University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Overview for PIs in Nursing
|
|
|
- Hilary McCarthy
- 5 years ago
- Views:
Transcription
1 University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Overview for PIs in Nursing WISPER Overview for Principal Investigators in Nursing WISPER facilitates the process of routing applications and agreements for extramural support by capturing the necessary data up and tracking through the generation of the award. As the Principal Investigator, you may be the initiator of record creation process. This document will walk through this process. Please feel free to leave fields empty and enter only known data, unless explicitly told to provide information within the following steps. This desk reference should be utilized based upon the assumption that this is the first record created of its kind by the PI. If previously created records are available to copy, you may refer to the Copy a Record desk reference for information on how to quickly create a new record based on a previous record. CREATE NEW RECORD...2 Login...2 Enter all Data on the Create New Record page...2 GENERAL PAGE INFORMATION...3 Basic Information:...3 Submission Instructions...4 Compliance...4 Personnel...5 Proposed Budget...5 Keywords/Comments...5 ATTACHMENTS...6 Upload Attachments...6 PRINCIPAL INVESTIGATOR SIGNATURE...7 Electronically Sign for the Proposal...7 Electronically Sign for the Award...8 ADD APPROVAL...9 Approval...9 ROUTE THE RECORD...10 Routing:...10 TRACK THE RECORD...11 Tracking the Status of 11
2 CREATE NEW RECORD 1. Login: 1 Navigate to the WISPER login page via to enter your PantherID and password. You will be directed to your MY WISPER page. 2 Select the Create New Record button at the top of the page. 2. Enter all Data on the Create New Record page: 1 Enter the following key data to create the initial record: a. Short Title working title for internal communication purposes b. Contact PI your name should already appear c. Department - Enter in Dept ID field d. Sponsor Search for and select Sponsor name from list 2 Click Create Record to advance to enter more information Navigation: UW WISPER > My WISPER > Create New Record button 2 of 11
3 GENERAL PAGE INFORMATION Note that you now have the WISPER ID number available to you in the header. This number can be used to track this record. The General page captures the bulk of the information about your record. 3. Basic Information: 1 Enter as much of the following information as possible: a. Official proposal title b. Start and End dates c. Proposal type d. Document type (usually Grant) 2 If the current record is related to an already existing WISPER record, click the Add Related Document button for the option of linking this record to an existing WISPER record such as a record for which the current record is a supplement or continuation, a CDA or MTA preceding the current record, or any other relationship you can think of. 3 The contact PI, department, and Sponsor also may be changed in this section. *This record can always be found in your My WISPER page under Records for which I am the PI. 3 of 11
4 4. Submission Instructions: This section captures detailed sponsor submission instructions and the campus contact information. 1 Enter the necessary data for the following fields: a. Sponsor deadline b. Submission method c. UWM campus contact - Search for and select the name of anyone assisting you in the development of this proposal or other WISPER document 2 If known, note the sponsor program number and reference number along with any detailed submission instructions. NOTE: If the Sponsor is UWM Foundation, enter UWM Foundation Account Number (####.##) in the Sponsor Reference Number field. 5. Compliance: All compliance questions must be answered before the PI will have the ability to sign the record. Answer each question by selecting Yes or No. 4 of 11
5 6. Personnel: The personnel section allows the listing of additional employees working on the project as well as role designation such as Co-Investigator, Fellow, and other project roles. If the record has multiple PIs, all PIs must be entered at this time. Entering other personnel is optional. 1 Click the Add Other Personnel to Record button which will open a new page allowing an employee search by name or Dept ID (UDDS). 2 After employee selection, identify their project role from the dropdown menu. 3 Repeat this process for as many personnel as you want to enter. 4 To delete personnel from the record click the trashcan to the right of the employee name. 7. Proposed Budget: If known, enter the proposed budget, F&A rate, and cost sharing information 8. Keywords/Comments: 1 If this proposal is related to the Return on Investment for an RGI award, enter RGI as a keyword 2 In addition, keywords assist in tracking research in major scientific areas. Enter relevant keywords separated by a comma or semi-colon. 3 Click the Add Comments button to access a large text field, which can be used to enter any information wished to be communicated to others accessing the record. *This record can always be found in your My WISPER page under Records for which I am the PI. 5 of 11
6 ATTACHMENTS Any type of file attachment can be uploaded via this page including PureEdge files, budget spreadsheets for review, and the complete application. 9. Upload Attachments: 1 Navigate to the attachment tab 2 Click the Upload Attachment button. 3 Browse for and upload the documents. (This process may take a couple of minutes depending on file size). 4 After the upload is complete, enter a file description such as draft narrative or proposal abstract, or budget justification. 5 To add multiple attachments click the Add another attachment hyperlink. 6 To delete an attachment, click on the trash can next to the Upload Attachment button. 6 of 11
7 PRINCIPAL INVESTIGATOR SIGNATURE Principal Investigators will be required to verify the information provided in the record prior to routing the document to RSA. This signature may be provided at any time following the initial data entry as described above. In order for the PI(s) to electronically sign the record, all compliance questions must be answered. 10. Electronically Sign for the Proposal: 1. Click on the Sign the Record hyperlink at the bottom of the General page. This will open the PI Signature page. 2. Read the statement to which you are attesting. 3. Sign for the proposal by clicking the I Sign button. 4. Verify that the PI name along with the date and time is stamped. If physical signatures are required on a record due to the PI(s) not being in the appointment tables, the PRINT button should be selected on the General page for a paper copy of the record 7 of 11
8 11. Electronically Sign for the Award: Below the Signature for Proposal is the Principal Investigator Signature for Award. If award terms and conditions are available at this time, PIs have the option to read and sign the award terms and conditions. In order for the PI to sign the Award signature, the Proposal signature must have previously been signed. After signing each statement, the PI name along with signature date and time will be stamped on the page as well as on the History page. The PI Signature page is accessible only to Principal Investigators; others will access the Document History page to verify that the signatures have been executed. 8 of 11
9 ADD AN APPROVAL 12. Approval: One way to share a record with another person is to add an approval. 1 Navigate to the Approvals Tab 2 Click the Add Approval button. This will open the details page. 3 Use the Approval Type Drop Down to select an appropriate approval type or other. 4 Click the Select Employee Button 5 Search for and select the name of the person with whom the record should be shared. 6 Click the Add Approval button. The main Approvals page will re-open with the newly requested approval documented with date and time. 9 of 11
