release 5.1 Enhancements J Program Sponsor Users 2 New Functionality in Release 5.1 for J Program
United States Department of State SEVIS Real-Time Interface Release 5.1 & Release 5.2 Training December 2005 Release 5.1 Enhancements J Program Sponsor Users 2 New Functionality in Release 5.1 for J Program
Communication plan template user guide
 Communication plan template user guide This user guide is designed to help you fill out the communication plan template docdm-22868. The diagram below provides an overview of the template. Each box represents
Communication plan template user guide This user guide is designed to help you fill out the communication plan template docdm-22868. The diagram below provides an overview of the template. Each box represents
JeffTrial Subject Registration Clinical Coordinator Training. Kimmel Cancer Center JeffTrials version 13.0 Ver. 1.2
 JeffTrial Subject Registration Clinical Coordinator Training Kimmel Cancer Center 1.2.14 JeffTrials version 13.0 Ver. 1.2 Coordinator Register a Patient 1. Log in to JeffTrial https://jefftrial.tjh.tju.edu/login
JeffTrial Subject Registration Clinical Coordinator Training Kimmel Cancer Center 1.2.14 JeffTrials version 13.0 Ver. 1.2 Coordinator Register a Patient 1. Log in to JeffTrial https://jefftrial.tjh.tju.edu/login
Instructions for the Hearst Foundations Online Grant Application
 Instructions for the Hearst Foundations Online Grant Application NOTE: All applicants should read this document thoroughly BEFORE starting the application process or seeking assistance from the Hearst
Instructions for the Hearst Foundations Online Grant Application NOTE: All applicants should read this document thoroughly BEFORE starting the application process or seeking assistance from the Hearst
Guide to Completing the Online Application
 Guide to Completing the Online Application Table of Contents 1. Before You Begin.......2 a. Invitation to Apply....2 b. Please Sign In.....2 c. Application Time Out...........3 d. Navigating the Online
Guide to Completing the Online Application Table of Contents 1. Before You Begin.......2 a. Invitation to Apply....2 b. Please Sign In.....2 c. Application Time Out...........3 d. Navigating the Online
How to Create and Submit Data Filings Using the Florida Office of Insurance Regulation Filing System (IRFS)
 The Florida Office of Insurance Regulation has launched the Insurance Regulation Filing System (IRFS) -- a new online application -- to replace the DCAM system. How to Create and Submit Data Filings Using
The Florida Office of Insurance Regulation has launched the Insurance Regulation Filing System (IRFS) -- a new online application -- to replace the DCAM system. How to Create and Submit Data Filings Using
Job Aid. Enter bid Using XML Upload Functionality
 Enter bid Using XML Upload Functionality Scenario: You need to respond to a bid using the XML upload functionality. The XML functionality allows your responses to be transferred automatically from an Excel
Enter bid Using XML Upload Functionality Scenario: You need to respond to a bid using the XML upload functionality. The XML functionality allows your responses to be transferred automatically from an Excel
How to Sign Off an Audit using the Online Audit Tool
 How to Sign Off an Audit using the Online Audit Tool ARC System Manager, September 2013 Contents Contents... 2 1 Accessing POPPI... 3 1.1 Placement Areas... 4 1.2 Audit Practice Placement Area Audits...
How to Sign Off an Audit using the Online Audit Tool ARC System Manager, September 2013 Contents Contents... 2 1 Accessing POPPI... 3 1.1 Placement Areas... 4 1.2 Audit Practice Placement Area Audits...
Section 1: Accessing the Extenuating Circumstances (ECs) task in e:vision
 Reporting Extenuating Circumstances (ECs) If you have experienced any difficulties such as illness, family problems or bereavement which may have affected your studies, you can submit an EC request via
Reporting Extenuating Circumstances (ECs) If you have experienced any difficulties such as illness, family problems or bereavement which may have affected your studies, you can submit an EC request via
Exporting Key Stage 2 Teacher Assessments Summer 2015
 Exporting Key Stage 2 Teacher Assessments Summer 2015 This HelpCard is about creating and exporting your Key Stage 2 TAs You must follow this HelpCard carefully in order to create and export your Key Stage
Exporting Key Stage 2 Teacher Assessments Summer 2015 This HelpCard is about creating and exporting your Key Stage 2 TAs You must follow this HelpCard carefully in order to create and export your Key Stage
You can access the AIMS user guide in the Related Links section at the top right of the page.
