HMMcopy: A package for bias-free copy number estimation and robust CNA detection in tumour samples from WGS HTS data
|
|
|
- Brian McGee
- 5 years ago
- Views:
Transcription
1 HMMcopy: A package for bias-free copy number estimation and robust CNA detection in tumour samples from WGS HTS data Daniel Lai and Gavin Ha June 13, 2018 Contents 1 Introduction 1 2 Generating Copy Number Profiles Obtaining HTS copy number data Loading HTS copy number data Correcting HTS copy number data Visualizing the effects of correction Visualizing corrected copy number profiles Correcting and visualizing tumour copy number profiles Segmentation and Classification of Copy Number Profiles Visualizing segments and classified states Improving segmentation performance Reducing the number of segments Adjusting copy number state ranges Understanding parameter convergence Overriding parameter convergence Matched Tumour-Normal Sample Correction Normalizing tumour by normal copy number profiles Session Information 13 1 Introduction High-throughput whole genome DNA sequencing results in millions of reads which after short-read mapping, can be used to infer the original sequence of the sample. An additional piece of information we gain from mapping is the coverage or number of reads overlapping each position, which can be used as a rough estimate of copy number. In practice, it is often easier to work at a lower resolution, and so we divide the genome into non-overlapping windows of fixed width (e.g. 1000), and use the readcount or number of reads starting in each of these windows (henceforth termed bins). The main assumption of using readcount as a proxy for copy number is that during the sequencing protocol, reads are uniformly sampled from all genetic material present in the original input sample. This assumption has been shown to be incorrect, with reads being sampled at unequal rates depending on the GC content, among other things [1]. To different extents, this bias exists and varies in all platforms, protocols, and samples. Additionally, Benjamini and Speed [1] show that two libraries prepared from the same DNA source show different biases, 1
2 which highlights the fact that matched sample normalization (i.e. normalizing tumour copy by matched normal reference) is insufficient to eliminate this bias. 2 Generating Copy Number Profiles For the remainder of this vignette, we will go through a short example using a matched normal tumour dataset, and show the ease and effects of normalization readcount by GC and mappability. The dataset that is described here belongs to a female triple-negative breast cancer patient from the published dataset [2, 3]. This genome library was sequenced on the ABI/Life SOLiD platform, generating hybrid lengths of 25bp and 50bp paired-end reads. The reads were aligned using BioScope ( where reads mapped to multiple sites were ignored. 2.1 Obtaining HTS copy number data It should be noted that due to memory and speed considerations, the raw input that feeds in to this package are required to be generated by programs distributed as part of the HMMcopy Suite available at: In short, the suite has tools to: ˆ Obtain high resolution bin counts for large ( 250GB) BAM files within a few hours. ˆ Obtain GC content for bins from standard FASTA files within minutes for a human genome. ˆ Obtain average mappability for bins from BigWig within minutes, or FASTA files within a day for a human genome. 2.2 Loading HTS copy number data To show the effects of correction in a simple case, let us begin by correcting data obtained from a normal genome sample. To start, we load in WIG files containing three sets of data, namely readcounts, GC content, and average mappability, pre-computed for each bin with an external applications provided by the HMMcopy Suite. Briefly, the readcounts data consists of the number of reads mapped within the boundaries of each bin. The GC content file consists of G and C base proportions for each bin; -1 is used when the reference sequence for the bin contains at least one unknown nucleotide base (i.e. N, rather than A,C, T or G). The mappability file, which was generated using ENCODE Duke Uniqueness of 35bp sequences track from UCSC ( provides scores indicating how mappable the reference sequence at each bin is to aligners; the higher the score, the more mappable the sequence. library(hmmcopy) rfile <- system.file("extdata", "normal.wig", package = "HMMcopy") gfile <- system.file("extdata", "gc.wig", package = "HMMcopy") mfile <- system.file("extdata", "map.wig", package = "HMMcopy") normal_reads <- wigstorangeddata(rfile, gfile, mfile) normal_reads[1000:1010, ] RangedData with 11 rows and 3 value columns across 1 space space ranges reads gc map <factor <IRanges <integer <numeric <numeric