10 ROUTE THE RECORD 13. Routing: After completion of the PI signature process, you will need to route the record to your Dean s office who will approve the record and forward to RSA for submission to sponsor or other processing. 1 Click the Route to Another Person button at the bottom of the PI Signature page. 2 Select the bottom button. 3 Note that the top button Route to a specific person will hardly ever be used at UWM. After routing, the record will be sent to your Dean s office and you can select the Sign Out link on the top right of the page or select the MY WISPER worklist link on the bottom of the page to continue further actions. 10 of 11
11 TRACK THE RECORD 14. Tracking the Status: You can always track the status of the record using WISPER to see who is working on it and its status. 1 Navigate to MY WORKLIST which is your home page when you log into WISPER 2 Scroll to the bottom of the page & see the Records for Which I am a PI Additional information and help is available at: or WISPER-rsa@uwm.edu or call of 11
University of Wisconsin Grants Project Desk Reference Madison * Milwaukee * Extension
 University of Wisconsin Grants Project Desk Reference Madison * Milwaukee * Extension WISPER Record Creation Overview WISPER facilitates the process of routing applications and agreements for extramural
University of Wisconsin Grants Project Desk Reference Madison * Milwaukee * Extension WISPER Record Creation Overview WISPER facilitates the process of routing applications and agreements for extramural
University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Update a Pre-Proposal for Full Submission
 University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Update a Pre-Proposal for Full Submission In order to obtain external funding for their research programs, sometimes investigators
University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Update a Pre-Proposal for Full Submission In order to obtain external funding for their research programs, sometimes investigators
University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Record Creation for Principal Investigators
 University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Record Creation for Principal Investigators WISPER - Record Creation for Principal Investigators Once a Pre-Proposal has been submitted
University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Record Creation for Principal Investigators WISPER - Record Creation for Principal Investigators Once a Pre-Proposal has been submitted
University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Supplement for Letters & Science Administrators
 University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Supplement for Letters & Science Administrators WISPER Supplement for Letters & Science Administrators WISPER facilitates the process
University of Wisconsin - Milwaukee Grants Project Desk Reference WISPER Supplement for Letters & Science Administrators WISPER Supplement for Letters & Science Administrators WISPER facilitates the process
NCURA 2011 Region IV Spring Meeting. Introduction to Electronic Document Routing
 NCURA 2011 Region IV Spring Meeting Introduction to Electronic Document Routing Melanie Jacobs Senior Grants & Contracts Specialist mrjacobs3@rsp.wisc.edu Nick Novak Senior Grants & Contracts Specialist
NCURA 2011 Region IV Spring Meeting Introduction to Electronic Document Routing Melanie Jacobs Senior Grants & Contracts Specialist mrjacobs3@rsp.wisc.edu Nick Novak Senior Grants & Contracts Specialist
Grants Project Reference Manual WISPER
 Grants Project Reference Manual Grants Project Reference Manual Revised 11/18/2009 Table of Contents BUSINESS PROCESS OVERVIEW... 1 Assumptions... 1 Key Impacts... 2 Related Forms and Reports... 2 Process
Grants Project Reference Manual Grants Project Reference Manual Revised 11/18/2009 Table of Contents BUSINESS PROCESS OVERVIEW... 1 Assumptions... 1 Key Impacts... 2 Related Forms and Reports... 2 Process
University of Wisconsin-Madison Grants Project WISPER Manual
 University of Wisconsin-Madison I. BUSINESS PROCESS OVERVIEW... 2 A. ASSUMPTIONS... 2 B. PROCESS OUTLIERS/SUB-PROCESSES... 3 C. ROLES AND RESPONSIBILITIES... 3 D. WISPER USER ROLES... 3 E. WISPER HIGH
University of Wisconsin-Madison I. BUSINESS PROCESS OVERVIEW... 2 A. ASSUMPTIONS... 2 B. PROCESS OUTLIERS/SUB-PROCESSES... 3 C. ROLES AND RESPONSIBILITIES... 3 D. WISPER USER ROLES... 3 E. WISPER HIGH
JDRF Operations External FAQs Preaward and Postaward
 JDRF Operations External FAQs Preaward and Postaward Page 1 RMS360 Preaward FAQ Application Content 1. How do I apply for a grant? 2. How do I add my RO (Research Office contact) and FO (Finance Officer)
JDRF Operations External FAQs Preaward and Postaward Page 1 RMS360 Preaward FAQ Application Content 1. How do I apply for a grant? 2. How do I add my RO (Research Office contact) and FO (Finance Officer)
The most current protocol information should be entered. This includes incorporation of all past amendments and modifications.
 Georgetown University Institutional Review Board Frequently Asked Questions (FAQs) about eric 1. What is eric? eric stands for Electronic Research and Institutional Compliance. This is an on-line IRB application
Georgetown University Institutional Review Board Frequently Asked Questions (FAQs) about eric 1. What is eric? eric stands for Electronic Research and Institutional Compliance. This is an on-line IRB application
OMNI GRANTS PROPOSAL APPROVAL WORKFLOW
 OMNI GRANTS PROPOSAL APPROVAL WORKFLOW OVERVIEW Training/Reference Material Proposal Development Guide creating& submitting a proposal Job Aids short instructions for accomplishing a specific task Overview
OMNI GRANTS PROPOSAL APPROVAL WORKFLOW OVERVIEW Training/Reference Material Proposal Development Guide creating& submitting a proposal Job Aids short instructions for accomplishing a specific task Overview
NAU New Project Submission. Guidance for creating a new project
 NAU New Project Submission Guidance for creating a new project Go to IRBNet Website address: https://www.irbnet.org/release/index.html Login to your account Enter your Username and Password and select
NAU New Project Submission Guidance for creating a new project Go to IRBNet Website address: https://www.irbnet.org/release/index.html Login to your account Enter your Username and Password and select
ELECTRONIC BIOMEDICAL RESEARCH APPLICATION PORTAL (ebrap) User Guide
 ELECTRONIC BIOMEDICAL RESEARCH APPLICATION PORTAL (ebrap) User Guide 1 Table of Contents Overview 5 Purpose and User Roles 5 Help Desk Contact Information 6 Operating Environment 6 Welcome to ebrap 7 Register
ELECTRONIC BIOMEDICAL RESEARCH APPLICATION PORTAL (ebrap) User Guide 1 Table of Contents Overview 5 Purpose and User Roles 5 Help Desk Contact Information 6 Operating Environment 6 Welcome to ebrap 7 Register
ORSP Online Application User Guide
 ORSP Online Application User Guide For submission of the following Internal Awards: Adjunct Faculty Opportunity Fund Provost Faculty Opportunities Fund Career Development Committee Funds (CDC) PI Travel
ORSP Online Application User Guide For submission of the following Internal Awards: Adjunct Faculty Opportunity Fund Provost Faculty Opportunities Fund Career Development Committee Funds (CDC) PI Travel
PI Certification Quick Guide
 PI Certification Quick Guide 1 P age Table of Contents Certifications... 3 PI Self-Certification... 3 PI is Certifying via Link in Email Notification... 3 PI Goes Directly to the Proposal... 6 Certification
PI Certification Quick Guide 1 P age Table of Contents Certifications... 3 PI Self-Certification... 3 PI is Certifying via Link in Email Notification... 3 PI Goes Directly to the Proposal... 6 Certification
Researcher User Manual
 Researcher User Manual Cloning Applications, Creating and Managing Events Audience: Principal Investigators & Project Team Members Updated: November 24, 2017 Checkpoint *PLEASE NOTE* Prior to leveraging