 Tender response form Click on any of the underlined headings to start your form. Once you have completed all mandatory questions within a section and moved to the next, you will see a tick appear on the
Tender response form Click on any of the underlined headings to start your form. Once you have completed all mandatory questions within a section and moved to the next, you will see a tick appear on the
This tutorial will guide a curator/user to create the files to upload phenotype experiment annotation and data onto T3.
 This tutorial will guide a curator/user to create the files to upload phenotype experiment annotation and data onto T3. Updated December 2012. Any feedback is welcome! Sections 1. Download the Template
This tutorial will guide a curator/user to create the files to upload phenotype experiment annotation and data onto T3. Updated December 2012. Any feedback is welcome! Sections 1. Download the Template
Visual Streamline FAQ
 Visual Streamline FAQ How does the Template Import Work? The Template Import routine enables you to import new inventory items, as well as New Supplier List Records, and make changes to existing Inventory
Visual Streamline FAQ How does the Template Import Work? The Template Import routine enables you to import new inventory items, as well as New Supplier List Records, and make changes to existing Inventory
Updated: 10/2016. Regional Meeting/ Specialty Conference. Symposium Organizer. Box & Workbook. User Guide
 Updated: 10/2016 Regional Meeting/ Specialty Conference Symposium Organizer Box & Workbook User Guide Table of Contents WHAT S NEW...2 Accepting/Rejecting Abstracts Creating Sessions Editing Abstracts
Updated: 10/2016 Regional Meeting/ Specialty Conference Symposium Organizer Box & Workbook User Guide Table of Contents WHAT S NEW...2 Accepting/Rejecting Abstracts Creating Sessions Editing Abstracts
2016 RESIDENTIAL AND LOW INCOME HOME ENERGY SERVICE PROGRAMS USER GUIDE FOR PROJECT SPONSORS
 XCEL NEW MEXICO 2016 RESIDENTIAL AND LOW INCOME HOME ENERGY SERVICE PROGRAMS USER GUIDE FOR PROJECT SPONSORS Updated January 2016 Table of Contents I. Introduction... 3 II. Registration... 4 Step 1: Login
XCEL NEW MEXICO 2016 RESIDENTIAL AND LOW INCOME HOME ENERGY SERVICE PROGRAMS USER GUIDE FOR PROJECT SPONSORS Updated January 2016 Table of Contents I. Introduction... 3 II. Registration... 4 Step 1: Login
Responding to RFQs/ITBs on the KNPC esourcing Portal and Sending Clarification Messages. Quick User Guide. Version 1.0
 Responding to RFQs/ITBs on the KNPC esourcing Portal and Sending Clarification Messages Quick User Guide Version 1.0 March 2016 Visit https://esourcing.knpc.com and login to the KNPC esourcing Portal.
Responding to RFQs/ITBs on the KNPC esourcing Portal and Sending Clarification Messages Quick User Guide Version 1.0 March 2016 Visit https://esourcing.knpc.com and login to the KNPC esourcing Portal.
Creating an MCRU Time and Events Table
 Creating an MCRU Time and Events Table This process builds an MCRU report from scratch. If you already have a billing calendar in MBECT, the steps that were completed as part of making the billing calendar
Creating an MCRU Time and Events Table This process builds an MCRU report from scratch. If you already have a billing calendar in MBECT, the steps that were completed as part of making the billing calendar
Creating a grant submission record in Themis
 Creating a grant submission record in Themis Creating a submission record for a grant requires completion of a number of screens. You will be able to monitor your progress on the progress train that displays
Creating a grant submission record in Themis Creating a submission record for a grant requires completion of a number of screens. You will be able to monitor your progress on the progress train that displays
SETTING UP YOUR SAFE KIDS DAY PAGE
 SETTING UP YOUR SAFE KIDS DAY PAGE As discussed in the website training webinar, you must set-up an individual page before you can join a team or become the captain of a team. The steps below walk you
SETTING UP YOUR SAFE KIDS DAY PAGE As discussed in the website training webinar, you must set-up an individual page before you can join a team or become the captain of a team. The steps below walk you
Project and Award Searches
 Project and Award Searches Contents Find Awards/Projects Search... 2 Award Page... 4 Project Inclusion... 4 Financials... 5 Summary View... 5 Calculator... 7 Projects View... 7 Period Summary... 8 AR...