3 Correcting HTS copy number data The resultant output is an IRanges object, which can easily use other Bioconductor functions and packages for analysis and visualzation. Although it is recommended that the output be left untouched until furthur correction as follows: normal_copy <- correctreadcount(normal_reads) normal_copy[1000:1010, ] RangedData with 11 rows and 8 value columns across 1 space space ranges reads gc map valid <factor <IRanges <integer <numeric <numeric <logical TRUE TRUE TRUE TRUE TRUE TRUE TRUE TRUE TRUE TRUE TRUE ideal cor.gc cor.map copy <logical <numeric <numeric <numeric 1 FALSE TRUE TRUE TRUE FALSE TRUE TRUE TRUE FALSE TRUE TRUE The corrected reads are in the two columns cor.gc and cor.map, which are corrected and normalized, effectively a copy number estimation for each bin. For downstream analysis, copy is used, which is simply the base 2 log values of cor.map, to normalize the differences between values above and below 1. Specifically, the columns output from this process are: valid Bins with valid GC and average mappability and non-zero read 3
4 ideal Valid bins of high mappability and reads that are not outliers cor.gc Readcounts after the first GC correction step cor.map cor.gc readcounts after a furthur mappability correction copy cor.map transformed into log2 space 2.4 Visualizing the effects of correction To best visualize the biases in the original sample and the effects of correction, convenience plotting functions are available as follows: par(cex.main = 0.7, cex.lab = 0.7, cex.axis = 0.7, mar = c(4, 4, 2, 0.5)) plotbias(normal_copy, pch = 20, cex = 0.5) 4
5 As observed, a unimodal relationship typically exists between readcount and GC content (excuse the weak tail), which is corrected and eliminated in the GC correction step. The GC-corrected readcounts exhibit a slight correlation with mappability, which is corrected in the second step. 2.5 Visualizing corrected copy number profiles To see the effects of correction for copy number estimation, we can plot the copy number along a segment of a chromosome as follows: par(mar = c(4, 4, 2, 0)) plotcorrection(normal_copy, pch = ".") 5
6 In the top track, the original uncorrected reads are used to estimate copy number (by simply dividing by the median value), followed by tracks with additional GC then mappability correction. Mediam absolute deviations (MAD) measuring the variance of each track can be seen to decrease after each correction step. 2.6 Correcting and visualizing tumour copy number profiles The identical process can be applied to tumour genomes using the same GC and mappability files as follows: tfile <- system.file("extdata", "tumour.wig", package = "HMMcopy") tumour_copy <- correctreadcount(wigstorangeddata(tfile, gfile, mfile)) par(mar = c(4, 4, 2, 0)) plotcorrection(tumour_copy, pch = ".") 6
7 Note that at this stage, the correction of the normal and tumour samples were done indepedently, meaning that HMMcopy is usable both in single sample and matched sample libraries. 3 Segmentation and Classification of Copy Number Profiles While corrected copy number data points are pleasing to stare at, they are often of much more practical use if we could group and classify them segments of specific copy number variation events. Included in the package is an implementation of an Hidden Markov Model [4] which does two main things: 1. Segments the copy number profile in regions predicted to be generated by the same copy number variation event 7
8 2. Predicts the copy number variation event for each segment, based on the biological likelihood of the event occuring tumour_segments <- HMMsegment(tumour_copy) 3.1 Visualizing segments and classified states We can visualize both the resultant segments and the states assigned to each segment easily as follows: par(mfrow = c(1, 1)) par(cex.main = 0.5, cex.lab = 0.5, cex.axis = 0.5, mar = c(2, 1.5, 0, 0), mgp = c(1, 0.5, 0)) plotsegments(tumour_copy, tumour_segments, pch = ".", + ylab = "Tumour Copy Number", xlab = "Chromosome Position") cols <- statecols() # 6 default state colours legend("topleft", c("homd", "HETD", "NEUT", "GAIN", "AMPL", "HLAMP"), + fill = cols, horiz = TRUE, bty = "n", cex = 0.5) As observed, there are six coloured states, corresponding to six biological CNA events: HOMD Homozygous deletion, 0 copies HETD Heterozygous deletion, 1 copy NEUT Neutral change, 2 copies GAIN Gain of chromosome, 3 copies AMPL Amplification event, 4 copies HLAMP High level amplification, 5 copies Segments are seen as neon green lines running through regions estimate to belong to the same CNA event. 