Researcher User Manual Cloning Applications, Creating and Managing Events Audience: Principal Investigators & Project Team Members Updated: November 24, 2017 Checkpoint *PLEASE NOTE* Prior to leveraging
Reference Card for ORSP
 Reference Card for ORSP https://eresearch.umich.edu Log in from eresearch Homepage 1. Go to https://eresearch.umich.edu. 2. Click Login in the Proposal Management box. 3. Enter your Login ID (uniqname)
Reference Card for ORSP https://eresearch.umich.edu Log in from eresearch Homepage 1. Go to https://eresearch.umich.edu. 2. Click Login in the Proposal Management box. 3. Enter your Login ID (uniqname)
ELECTRONIC BIOMEDICAL RESEARCH APPLICATION PORTAL (ebrap) User Guide
 ELECTRONIC BIOMEDICAL RESEARCH APPLICATION PORTAL (ebrap) User Guide 1 Table of Contents Overview 5 Purpose and User Roles 5 Help Desk Contact Information 6 Operating Environment 6 Welcome to the ebrap
ELECTRONIC BIOMEDICAL RESEARCH APPLICATION PORTAL (ebrap) User Guide 1 Table of Contents Overview 5 Purpose and User Roles 5 Help Desk Contact Information 6 Operating Environment 6 Welcome to the ebrap
erequest How to apply guide
 Overview is an application that assists UCB in request life cycle management. UCB has clear guidance in place on what they can support or sponsor. Online requests will go through an internal review and
Overview is an application that assists UCB in request life cycle management. UCB has clear guidance in place on what they can support or sponsor. Online requests will go through an internal review and
IRBNet User Manual. University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB)
 University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB) IRBNet User Manual Office of Research Integrity and Education Mary Reed Building 222 INTRODUCTION The Office
University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB) IRBNet User Manual Office of Research Integrity and Education Mary Reed Building 222 INTRODUCTION The Office
Analyst User Guide for PAMIS ecaf
 Analyst User Guide for PAMIS ecaf 05/29/2009 PAMIS ecaf Analyst User Guide Slide 1 Table of Contents Introduction First Use of ecaf Creating an ecaf for a New Proposal Creating an ecaf for a Renewal, Continuation,
Analyst User Guide for PAMIS ecaf 05/29/2009 PAMIS ecaf Analyst User Guide Slide 1 Table of Contents Introduction First Use of ecaf Creating an ecaf for a New Proposal Creating an ecaf for a Renewal, Continuation,
Revision Control Date By Action Pages 12/3/2018 Erica Escamilla Created 12
 Business Process Guide Process: (Staff and Management) Module: Recruiting Solutions High Level Description Process Module Document Type How to Search and Apply for a Job at Cal State East Bay Recruiting
Business Process Guide Process: (Staff and Management) Module: Recruiting Solutions High Level Description Process Module Document Type How to Search and Apply for a Job at Cal State East Bay Recruiting
Office for Sponsored Research. Electronic Sponsored Project Requests (ESPR) System User Guide
 Office for Sponsored Research Electronic Sponsored Project Requests (ESPR) System User Guide Updated May 1, 2015 Contents 1.0 Introduction to ESPR... 3 1.1 Overview... 3 1.2 Accessing ESPR... 3 1.3 ESPR
Office for Sponsored Research Electronic Sponsored Project Requests (ESPR) System User Guide Updated May 1, 2015 Contents 1.0 Introduction to ESPR... 3 1.1 Overview... 3 1.2 Accessing ESPR... 3 1.3 ESPR
Quick guide to the SmartSimple on-line portal (making an application)
 EPA Research Programme 2014-2020 Quick guide to the SmartSimple on-line portal (making an application) POWERED BY SMARTSIMPLE Disclaimer Please read this document carefully prior to using the on-line portal.
EPA Research Programme 2014-2020 Quick guide to the SmartSimple on-line portal (making an application) POWERED BY SMARTSIMPLE Disclaimer Please read this document carefully prior to using the on-line portal.
Getting Help. Intra/Inter Departmental Transfer. IDT Journal Approvers Quick Guide. Help with IDT Journals: Michelle Duong
 Intra/Inter Departmental Transfer An Intra/Inter Departmental Transfer (IDT) is a PeopleSoft journal enabling an authorized user to transfer revenue or expenses from one cost center to another. This process
Intra/Inter Departmental Transfer An Intra/Inter Departmental Transfer (IDT) is a PeopleSoft journal enabling an authorized user to transfer revenue or expenses from one cost center to another. This process
User s Guide: Sponsored Programs PreAward Module
 User s Guide: Sponsored Programs PreAward Module Adding a New Proposal NUgrant is a web portal through which you can route proposals, file disclosures of interest, and submit IRB or IACUC protocols. This
User s Guide: Sponsored Programs PreAward Module Adding a New Proposal NUgrant is a web portal through which you can route proposals, file disclosures of interest, and submit IRB or IACUC protocols. This
Project Modification Request 9.2 Financial System Campus Users Guide Explanation of Form & Header Sections
 Project Modification Request 9.2 Financial System Campus Users Guide Explanation of Form & Header Sections 6/2014 Revised 3/2018 Project Modification Request INTRODUCTION: A project modification request
Project Modification Request 9.2 Financial System Campus Users Guide Explanation of Form & Header Sections 6/2014 Revised 3/2018 Project Modification Request INTRODUCTION: A project modification request
Submitting a Response to Modifications Required by the IRB to Secure Approval
 Submitting a Response to Modifications Required by the IRB to Secure Approval If you have submitted an item to the IRB, and the IRB has responded with a Modifications Required to Secure Approval memorandum,
Submitting a Response to Modifications Required by the IRB to Secure Approval If you have submitted an item to the IRB, and the IRB has responded with a Modifications Required to Secure Approval memorandum,
Chartfield Request C&G Contracts & Grants Roles 9.1 Financial System
 Chartfield Request C&G Contracts & Grants Roles 9.1 Financial System 1 Chartfield Request Processing Under specific criterion, segments are established to separately account for expenditures related to
Chartfield Request C&G Contracts & Grants Roles 9.1 Financial System 1 Chartfield Request Processing Under specific criterion, segments are established to separately account for expenditures related to
Weill Research Gateway
 Table of Contents The - What is the? - Logging In to WRG - The WRG Homepage Conflicts of Interest - What are Conflicts of Interest? - Submitting your Conflicts Survey - Submitting a Travel Disclosure -
Table of Contents The - What is the? - Logging In to WRG - The WRG Homepage Conflicts of Interest - What are Conflicts of Interest? - Submitting your Conflicts Survey - Submitting a Travel Disclosure -
CMS for Accounting. Examiner Certification System User Guide
 CMS for Accounting Examiner Certification System User Guide VERSION 1.0 JANUARY 2019 0 Table of Contents The Examiner Certification System Overview...2 User Privileges and Actions...2 Examiner Certification
CMS for Accounting Examiner Certification System User Guide VERSION 1.0 JANUARY 2019 0 Table of Contents The Examiner Certification System Overview...2 User Privileges and Actions...2 Examiner Certification
SUBMITTING A NEW IBC PROJECT APPLICATION (INITIAL)
 SUBMITTING A After you have established and activated your username and password, you can begin to create an IBC application in IRBNet. To start building a new IBC application, you must first CREATE A
SUBMITTING A After you have established and activated your username and password, you can begin to create an IBC application in IRBNet. To start building a new IBC application, you must first CREATE A
Cayuse424 Reference Manual for UW-Madison Revised January 2010 Check for updates to both manual and software.