Project and Award Searches Contents Find Awards/Projects Search... 2 Award Page... 4 Project Inclusion... 4 Financials... 5 Summary View... 5 Calculator... 7 Projects View... 7 Period Summary... 8 AR...
Industry Access Portal MUNICIPALITY MODULE User Guide
 2017 Industry Access Portal MUNICIPALITY MODULE User Guide LOUISIANA DEPARTMENT OF INSURANCE INDUSTRY ACCESS Contents Register for Industry Access... 2 Sign Up... 4 Log into Industry Access... 8 Access
2017 Industry Access Portal MUNICIPALITY MODULE User Guide LOUISIANA DEPARTMENT OF INSURANCE INDUSTRY ACCESS Contents Register for Industry Access... 2 Sign Up... 4 Log into Industry Access... 8 Access
Researchfish outcome reporting system. User Guide for Researchers
 Researchfish outcome reporting system User Guide for Researchers January 2017 How Researchfish works Researchfish is a system used by research funders to gather information on the outcomes of the research
Researchfish outcome reporting system User Guide for Researchers January 2017 How Researchfish works Researchfish is a system used by research funders to gather information on the outcomes of the research
ACP reporting. This guide provides a general overview on how to run ACP reports. In this guide: Available reports Page 3
 ACP reporting For ACP Supervisor ACP Reporting Administrator ACP reporting Last Updated March 2016 (Version 1.1) Menu My ACP run a report This guide provides a general overview on how to run ACP reports.
ACP reporting For ACP Supervisor ACP Reporting Administrator ACP reporting Last Updated March 2016 (Version 1.1) Menu My ACP run a report This guide provides a general overview on how to run ACP reports.
Policy on the Standardisation of Documentation
 Policy on the Standardisation of Documentation Policy Number Target Audience Approving Committee IMTD001 CCG Board members and staff CCG Executive Date Approved November 2013 Last Review Date July 2016
Policy on the Standardisation of Documentation Policy Number Target Audience Approving Committee IMTD001 CCG Board members and staff CCG Executive Date Approved November 2013 Last Review Date July 2016
Information Sheet for Scientific Approvers
 1 Information Sheet for Scientific Approvers This document will provide you with the basic instructions for your role as a scientific approver in OSIRIS. All listed approvers have been designated by their
1 Information Sheet for Scientific Approvers This document will provide you with the basic instructions for your role as a scientific approver in OSIRIS. All listed approvers have been designated by their
Reviewers Guide on Clinical Trials
 Reviewers Guide on Clinical Trials Office of Research Integrity & Compliance Version 2 Updated: June 26, 2017 This document is meant to help board members conduct reviews for Full Board: Clinical Trial
Reviewers Guide on Clinical Trials Office of Research Integrity & Compliance Version 2 Updated: June 26, 2017 This document is meant to help board members conduct reviews for Full Board: Clinical Trial
Project Modification Request 9.2 Financial System Campus Users Guide Explanation of Form & Header Sections
 Project Modification Request 9.2 Financial System Campus Users Guide Explanation of Form & Header Sections 6/2014 Revised 3/2018 Project Modification Request INTRODUCTION: A project modification request
Project Modification Request 9.2 Financial System Campus Users Guide Explanation of Form & Header Sections 6/2014 Revised 3/2018 Project Modification Request INTRODUCTION: A project modification request
Office of Human Research
 Office of Human Research JeffTrial OHR Training Document OHR Workflow and Data Entry Office of Human Research 8/16/2013 Ver. 1.0 Table of Contents OHR and JeffTrial... 2 JeffTrial Workflow... 2 Workflow
Office of Human Research JeffTrial OHR Training Document OHR Workflow and Data Entry Office of Human Research 8/16/2013 Ver. 1.0 Table of Contents OHR and JeffTrial... 2 JeffTrial Workflow... 2 Workflow
SOP Owner: Finance Last Reviewed/Update Date: 10/05/2015. Petty Cash Reimbursement on Grants Standard Operating Procedure
 Petty Cash Reimbursement on Grants Standard Operating Procedure 1. Purpose WCMC is required by federal regulation to monitor and review any research related costs associated to Grants. All transactions
Petty Cash Reimbursement on Grants Standard Operating Procedure 1. Purpose WCMC is required by federal regulation to monitor and review any research related costs associated to Grants. All transactions
Installing TeamViewer
 Installing TeamViewer 1 2008-05-03 Installing TeamViewer 1 Introduction TeamViewer is commercial software produced by TeamViewer GmbH that provides screen sharing, remote control, file transfer and a text
Installing TeamViewer 1 2008-05-03 Installing TeamViewer 1 Introduction TeamViewer is commercial software produced by TeamViewer GmbH that provides screen sharing, remote control, file transfer and a text
Construction Document: Petitions, Claims and Complaints Management Bizagi Process Modeler
 Construction Document: Petitions, Claims and Complaints Management Bizagi Process Modeler Copyright 2011 - bizagi Table of Contents 1. 2. Process Description... 4 Main Facts in the Process Construction...