3.2 Improving segmentation performance The segmentation program will attempt its best at creating the most biologically likely state assignment, but unfortunately and fortunately for us humans, the automated program can often fail, as seen in this specific example. In cases like these, there is then a need to adjust the parameters generated by the algorithm. Instead of getting parameters from scratch however, one can easily retrieve the default set of parameters as follows: 8
9 default_param <- HMMsegment(tumour_copy, getparam = TRUE) default_param strength e mu lambda nu kappa m eta gamma 1 1e e e e e e e e e e e e+04 3 S By calling the same segmentation algorithm with the getparam argument set to TRUE, the algorithm generates parameters without actually running the segmentation and returns it. There are 10 parameters explained in detail in the documentation (i.e.?hmmsegment), each having values across six states in each row. 3.3 Reducing the number of segments To reduce the segment, the two parameters we need to adjust are e and strength, which are the suggested probability of staying in a segment, and the strength of your suggestion to the algorithm. Once set, we run the segmentation algorithm, but this time expliciting providing our new parameters. longseg_param <- default_param longseg_param$e < longseg_param$strength <- 1e30 longseg_segments <- HMMsegment(tumour_copy, longseg_param, verbose = FALSE) And finally, we can visualize the results to confirm a decrease in segments as intended. par(cex.main = 0.5, cex.lab = 0.5, cex.axis = 0.5, mar = c(2, 1.5, 0, 0), mgp = c(1, 0.5, 0)) plotsegments(tumour_copy, longseg_segments, pch = ".", + ylab = "Tumour Copy Number", xlab = "Chromosome Position") legend("topleft", c("homd", "HETD", "NEUT", "GAIN", "AMPL", "HLAMP"), + fill = cols, horiz = TRUE, bty = "n", cex = 0.5) 9
10 As one can imagine, if for some odd reason you d like to increase the number of segments, simply decrease e and strength to a small value (close to 0). Although the algorithm will almost certainly take a few times longer to run. 3.4 Adjusting copy number state ranges A second more subtle and debilitating problem seen in this profile is actually the incorrect median of each copy number state. Specifically, this is a problem with the mu parameter. Accessing one of the output values of the segmentation process: longseg_segments$mus [,1] [,2] [,3] [,4] [,5] [,6] [1,] [2,] [3,] [4,] [5,] [6,] [,7] [1,] [2,] [3,] [4,] [5,] [6,] We see the median of 6 states (rows) after each iteration of the optimization algorithm (columns). The first column is our initial suggested mu: longseg_param$mu [1] And the last column is the actual values used during the segmentation process, which we can visualize as follows: 10
11 par(cex.main = 0.5, cex.lab = 0.5, cex.axis = 0.5, mar = c(2, 1.5, 0, 0), mgp = c(1, 0.5, 0)) plotsegments(tumour_copy, longseg_segments, pch = ".", + ylab = "Tumour Copy Number", xlab = "Chromosome Position") for(i in 1:nrow(longseg_segments$mus)) { + abline(h = longseg_segments$mus[i,ncol(longseg_segments$mus)], col = cols[i], + lwd = 2, lty = 3) + } abline(v = 7.68e7, lwd = 2, lty = 3) abline(v = 8.02e7, lwd = 2, lty = 3) From this, it becomes obvious that the medians are clearly not running through the middle of the visually obvious segments for many of the states. For example, the red medians for GAIN, AMPL and HLAMP are much too high, the green medians for HOMD and HETDare much too close together. Finally there is a clear segment flanked by vertical black dashed lines near the centre of the chromosome that is stuck between medians for green HETD and blue NEUT, when it should definitively belong to one or the other. Staring at such a diagram, it is easy to suggest new mu params, which can be put back into the segmentation algorithm once again and visualized as follows. newmu_param <- longseg_param newmu_param$mu <- c(-0.5, -0.4, -0.15, 0.1, 0.4, 0.7) newmu_segments <- HMMsegment(tumour_copy, newmu_param, verbose = FALSE) par(cex.main = 0.5, cex.lab = 0.5, cex.axis = 0.5, mar = c(2, 1.5, 0, 0), mgp = c(1, 0.5, 0)) plotsegments(tumour_copy, newmu_segments, pch = ".", + ylab = "Tumour Copy Number", xlab = "Chromosome Position") 11