 Cayuse424 Reference Manual for UW-Madison Revised January 2010 Check www.rsp.wisc.edu/cayuse for updates to both manual and software. How to Use This Manual This manual was designed to guide you through
Cayuse424 Reference Manual for UW-Madison Revised January 2010 Check www.rsp.wisc.edu/cayuse for updates to both manual and software. How to Use This Manual This manual was designed to guide you through
Top Ten Questions About IRBNet.org
 Top Ten Questions About IRBNet.org 1. How do I register? Registration is easy. To register, simply navigate your web browser www.irbnet.org. The login area for IRBNet is in the upper right corner of the
Top Ten Questions About IRBNet.org 1. How do I register? Registration is easy. To register, simply navigate your web browser www.irbnet.org. The login area for IRBNet is in the upper right corner of the
Electronic MTA Training Manual & User Guide for Administrative Staff & Principal Investigators
 Electronic MTA Training Manual & User Guide for Administrative Staff & Principal Investigators 1 P a g e Table of Contents Getting Started... 3 Introduction... 3 Helpful Hints... 3 Login Process... 6 Profile...
Electronic MTA Training Manual & User Guide for Administrative Staff & Principal Investigators 1 P a g e Table of Contents Getting Started... 3 Introduction... 3 Helpful Hints... 3 Login Process... 6 Profile...
TRAQ DSS Form. Researchers - User Manual
 TRAQ DSS Form Researchers - User Manual January 2014 Introduction to TRAQ TRAQ (Tools for Research at Queen s) is an electronic research management system which replaced all internal certification paper
TRAQ DSS Form Researchers - User Manual January 2014 Introduction to TRAQ TRAQ (Tools for Research at Queen s) is an electronic research management system which replaced all internal certification paper
Research Funding Module InfoEd User Guide. Version 3.3
 Research Funding Module InfoEd User Guide Version 3.3 Table of Contents Login... 3 Create New Proposal or Expression of Interest (EOI)... 3 Primary Application Details... 4 Principal Investigator Information...
Research Funding Module InfoEd User Guide Version 3.3 Table of Contents Login... 3 Create New Proposal or Expression of Interest (EOI)... 3 Primary Application Details... 4 Principal Investigator Information...
INSTRUCTIONS FOR THE eiaf COMPLETING THE FORM AND REQUIRED APPROVALS AND CERTIFICATIONS
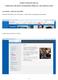 INSTRUCTIONS FOR THE eiaf COMPLETING THE FORM AND REQUIRED APPROVALS AND CERTIFICATIONS ACCESSING A NEW BLANK FORM From the UK website, click on link blue. (Click refers to pressing the mouse button.)
INSTRUCTIONS FOR THE eiaf COMPLETING THE FORM AND REQUIRED APPROVALS AND CERTIFICATIONS ACCESSING A NEW BLANK FORM From the UK website, click on link blue. (Click refers to pressing the mouse button.)
Click For Industry Professionals From the toolbar click Regulatory Filing & Reporting Under Related Links, click Entitlement Program
 Table of Contents Overview of the Advertising Regulation Electronic Files (AREF) System... 1 Getting Started Entitlement... 1 AREF Users... 2 Adding Submit an Advertisement to My Quicklinks... 2 The AREF
Table of Contents Overview of the Advertising Regulation Electronic Files (AREF) System... 1 Getting Started Entitlement... 1 AREF Users... 2 Adding Submit an Advertisement to My Quicklinks... 2 The AREF
Reminder: Please refer to the 2018 Convening & Collaborating (C 2 ) competition page for more details.
 Reminder: Please refer to the 2018 Convening & Collaborating (C 2 ) competition page for more details. Starting the Process Login to MSFHR ApplyNet. IMPORTANT: If you have forgotten your login ID for your
Reminder: Please refer to the 2018 Convening & Collaborating (C 2 ) competition page for more details. Starting the Process Login to MSFHR ApplyNet. IMPORTANT: If you have forgotten your login ID for your
Human Subjects Development Guide
 Research Integrity & Compliance Institutional Review Board Human Subjects Development Guide ERA Version 15, Nov. 2016 Table of Contents Logging In... 3 Adding a Delegate... 4 Viewing Items as a Delegate...
Research Integrity & Compliance Institutional Review Board Human Subjects Development Guide ERA Version 15, Nov. 2016 Table of Contents Logging In... 3 Adding a Delegate... 4 Viewing Items as a Delegate...
Project Modification Request 9.1 Financial System Campus Users Guide Sections of the PMR Form/Template
 Project Modification Request 9.1 Financial System Campus Users Guide Sections of the PMR Form/Template 6/2014 Project Modification Request INTRODUCTION: The Project Modification Request (PMR) Module has
Project Modification Request 9.1 Financial System Campus Users Guide Sections of the PMR Form/Template 6/2014 Project Modification Request INTRODUCTION: The Project Modification Request (PMR) Module has
Cayuse Submission Guidelines University Facilitating Fund. Table of Contents. Table of Contents
 Cayuse Submission Guidelines University Facilitating Fund Table of Contents Each topic is linked to its location in the guidance document. Table of Contents Browser Settings 2 Registration 2 Logging Into
Cayuse Submission Guidelines University Facilitating Fund Table of Contents Each topic is linked to its location in the guidance document. Table of Contents Browser Settings 2 Registration 2 Logging Into
ARCHIBUS Web Central: Service Requests
 : Service Requests Table of Contents SERVICE REQUESTS... 3 ENTERING A SERVICE REQUEST... 3 Rand Worldwide Enabling Engineering Innovation 2 Service Requests Entering a Service Request As an employee of
: Service Requests Table of Contents SERVICE REQUESTS... 3 ENTERING A SERVICE REQUEST... 3 Rand Worldwide Enabling Engineering Innovation 2 Service Requests Entering a Service Request As an employee of
DISCOVR-e USER MANUAL. Vanderbilt University Human Research Protection Program
 DISCOVR-e USER MANUAL Vanderbilt University Human Research Protection Program Table of Contents Introduction and Overview... 3 Log into the System... 4 Investigator Dashboard... 5 Submitting a New Study...