Construction Document: Petitions, Claims and Complaints Management Bizagi Process Modeler Copyright 2011 - bizagi Table of Contents 1. 2. Process Description... 4 Main Facts in the Process Construction...
ELECTRONIC ADDITIONAL PAYMENT FORM Table of Contents
 ELECTRONIC ADDITIONAL PAYMENT FORM Table of Contents Electronic payment form options/overview.page 2-3 Individual Entry process..page 4-5 Group Entry Process Page 6-7 Excel Spreadsheet mass upload process..page
ELECTRONIC ADDITIONAL PAYMENT FORM Table of Contents Electronic payment form options/overview.page 2-3 Individual Entry process..page 4-5 Group Entry Process Page 6-7 Excel Spreadsheet mass upload process..page
PRS Desk Reference Guide. Bulk CREFC Template Desk Reference
 PRS Desk Reference Guide Bulk CREFC Template Desk Reference Freddie Mac Multifamily July 2017 Bulk CREFC Template 2 P a g e Table of Contents Bulk CREFC Template... 4 General Form Guidance... 4 Examples
PRS Desk Reference Guide Bulk CREFC Template Desk Reference Freddie Mac Multifamily July 2017 Bulk CREFC Template 2 P a g e Table of Contents Bulk CREFC Template... 4 General Form Guidance... 4 Examples
NHS e-referrals User Guide - SystmOne TPP (C&B) Please Use This Guide for Practices in: North West London CCG s
 NHS e-referrals User Guide SystmOne TPP Please Use This Guide for Practices in: North West London CCG s Version: 3.4 Author Filename: Path: Primary Care Systems Team / CWHHECCG System One Implementation
NHS e-referrals User Guide SystmOne TPP Please Use This Guide for Practices in: North West London CCG s Version: 3.4 Author Filename: Path: Primary Care Systems Team / CWHHECCG System One Implementation
NHS Education for Scotland Portal https://www.portal.scot.nhs.uk Dental Audit: A user guide from application to completion
 Dental Audit: A user guide from application to completion 1. Audit Guidance 2. New Application: Getting Started 3. New Application: The Audit Application Form 4. New Application: Submitting Your Application
Dental Audit: A user guide from application to completion 1. Audit Guidance 2. New Application: Getting Started 3. New Application: The Audit Application Form 4. New Application: Submitting Your Application
Mentor eirb Researcher User Manual
 Ascension Wisconsin IRB Mentor eirb Researcher User Manual Contents 1 Introduction 1.1 Purpose of this Manual 1.2 User Support 2 Mentor eirb & User Accounts 2.1 About Mentor 2.2 User Roles 2.3 Requesting
Ascension Wisconsin IRB Mentor eirb Researcher User Manual Contents 1 Introduction 1.1 Purpose of this Manual 1.2 User Support 2 Mentor eirb & User Accounts 2.1 About Mentor 2.2 User Roles 2.3 Requesting
MAFSI BUSINESS BAROMETER REPORTING TOOL HOW-TO GUIDE
 MAFSI BUSINESS BAROMETER REPORTING TOOL HOW-TO GUIDE Thank you for taking the time to participate in the MAFSI Business Barometer Surveys. The following guide is intended to make using the Barometer Calculator
MAFSI BUSINESS BAROMETER REPORTING TOOL HOW-TO GUIDE Thank you for taking the time to participate in the MAFSI Business Barometer Surveys. The following guide is intended to make using the Barometer Calculator
AmpliFund Applicant Portal
 AmpliFund Applicant Portal Contents Applicant Portal Overview... 2 Registering on the Applicant Portal... 2 Logging In To the Applicant Portal... 2 User Menu... 2 Account Information... 2 Change Password...
AmpliFund Applicant Portal Contents Applicant Portal Overview... 2 Registering on the Applicant Portal... 2 Logging In To the Applicant Portal... 2 User Menu... 2 Account Information... 2 Change Password...
Membership Database Application
 Membership Database Application This application tracks member payments for dues, programs, and events; generates form letters for past due notices, endof-year summaries; provides the capability for users
Membership Database Application This application tracks member payments for dues, programs, and events; generates form letters for past due notices, endof-year summaries; provides the capability for users
ES FINANCIALS User Training
 ES FINANCIALS User Training MAKING JOURNAL ENTRIES PARTICIPANT GUIDE Date: July 2016 Page 1 of 36 Making Journal Entries Exercise 1: Creating an Online Journal including Email Notification & Attachments...
ES FINANCIALS User Training MAKING JOURNAL ENTRIES PARTICIPANT GUIDE Date: July 2016 Page 1 of 36 Making Journal Entries Exercise 1: Creating an Online Journal including Email Notification & Attachments...
Rev 2 May Health and Safety Authority. Function and Scope of REACH and CLP Helpdesk
 Rev 2 May 2011 Health and Safety Authority Function and Scope of REACH and CLP Helpdesk Introduction The EU Regulation for Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) entered
Rev 2 May 2011 Health and Safety Authority Function and Scope of REACH and CLP Helpdesk Introduction The EU Regulation for Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) entered
AIR QUALITY MANAGEMENT DATABASE: ANNEX OF UBA REPORT (05/2010) "NOTIFICATIONS FOR TIME EXTENSION"
 AIR QUALITY MANAGEMENT DATABASE: ANNEX OF UBA REPORT (05/2010) "NOTIFICATIONS FOR TIME EXTENSION" Umweltbundesamt Error! No text of specified style in document. 1 Analysis of notifications Error! Style
AIR QUALITY MANAGEMENT DATABASE: ANNEX OF UBA REPORT (05/2010) "NOTIFICATIONS FOR TIME EXTENSION" Umweltbundesamt Error! No text of specified style in document. 1 Analysis of notifications Error! Style
PERFORMANCE IN INITIATING AND DELIVERING CLINICAL RESEARCH CTP PLATFORM INSTRUCTIONS
 PERFORMANCE IN INITIATING AND DELIVERING CLINICAL RESEARCH CTP PLATFORM INSTRUCTIONS Version Control Entire Document Quick Tips Quick Tips, Item 27.a.xix and xxx Item 27.a.xxv Item 27.a.xvi Item 27.a.xxvi
PERFORMANCE IN INITIATING AND DELIVERING CLINICAL RESEARCH CTP PLATFORM INSTRUCTIONS Version Control Entire Document Quick Tips Quick Tips, Item 27.a.xix and xxx Item 27.a.xxv Item 27.a.xvi Item 27.a.xxvi
edofe Management Toolkit
 edofe Management Toolkit A guide to effective edofe management for Directly Licensed Centres 1 2 Contents Section one: Setting up the correct infrastructure on edofe... 4 Creating a group... 4 Editing
edofe Management Toolkit A guide to effective edofe management for Directly Licensed Centres 1 2 Contents Section one: Setting up the correct infrastructure on edofe... 4 Creating a group... 4 Editing