DISCOVR-e USER MANUAL Vanderbilt University Human Research Protection Program Table of Contents Introduction and Overview... 3 Log into the System... 4 Investigator Dashboard... 5 Submitting a New Study...
NYU Cayuse IRB Manual
 IRB NYU Cayuse IRB Manual prepared by the NYU UCAIHS (University Committee on Activities Involving Human Subjects) What is Cayuse? The Cayuse Research Suite is NYU s system to support the submission of
IRB NYU Cayuse IRB Manual prepared by the NYU UCAIHS (University Committee on Activities Involving Human Subjects) What is Cayuse? The Cayuse Research Suite is NYU s system to support the submission of
Wisconsin Department of Justice. Egrants System User Guide School Safety Initiative
 Wisconsin Department of Justice Egrants System User Guide School Safety Initiative 2 What is in this guide? This guide describes technical steps describing how to apply for and manage a grant in Egrants.
Wisconsin Department of Justice Egrants System User Guide School Safety Initiative 2 What is in this guide? This guide describes technical steps describing how to apply for and manage a grant in Egrants.
PCORI Online: Awardee User Guide Research Awards
 PCORI Online: Awardee User Guide Research Awards Updated as of 1/31/18 1 Table of Contents Section 1: Introduction to PCORI Online... 3 1.1 Getting Started - Tips for Using PCORI Online... 4 1.2 Logging
PCORI Online: Awardee User Guide Research Awards Updated as of 1/31/18 1 Table of Contents Section 1: Introduction to PCORI Online... 3 1.1 Getting Started - Tips for Using PCORI Online... 4 1.2 Logging
2018 National Children s Alliance
 2018 National Children s Alliance NCA is using Micro Edge IGAM software. This software supports an online application, grant review and report submissions. Before beginning the online application process,
2018 National Children s Alliance NCA is using Micro Edge IGAM software. This software supports an online application, grant review and report submissions. Before beginning the online application process,
Cayuse Submission Guidelines Cross-Disciplinary Research Fund Table of Contents
 Cayuse Submission Guidelines Cross-Disciplinary Research Fund Table of Contents Each topic is linked to its location in the guidance document. Browser Settings... 2 Registration... 2 Logging Into Cayuse...
Cayuse Submission Guidelines Cross-Disciplinary Research Fund Table of Contents Each topic is linked to its location in the guidance document. Browser Settings... 2 Registration... 2 Logging Into Cayuse...
Project and Award Searches
 Project and Award Searches Contents Find Awards/Projects Search... 2 Award Page... 4 Project Inclusion... 4 Financials... 5 Summary View... 5 Calculator... 7 Projects View... 7 Period Summary... 8 AR...
Project and Award Searches Contents Find Awards/Projects Search... 2 Award Page... 4 Project Inclusion... 4 Financials... 5 Summary View... 5 Calculator... 7 Projects View... 7 Period Summary... 8 AR...
Concur California State University, Stanislaus Financial Services
 Concur California State University, Stanislaus Financial Services Employee Reimbursements Non-Travel Accounts Payable 1 Table of Contents Getting Started... 3 Set up your Profile... 5 Enter your Information...
Concur California State University, Stanislaus Financial Services Employee Reimbursements Non-Travel Accounts Payable 1 Table of Contents Getting Started... 3 Set up your Profile... 5 Enter your Information...
MyPermitNow.org. Search Permits. page 5. Click to Search Permits from the left navigation.
 Search Permits Click to Search Permits from the left navigation. Select your state. This will cause the Jurisdiction dropdown menu to load all participating jurisdictions for that state. Select your Jurisdiction.
Search Permits Click to Search Permits from the left navigation. Select your state. This will cause the Jurisdiction dropdown menu to load all participating jurisdictions for that state. Select your Jurisdiction.
proposalcentral Prepare and Submit a Proposal.
 proposalcentral Prepare and Submit a Proposal. If you need assistance, contact Customer Service by email at pcsupport@altum.com or by phone at 1-800-875-2562 1 Recommended Software proposalcentral Recommends
proposalcentral Prepare and Submit a Proposal. If you need assistance, contact Customer Service by email at pcsupport@altum.com or by phone at 1-800-875-2562 1 Recommended Software proposalcentral Recommends
UVMClick IRB Study Submission Guide
 UVMClick IRB Study Submission Guide September 2018 Table of Contents How to Login 3 How to Create a New Study 4 Find More Information 5 How to Edit a Study 6 Check the Study for Errors 7 Submit the Study
UVMClick IRB Study Submission Guide September 2018 Table of Contents How to Login 3 How to Create a New Study 4 Find More Information 5 How to Edit a Study 6 Check the Study for Errors 7 Submit the Study
eprost System Policies & Procedures
 eprost System Policies & Procedures Initial Approval Date: 12/07/2010 Revision Date: 02/25/2011 Introduction eprost [ Electronic Protocol Submission and Tracking ] is the Human Subject Research Office's
eprost System Policies & Procedures Initial Approval Date: 12/07/2010 Revision Date: 02/25/2011 Introduction eprost [ Electronic Protocol Submission and Tracking ] is the Human Subject Research Office's
eprotocol Investigator Role Manual
 eprotocol Investigator Role Manual Table of Contents Table of Contents Auto-Population of Stored User Data...17 5.4 Module Specific Work Flow Sections...18 1 Overview...4 1.1 Things to Remember...4 2 Creating
eprotocol Investigator Role Manual Table of Contents Table of Contents Auto-Population of Stored User Data...17 5.4 Module Specific Work Flow Sections...18 1 Overview...4 1.1 Things to Remember...4 2 Creating
Manual author: Thomas Bozza Program Manager- Streamlyne (NJIT Grants Management Software)
 PROPOSAL DEVELOPMENT- PRE AWARD TRAINING MANUAL: PROPOSALS & BUDGETS Manual author: Thomas Bozza Program Manager- Streamlyne (NJIT Grants Management Software) TABLE OF CONTENTS Slide # Content/Streamlyne
PROPOSAL DEVELOPMENT- PRE AWARD TRAINING MANUAL: PROPOSALS & BUDGETS Manual author: Thomas Bozza Program Manager- Streamlyne (NJIT Grants Management Software) TABLE OF CONTENTS Slide # Content/Streamlyne
DEPARTMENT OF EDUCATION. LEA Accounting
 DEPARTMENT OF EDUCATION LEA ACCOUNTING Contents Revision History... 2 Revision History Chart... 2 Application Permissions... 3 Creating an AIM Account... 4 Create an Account Email Address Entered... 5
DEPARTMENT OF EDUCATION LEA ACCOUNTING Contents Revision History... 2 Revision History Chart... 2 Application Permissions... 3 Creating an AIM Account... 4 Create an Account Email Address Entered... 5
Researcher User Manual
 Researcher User Manual Post-Review Application Management Cloning Applications, Creating and Managing Events Audience: Principal Investigators & Project Team Members Updated: December 1, 2016 Checkpoint
Researcher User Manual Post-Review Application Management Cloning Applications, Creating and Managing Events Audience: Principal Investigators & Project Team Members Updated: December 1, 2016 Checkpoint
HOW TO SUBMIT A POSTER PRESENTATION FOR THE NAN ANNUAL CONFERENCE
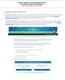 HOW TO SUBMIT A POSTER PRESENTATION FOR THE NAN ANNUAL CONFERENCE Submission Deadline: March 29, 2019 1. Click here to access the submission site. 2. NAN Account Holders: please click on the Login. This
HOW TO SUBMIT A POSTER PRESENTATION FOR THE NAN ANNUAL CONFERENCE Submission Deadline: March 29, 2019 1. Click here to access the submission site. 2. NAN Account Holders: please click on the Login. This
SUBMITTING AN IBC AMENDMENT
 The submission of an amendment/modification to an approved IBC protocol requires the creation of a subsequent PACKAGE in a project. After an IBC application is approved, the project may require modifications
The submission of an amendment/modification to an approved IBC protocol requires the creation of a subsequent PACKAGE in a project. After an IBC application is approved, the project may require modifications
Heart and Stroke Foundation CIRCUlink
 Heart and Stroke Foundation CIRCUlink APPLICANT USER GUIDE How to submit a Grant-in-Aid application online Page 1 Contents 1. Introduction... 3 2. Before you begin using CIRCUlink... 3 3. Accessing CIRCUlink...