Patient Services - Appointments User Guide
 Vision 3 Patient Services - Appointments User Guide Copyright INPS Ltd 2017 The Bread Factory, 1A Broughton Street, Battersea, London, SW8 3QJ T: +44 (0) 207 5017000 F:+44 (0) 207 5017100 W: www.inps.co.uk
Vision 3 Patient Services - Appointments User Guide Copyright INPS Ltd 2017 The Bread Factory, 1A Broughton Street, Battersea, London, SW8 3QJ T: +44 (0) 207 5017000 F:+44 (0) 207 5017100 W: www.inps.co.uk
Creating and using Moodle Rubrics
 Creating and using Moodle Rubrics Rubrics are marking methods that allow staff to ensure that consistent grading practices are followed, especially when grading as a team. They comprise a set of criteria
Creating and using Moodle Rubrics Rubrics are marking methods that allow staff to ensure that consistent grading practices are followed, especially when grading as a team. They comprise a set of criteria
EPA Research Programme EPA s Online Grant Application & Project Management Portal
 EPA Research Programme 2014-2020 EPA s Online Grant Application & Project Management Portal QUICK GUIDE TO MAKING AN APPLICATION April 2018 Version 3 Powered by SmartSimple The EPA Research Programme is
EPA Research Programme 2014-2020 EPA s Online Grant Application & Project Management Portal QUICK GUIDE TO MAKING AN APPLICATION April 2018 Version 3 Powered by SmartSimple The EPA Research Programme is
Ethics and Omics. Jeffrey Engler, Ph.D. Dept of Biochemistry and Molecular Genetics Associate Dean, UAB Graduate School
 Ethics and Omics Jeffrey Engler, Ph.D. Dept of Biochemistry and Molecular Genetics Associate Dean, UAB Graduate School Office of Research Integrity/DHHS http://ori.hhs.gov/education/products /clinicaltools/data.pdf
Ethics and Omics Jeffrey Engler, Ph.D. Dept of Biochemistry and Molecular Genetics Associate Dean, UAB Graduate School Office of Research Integrity/DHHS http://ori.hhs.gov/education/products /clinicaltools/data.pdf
Office of Human Research
 Office of Human Research JeffTrial End-User Training Document Regulatory Coordinator Training for Non-Oncology personnel Office of Human Research 8/16/2013 Ver. 1.0 Contents The REG Role: Completing Basic
Office of Human Research JeffTrial End-User Training Document Regulatory Coordinator Training for Non-Oncology personnel Office of Human Research 8/16/2013 Ver. 1.0 Contents The REG Role: Completing Basic
REPORTING Copyright Framework Private Equity Investment Data Management Ltd
 REPORTING Copyright Framework Private Equity Investment Data Management Ltd - 2016 Table of Contents Standard Reports... 3 Standard Report Pack... 4 General Data Protection and Framework... 7 Partner Bank
REPORTING Copyright Framework Private Equity Investment Data Management Ltd - 2016 Table of Contents Standard Reports... 3 Standard Report Pack... 4 General Data Protection and Framework... 7 Partner Bank
Table of Contents. SV3 Visitor Registration Tenant Job Aid. 1. Introduction Accessing the SV3 Portal Schedule Visitor...