Heart and Stroke Foundation CIRCUlink APPLICANT USER GUIDE How to submit a Grant-in-Aid application online Page 1 Contents 1. Introduction... 3 2. Before you begin using CIRCUlink... 3 3. Accessing CIRCUlink...
IDT Quick Reference Guide 2017
 This reference guide will help campus users navigate through the most common method to reclassify transactions between accounts or cost centers. In most cases, the Easy IDT button embedded within the University
This reference guide will help campus users navigate through the most common method to reclassify transactions between accounts or cost centers. In most cases, the Easy IDT button embedded within the University
HIRING MANAGER S JOB SITE USER S GUIDE. Fitchburg State University Hiring System
 HIRING MANAGER S JOB SITE USER S GUIDE Fitchburg State University Hiring System TABLE OF CONTENTS INTRODUCTION... 3 GETTING STARTED... 5 CREATING A POSTING.7 Creating Posting from Position Type... 7 Posting
HIRING MANAGER S JOB SITE USER S GUIDE Fitchburg State University Hiring System TABLE OF CONTENTS INTRODUCTION... 3 GETTING STARTED... 5 CREATING A POSTING.7 Creating Posting from Position Type... 7 Posting
Guide to Completing the Online Application
 Guide to Completing the Online Application Table of Contents 1. Before You Begin.......2 a. Invitation to Apply....2 b. Please Sign In.....2 c. Application Time Out...........3 d. Navigating the Online
Guide to Completing the Online Application Table of Contents 1. Before You Begin.......2 a. Invitation to Apply....2 b. Please Sign In.....2 c. Application Time Out...........3 d. Navigating the Online
The IACUC is charged with the responsibility of ensuring that the Animal Welfare Act policies and procedures are followed.
 Protis is an online application system in support of research activities that require approval by the Institutional Animal Care and Use Committee (IACUC) or the Institutional Review Board (IRB). The IACUC
Protis is an online application system in support of research activities that require approval by the Institutional Animal Care and Use Committee (IACUC) or the Institutional Review Board (IRB). The IACUC
Animal Protocol Development Instructions for Researchers
 Animal Protocol Development Instructions for Researchers OFFICE OF THE VICE PRESIDENT FOR RESEARCH TOPAZ Elements - Protocol Development Instructions for Researchers Page 1 Topaz Elements Animal Protocol
Animal Protocol Development Instructions for Researchers OFFICE OF THE VICE PRESIDENT FOR RESEARCH TOPAZ Elements - Protocol Development Instructions for Researchers Page 1 Topaz Elements Animal Protocol
NAU IRBNet Application
 NAU IRBNet Application Guidance for NAU Research Application Revised 17-0202 Go to IRBNet Website address: irbnet.org/release/index.html Login to your account - Enter your Username and Password and select
NAU IRBNet Application Guidance for NAU Research Application Revised 17-0202 Go to IRBNet Website address: irbnet.org/release/index.html Login to your account - Enter your Username and Password and select
Grants Module 101. A Quick Reference Guide for Inquiry into Grants Contracts and Grants April 2016
 Grants Module 101 A Quick Reference Guide for Inquiry into Grants Contracts and Grants April 2016 Table of Contents Adding Pages to Favorites Pages 2-3 Viewing Information on the Award Profile Pages 4-8
Grants Module 101 A Quick Reference Guide for Inquiry into Grants Contracts and Grants April 2016 Table of Contents Adding Pages to Favorites Pages 2-3 Viewing Information on the Award Profile Pages 4-8
User Home Screen Quick Reference Guide
 User Home Screen USER HOME The User Home screen is the hub by which you can access all the features of UNCG Jobsearch. SHORTCUTS Quick access for commonly used tasks. MY LINKS Links to relevant info such
User Home Screen USER HOME The User Home screen is the hub by which you can access all the features of UNCG Jobsearch. SHORTCUTS Quick access for commonly used tasks. MY LINKS Links to relevant info such
Office of Research Integrity / Office of Sponsored Projects IACUC PROTOCOL SUBMISSION GUIDE
 Office of Research Integrity / Office of Sponsored Projects IACUC PROTOCOL SUBMISSION GUIDE Coeus Lite 4.5.1.Px Document Version 3 Updated May 2016 Table of Contents ABOUT COEUS... 3 o About the Coeus
Office of Research Integrity / Office of Sponsored Projects IACUC PROTOCOL SUBMISSION GUIDE Coeus Lite 4.5.1.Px Document Version 3 Updated May 2016 Table of Contents ABOUT COEUS... 3 o About the Coeus
STEP-BY-STEP: PI OFFLINE CERTIFICATION
 STEP-BY-STEP: PI OFFLINE CERTIFICATION APRIL 24 TH, 2015 ***YOU MUST HAVE A PI SIGNED AND DATED COPY OF THE CERTIFICATON UPLOADED IN THE PROPOSAL RECORD IN ORDER TO CERTIFY ON BEHALF OF A PI*** OFFLINE
STEP-BY-STEP: PI OFFLINE CERTIFICATION APRIL 24 TH, 2015 ***YOU MUST HAVE A PI SIGNED AND DATED COPY OF THE CERTIFICATON UPLOADED IN THE PROPOSAL RECORD IN ORDER TO CERTIFY ON BEHALF OF A PI*** OFFLINE
Introduction to the Online Proposal Routing System
 Introduction to the Online Proposal Routing System Prepared by Ramu Ramachandran and Sangam Mulmi on behalf of The Office of Sponsored Projects Last revised: November 14, 2018 First things first: 1. Download
Introduction to the Online Proposal Routing System Prepared by Ramu Ramachandran and Sangam Mulmi on behalf of The Office of Sponsored Projects Last revised: November 14, 2018 First things first: 1. Download
Signing Authority Manual
 Signing Authority Manual Reviewing Applications for Department and Faculty St. John s Campus, Grenfell Campus, and Marine Institute Updated: November 24, 2017 1 Checkpoint *PLEASE NOTE* Prior to leveraging