 Table of Contents 1. Introduction... 2 2. Accessing the SV3 Portal... 2 3. Schedule Visitor... 3 1. Introduction This Job Aid is for entering registered visitors into SV3 using the new user experience
Table of Contents 1. Introduction... 2 2. Accessing the SV3 Portal... 2 3. Schedule Visitor... 3 1. Introduction This Job Aid is for entering registered visitors into SV3 using the new user experience
Audience Analytics Data Submission Guide Current Participants
 Audience Analytics Data Submission Guide Current Participants This material will guide you through the process of reviewing your current data in emerge and organizing, preparing, and uploading your new
Audience Analytics Data Submission Guide Current Participants This material will guide you through the process of reviewing your current data in emerge and organizing, preparing, and uploading your new
COC APR Finding and Fixing Data Quality Errors
 COC APR Finding and Fixing Data Quality Errors The new COC APR is now available (April 1, 2017). The new report: Has been formatted into a canned report found on the Reports Tab Utilized for projects with
COC APR Finding and Fixing Data Quality Errors The new COC APR is now available (April 1, 2017). The new report: Has been formatted into a canned report found on the Reports Tab Utilized for projects with
CMS RETROACTIVE ENROLLMENT & PAYMENT VALIDATION RETROACTIVE PROCESSING CONTRACTOR (RPC)
 CMS RETROACTIVE ENROLLMENT & PAYMENT VALIDATION RETROACTIVE PROCESSING CONTRACTOR (RPC) STATE & ENROLLMENT BROKER RETROACTIVE SUBMISSION STANDARD OPERATING PROCEDURE (FOR STATES PARTICIPATING IN THE FINANCIAL
CMS RETROACTIVE ENROLLMENT & PAYMENT VALIDATION RETROACTIVE PROCESSING CONTRACTOR (RPC) STATE & ENROLLMENT BROKER RETROACTIVE SUBMISSION STANDARD OPERATING PROCEDURE (FOR STATES PARTICIPATING IN THE FINANCIAL
Workforce Repository and Planning Tool Creating a Standard Baseline report
 Workforce Repository and Planning Tool Creating a Standard Baseline report 1 CONTENTS Section Detail Page 1 Introduction 2 Scoping 3 Information Governance 4 Data Collation 5 Populating the field creator
Workforce Repository and Planning Tool Creating a Standard Baseline report 1 CONTENTS Section Detail Page 1 Introduction 2 Scoping 3 Information Governance 4 Data Collation 5 Populating the field creator
SchARP Instruction Manual
 SchARP Instruction Manual Secure Payroll Upload & Loan Dashboard ALLIANCE BENEFIT GROUP OF ILLINOIS ADMINISTRATORS, CONSULTANTS, ACTUARIES 456 FULTON STREET, STE 345, PEORIA, IL 61602 www.abgil.com Welcome
SchARP Instruction Manual Secure Payroll Upload & Loan Dashboard ALLIANCE BENEFIT GROUP OF ILLINOIS ADMINISTRATORS, CONSULTANTS, ACTUARIES 456 FULTON STREET, STE 345, PEORIA, IL 61602 www.abgil.com Welcome
INAB CAB Portal User Guide
 INAB CAB Portal User Guide CRM 2 INAB Cab Portal User Guide CRM Documentation Issue 2 June 2018 Contents 1. Login...4 1.1. Set up Portal password... 5 1.2. Login... 6 1.3. Forgot my password... 7 1.4.
INAB CAB Portal User Guide CRM 2 INAB Cab Portal User Guide CRM Documentation Issue 2 June 2018 Contents 1. Login...4 1.1. Set up Portal password... 5 1.2. Login... 6 1.3. Forgot my password... 7 1.4.
Statutory Notifications
 Registration under the Health and Social Care Act 2008 Statutory Notifications Guidance for registered providers and managers of NHS GP and other primary medical services May 2013 Statutory notifications
Registration under the Health and Social Care Act 2008 Statutory Notifications Guidance for registered providers and managers of NHS GP and other primary medical services May 2013 Statutory notifications
The results section of a clinicaltrials.gov file is divided into discrete parts, each of which includes nested series of data entry screens.
 OVERVIEW The ClinicalTrials.gov Protocol Registration System (PRS) is a web-based tool developed for submitting clinical trials information to ClinicalTrials.gov. This document provides step-by-step instructions
OVERVIEW The ClinicalTrials.gov Protocol Registration System (PRS) is a web-based tool developed for submitting clinical trials information to ClinicalTrials.gov. This document provides step-by-step instructions
/Internet Random Moment Sampling. STATE OF ALASKA Department of Health and Social Services Division of Public Assistance
 E-mail/Internet Random Moment Sampling STATE OF ALASKA Department of Health and Social Services Division of Public Assistance RMS Training Objectives Goal: Upon completion of this training session, participants
E-mail/Internet Random Moment Sampling STATE OF ALASKA Department of Health and Social Services Division of Public Assistance RMS Training Objectives Goal: Upon completion of this training session, participants
Lorin Muhlmann V0.5 Last updated 17/04/18
 ClubGRANTS Online Training Manual CONVENORS Contents Logging in... 4 Opening a grant round... 6 Completing an application... 10 Reviewing and assessing applications... 15 Completing and reviewing acquittal
ClubGRANTS Online Training Manual CONVENORS Contents Logging in... 4 Opening a grant round... 6 Completing an application... 10 Reviewing and assessing applications... 15 Completing and reviewing acquittal
ClubGRANTS Online Training Manual
 1 ClubGRANTS Online Training Manual Contents Registration... 3 Opening a grant round... 5 Reviewing and Editing Aquittal Forms... 15 Exporting data to excel... 17 Sending customised correspondence to applicants...