Signing Authority Manual Reviewing Applications for Department and Faculty St. John s Campus, Grenfell Campus, and Marine Institute Updated: November 24, 2017 1 Checkpoint *PLEASE NOTE* Prior to leveraging
IRBNet User Manual IACUC
 IRBNet User Manual IACUC Version 5/29/2012 1 User Manual This user manual is designed to assist in the use of IRBNet. You will find assistance in registration, submission, continuing review, amendments
IRBNet User Manual IACUC Version 5/29/2012 1 User Manual This user manual is designed to assist in the use of IRBNet. You will find assistance in registration, submission, continuing review, amendments
IRBNet for IACUC The Electronic Platform for Submission and Processing of Animals Study Documents
 IRBNet for IACUC The Electronic Platform for Submission and Processing of Animals Study Documents Please see the attached information regarding the submission of your protocol via IRBNet for IACUC. NOTE:
IRBNet for IACUC The Electronic Platform for Submission and Processing of Animals Study Documents Please see the attached information regarding the submission of your protocol via IRBNet for IACUC. NOTE:
Documentation for Non-Medical Research Ethics Board Researchers Full Board and Delegated Board Review
 Documentation for Non-Medical Research Ethics Board Researchers Full Board and Delegated Board Review July 23, 2013 Office of Research Ethics If you run into any difficulties or have questions about Romeo,
Documentation for Non-Medical Research Ethics Board Researchers Full Board and Delegated Board Review July 23, 2013 Office of Research Ethics If you run into any difficulties or have questions about Romeo,
Getting Started With Web Mail Help Desk
 INET010 Feb 2008 Getting Started With Web Mail Help Desk Overview... 1 Login and Logout... 2 Basic Navigation... 2 Basic Tasks... 2 Using Folders... 5 Attachments... 7 Address Book... 8 Options and Other
INET010 Feb 2008 Getting Started With Web Mail Help Desk Overview... 1 Login and Logout... 2 Basic Navigation... 2 Basic Tasks... 2 Using Folders... 5 Attachments... 7 Address Book... 8 Options and Other
Proposal Approver Addendum COEUS LITE V Proposal Development: The Proposal Approver Addendum
 COEUS LITE V4.4.4.1 Proposal Development: The Proposal Approver Addendum Table of Contents Knowing When a Proposal is Available for Review... 3 Notifications in the COEUS Inbox... 3 Emailed Notification...
COEUS LITE V4.4.4.1 Proposal Development: The Proposal Approver Addendum Table of Contents Knowing When a Proposal is Available for Review... 3 Notifications in the COEUS Inbox... 3 Emailed Notification...
Main Toolbar. Toolbars Icon Cheat Sheet. The Coeus Main toolbar, located below the menu bar, is consistent in every module of Coeus.
 Toolbars Icon Cheat Sheet Main Toolbar The Coeus Main toolbar, located below the menu bar, is consistent in every module of Coeus. Maintain Inbox - Inbox contains messages regarding routing and approval
Toolbars Icon Cheat Sheet Main Toolbar The Coeus Main toolbar, located below the menu bar, is consistent in every module of Coeus. Maintain Inbox - Inbox contains messages regarding routing and approval
How do I set up or update my GENIUS Profile?
 Oct. 2 How do I set up or update my GENIUS Profile? General Information Your GENIUS Profile stores non-sensitive information about you and your research interests at UNH. InfoEd uses your profile to pre-populate
Oct. 2 How do I set up or update my GENIUS Profile? General Information Your GENIUS Profile stores non-sensitive information about you and your research interests at UNH. InfoEd uses your profile to pre-populate
Submitting an Protocol for De Novo Review
 Submitting an Protocol for De Novo Review Important: A protocol can be created, edited and submitted by the Principal Investigator (PI), Laboratory Contact or Alternate Lab Contact. However, ONLY the Principal
Submitting an Protocol for De Novo Review Important: A protocol can be created, edited and submitted by the Principal Investigator (PI), Laboratory Contact or Alternate Lab Contact. However, ONLY the Principal
Student Guide to Submitting a Thesis or Dissertation at Texas A&M
 Student Guide to Submitting a Thesis or Dissertation at Texas A&M To begin your submission please visit: http://etd.tamu.edu When you click Start your submittal, this is the screen you will see. You will
Student Guide to Submitting a Thesis or Dissertation at Texas A&M To begin your submission please visit: http://etd.tamu.edu When you click Start your submittal, this is the screen you will see. You will
Romeo. How to Apply for a Faculty Conference Travel Grant
 Romeo How to Apply for a Faculty Conference Travel Grant Please note: Romeo is compatible with Internet Explorer, Firefox, Edge, Google Chrome and Safari. If you have any problems or questions, please
Romeo How to Apply for a Faculty Conference Travel Grant Please note: Romeo is compatible with Internet Explorer, Firefox, Edge, Google Chrome and Safari. If you have any problems or questions, please
Faculty Recruitment using PeopleAdmin
 Table of Contents Accessing PeopleAdmin... 2 Determining the Appropriate Role... 2 Changing your Role... 3 Create a Posting... 4 Steps to take before beginning your posting... 4 Creating the posting...
Table of Contents Accessing PeopleAdmin... 2 Determining the Appropriate Role... 2 Changing your Role... 3 Create a Posting... 4 Steps to take before beginning your posting... 4 Creating the posting...
* Slide the scroll bar to display more fields to search.
 Brown University - OSP COEUS GUIDE The Basics for Viewers ACCESSING COEUS PREMIUM Click on the Coeus Premium link on the Coeus Homepage: https://coeus.brown.edu/prod/ Enter your Brown User Name and Password
Brown University - OSP COEUS GUIDE The Basics for Viewers ACCESSING COEUS PREMIUM Click on the Coeus Premium link on the Coeus Homepage: https://coeus.brown.edu/prod/ Enter your Brown User Name and Password
HOW TO SUBMIT A POSTER PRESENTATION FOR THE NAN ANNUAL CONFERENCE Submission Deadline: April 1, 2016
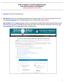 HOW TO SUBMIT A POSTER PRESENTATION FOR THE NAN ANNUAL CONFERENCE Submission Deadline: April 1, 2016 1. Click here to access the submission site. 2. NAN Members enter your email address and Member ID number
HOW TO SUBMIT A POSTER PRESENTATION FOR THE NAN ANNUAL CONFERENCE Submission Deadline: April 1, 2016 1. Click here to access the submission site. 2. NAN Members enter your email address and Member ID number
UNIVERSITY OF NORTH FLORIDA. Institutional Review Board (IRB) Guide to IRBNet:
 UNIVERSITY OF NORTH FLORIDA Institutional Review Board (IRB) Guide to IRBNet: Version 2 11-15-2013 Table of Contents i Introduction 2 IRBNet User Registration 2 IRBNet ID Number 2 Training and Credentials
UNIVERSITY OF NORTH FLORIDA Institutional Review Board (IRB) Guide to IRBNet: Version 2 11-15-2013 Table of Contents i Introduction 2 IRBNet User Registration 2 IRBNet ID Number 2 Training and Credentials
The Center for Translational Medicine at UT Southwestern Medical Center. The Research Launchpad: Your One-Stop Shop to CTSA Resources and Services
 The Center for Translational Medicine at UT Southwestern Medical Center The Research Launchpad: Your One-Stop Shop to CTSA Resources and Services Users Guide Table of Contents a. Introduction b. Where
The Center for Translational Medicine at UT Southwestern Medical Center The Research Launchpad: Your One-Stop Shop to CTSA Resources and Services Users Guide Table of Contents a. Introduction b. Where
From the Online Tools list, scroll down to SBS Connect, and click on the Register for SBS Connect link. The SBS Connect login screen loads.
 SBS EXTERNAL HEALTHCARE REVIEW USER GUIDE Create New Account Register an Entity View Attachment Upload Attachment SBS CONNECT CREATE NEW ACCOUNT Before using SBS Connect for the first time, 1) create an
SBS EXTERNAL HEALTHCARE REVIEW USER GUIDE Create New Account Register an Entity View Attachment Upload Attachment SBS CONNECT CREATE NEW ACCOUNT Before using SBS Connect for the first time, 1) create an
1) Log in to Therap https://secure.therapservices.net/auth/login 2) From the dashboard, check to see if your profile is set to Internal.
 Initiating a New Case Note How to Create a Case Note- Early Intervention Steps that will be used in this How-To process. 1. Initiating a New Case Note 2. Entering Data on a Case Note 3. Submitting a Case
Initiating a New Case Note How to Create a Case Note- Early Intervention Steps that will be used in this How-To process. 1. Initiating a New Case Note 2. Entering Data on a Case Note 3. Submitting a Case
FIELDS REQUIRED TO SAVE A PROTOCOL
 Human Subject Research Protocols If conducting human subject research, a protocol must be submitted in WVU+kc to the WVU Institutional Review Board for review. Click on the Create IRB Protocol link located
Human Subject Research Protocols If conducting human subject research, a protocol must be submitted in WVU+kc to the WVU Institutional Review Board for review. Click on the Create IRB Protocol link located
Applying for Funding in Fluxx. Quick Start Instructions
 Applying for Funding in Fluxx Quick Start Instructions GETTING STARTED The Hogg Foundation Fluxx Grant Portal is optimized for use with Chrome or Safari browsers and using another browser may cause technical
Applying for Funding in Fluxx Quick Start Instructions GETTING STARTED The Hogg Foundation Fluxx Grant Portal is optimized for use with Chrome or Safari browsers and using another browser may cause technical
Kuali Research User Guide: Create an IRB Protocol
 Kuali Research User Guide: Create an IRB Protocol Version.0: November 06 Purpose: To create an IRB Protocol document to be used for tracking new Human Subjects Research requests. Trigger / Timing / Frequency:
Kuali Research User Guide: Create an IRB Protocol Version.0: November 06 Purpose: To create an IRB Protocol document to be used for tracking new Human Subjects Research requests. Trigger / Timing / Frequency:
You may also scroll through the Table of Contents and click on the topic or question of interest.
 We have posted our IRB process related FAQs in a searchable PDF format. You may search by using the CTRL/F key combination. Just type the search word in the box that appears. You may also scroll through
We have posted our IRB process related FAQs in a searchable PDF format. You may search by using the CTRL/F key combination. Just type the search word in the box that appears. You may also scroll through
KIAS - Kansas Integrated Accountability System
 Quick Start Guide for LEA Users Discipline Data Collection 2018 Leader Services Introduction This document will explain how LEAs can use the Kansas Integrated Accountability System (KIAS) to complete the
Quick Start Guide for LEA Users Discipline Data Collection 2018 Leader Services Introduction This document will explain how LEAs can use the Kansas Integrated Accountability System (KIAS) to complete the
How To Use NCA s Online Grant Application System
 How To Use NCA s Online Grant Application System NCA is using MicroEdge IGAM software. This software supports an online application, grant review and report submissions. Before beginning the online application
How To Use NCA s Online Grant Application System NCA is using MicroEdge IGAM software. This software supports an online application, grant review and report submissions. Before beginning the online application
STUDY ASSISTANT. Study Assistant. Version 10.03
 STUDY ASSISTANT Study Assistant Version 10.03 Contents Introduction... 3 Add a New Study... 3 My Studies... 3 Find a Study... 9 Find a Study for All Users... 12 imedris Data Corporation 2 Study Assistant
STUDY ASSISTANT Study Assistant Version 10.03 Contents Introduction... 3 Add a New Study... 3 My Studies... 3 Find a Study... 9 Find a Study for All Users... 12 imedris Data Corporation 2 Study Assistant
COP Management Grid (Department)
 COP Management Grid (Department) Purpose...1 Considerations...2 Assignments:...2 Worklist:...3 COP Management Grid...3 Elements of the Page:...4 Automatic Closeout Packet Creation:...5 When COP Management
COP Management Grid (Department) Purpose...1 Considerations...2 Assignments:...2 Worklist:...3 COP Management Grid...3 Elements of the Page:...4 Automatic Closeout Packet Creation:...5 When COP Management
the address book training guide
 the address book training guide 2015, The Trustees of Indiana University 1 table of Contents Table of Contents 2 table of Contents 3 about this document 4 how to access the address book 5 address book
the address book training guide 2015, The Trustees of Indiana University 1 table of Contents Table of Contents 2 table of Contents 3 about this document 4 how to access the address book 5 address book