1 ClubGRANTS Online Training Manual Contents Registration... 3 Opening a grant round... 5 Reviewing and Editing Aquittal Forms... 15 Exporting data to excel... 17 Sending customised correspondence to applicants...
AER Returns Excel Workbook & Website User Manual
 AER Returns Excel Workbook & Website User Manual Table of Contents Version 2.1 January 2015 1.0 Downloading the Configured Excel Workbook or XSD Schema... 2 1.1 Logging In... 2 1.2 Downloading the Configured
AER Returns Excel Workbook & Website User Manual Table of Contents Version 2.1 January 2015 1.0 Downloading the Configured Excel Workbook or XSD Schema... 2 1.1 Logging In... 2 1.2 Downloading the Configured
Guidance for Completion of Form R Parts A & B
 1 Guidance for Completion of Form R Parts A & B 2 Table of Contents The following document details each field of the Form R giving guidance, examples and required actions. Please read through this document
1 Guidance for Completion of Form R Parts A & B 2 Table of Contents The following document details each field of the Form R giving guidance, examples and required actions. Please read through this document
pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation (Manufacturer)
 pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation (Manufacturer) Blinatumomab (Blincyto) for Philadelphia chromosome positive Acute Lymphoblastic
pan-canadian Oncology Drug Review Stakeholder Feedback on a pcodr Expert Review Committee Initial Recommendation (Manufacturer) Blinatumomab (Blincyto) for Philadelphia chromosome positive Acute Lymphoblastic
Using DCC DMPonline to write a Data Management Plan
 Using DCC DMPonline to write a Data Management Plan Laurence Horton, London School of Economics and Political Science datalibrary@lse.ac.uk Version 1.1 October 2017 Table of Contents Introduction... 2
Using DCC DMPonline to write a Data Management Plan Laurence Horton, London School of Economics and Political Science datalibrary@lse.ac.uk Version 1.1 October 2017 Table of Contents Introduction... 2
Updated Feature: Appointment Center User Interface
 Updated Feature: Appointment Center User Interface We have redesigned the user interface and interactions in Appointment Center as part of this release. There are new actions and abilities to manage locations
Updated Feature: Appointment Center User Interface We have redesigned the user interface and interactions in Appointment Center as part of this release. There are new actions and abilities to manage locations
QUALTRICS SURVEY TOOL (BlueQ 2015)
 QUALTRICS SURVEY TOOL (BlueQ 2015) DoIT Training Course Procedures Handout Table of Contents Logging In... 4 To Create a New Survey... 4 Create a New Item... 5 Adding Text... 5 Create a New Item Options...
QUALTRICS SURVEY TOOL (BlueQ 2015) DoIT Training Course Procedures Handout Table of Contents Logging In... 4 To Create a New Survey... 4 Create a New Item... 5 Adding Text... 5 Create a New Item Options...
ILM Recognition Endorsed and Development programme application form Part Two
 ILM Recognition Endorsed and Development programme application form Part Two Version 0317 01 tes for completion Where you see this paperclip icon: Please attach the relevant document to your application,
ILM Recognition Endorsed and Development programme application form Part Two Version 0317 01 tes for completion Where you see this paperclip icon: Please attach the relevant document to your application,
Report on the activities of the Independent Integrity Unit, November 2016 to September 2017
 Meeting of the Board 30 September 2 October 2017 Cairo, Arab Republic of Egypt Provisional agenda item 8 GCF/B.18/Inf.12 29 September 2017 Report on the activities of the Independent Integrity Unit, November
Meeting of the Board 30 September 2 October 2017 Cairo, Arab Republic of Egypt Provisional agenda item 8 GCF/B.18/Inf.12 29 September 2017 Report on the activities of the Independent Integrity Unit, November