12 3.4.1 Understanding parameter convergence The observant reader will noticed nothing has changed. And this was done to purposely highlight that the parameters you set are often just suggestions, and given sufficient flexibility, the algorithm will effectively ignore your suggestions. As seen below in newmu_segments, after several iterations our newly suggested values of mu (first column) effectively converge (in the last column) to the same default values of mu in longseg_param. newmu_segments$mus [,1] [,2] [,3] [,4] [,5] [,6] [1,] [2,] [3,] [4,] [5,] [6,] [,7] [,8] [1,] [2,] [3,] [4,] [5,] [6,] longseg_param$mu [1] Overriding parameter convergence The solution is to simply disallow the algorithm from making large shifts to mu, which we can achieve by setting the prior mean of n (i.e. m) to values identical to mu. par(cex.main = 0.5, cex.lab = 0.5, cex.axis = 0.5, mar = c(2, 1.5, 0, 0), mgp = c(1, 0.5, 0)) newmu_param$m <- newmu_param$mu realmu_segments <- HMMsegment(tumour_copy, newmu_param, verbose = FALSE) plotsegments(tumour_copy, realmu_segments, pch = ".", + ylab = "Tumour Copy Number", xlab = "Chromosome Position") 12
13 4 Matched Tumour-Normal Sample Correction So far, our entire process has been done on single samples, providing respectable results and aesthetically pleasing profiles. However, when give matched tumour and normal pairs, we can improve our copy number results in two ways: 1. We can divide our tumour copy number profile by normal copy number profile to eliminate any germline copy number variation (CNV) events present in both normal and tumour profiles. This will leave only somatic copy number aberration (CNA) events in the tumour. 2. A dramatic reduction in noise is also observed, furthur improving copy number profiles. 4.1 Normalizing tumour by normal copy number profiles The normalization is a simple division of tumour copy number by normal copy number in a bin-wise fashion as follows, or a subtracting of the values in log space as follows: somatic_copy <- tumour_copy # LOGARITHM IDENTITY: log(a) - log(b) == lob(a / b) somatic_copy$copy <- tumour_copy$copy - normal_copy$copy We can then do the same segmentation and visualization as follows: somatic_segments <- HMMsegment(somatic_copy, newmu_param, verbose = FALSE) par(cex.main = 0.5, cex.lab = 0.5, cex.axis = 0.5, mar = c(2, 1.5, 0, 0), mgp = c(1, 0.5, 0)) plotsegments(somatic_copy, somatic_segments, pch = ".", + ylab = "Tumour Copy Number", xlab = "Chromosome Position") The resultant corrected reads can easily be analyzed and exported with other R and Bioconductor packages. 5 Session Information The version number of R and packages loaded for generating the vignette were: ˆ R version Patched ( r74699), x86_64-pc-linux-gnu 13
14 ˆ Locale: LC_CTYPE=en_US.UTF-8, LC_NUMERIC=C, LC_TIME=en_US.UTF-8, LC_COLLATE=C, LC_MONETARY=en_US.UTF-8, LC_MESSAGES=en_US.UTF-8, LC_PAPER=en_US.UTF-8, LC_NAME=C, LC_ADDRESS=C, LC_TELEPHONE=C, LC_MEASUREMENT=en_US.UTF-8, LC_IDENTIFICATION=C ˆ Running under: Ubuntu LTS ˆ Matrix products: default ˆ BLAS: /home/biocbuild/bbs-3.8-bioc/r/lib/librblas.so ˆ LAPACK: /home/biocbuild/bbs-3.8-bioc/r/lib/librlapack.so ˆ Base packages: base, datasets, grdevices, graphics, methods, parallel, stats, stats4, utils ˆ Other packages: AnnotationDbi , Biobase , BiocGenerics , HMMcopy , IRanges , S4Vectors , XML , annotate , geneplotter , lattice ˆ Loaded via a namespace (and not attached): DBI 1.0.0, KernSmooth , RColorBrewer 1.1-2, RCurl , RSQLite 2.1.1, Rcpp , bit , bit , bitops 1.0-6, blob 1.1.1, compiler 3.5.0, digest , grid 3.5.0, memoise 1.1.0, tools 3.5.0, xtable References [1] Yuval Benjamini and Terence P Speed. Summarizing and correcting the gc content bias in highthroughput sequencing. Nucleic Acids Res, 40(10):e72, May [2] Gavin Ha, Andrew Roth, Daniel Lai, Ali Bashashati, Jiarui Ding, Rodrigo Goya, Ryan Giuliany, Jamie Rosner, Arusha Oloumi, Karey Shumansky, Suet-Feung Chin, Gulisa Turashvili, Martin Hirst, Carlos Caldas, Marco A Marra, Samuel Aparicio, and Sohrab P Shah. Integrative analysis of genome-wide loss of heterozygosity and mono-allelic expression at nucleotide resolution reveals disrupted pathways in triple negative breast cancer. Genome Research, (advanced online publication), May [3] S P Shah, A Roth, R Goya, A Oloumi, G Ha, Y Zhao, G Turashvili, J Ding, K Tse, G Haffari, A Bashashati, L M Prentice, J Khattra, A Burleigh, D Yap, V Bernard, A McPherson, K Shumansky, A Crisan, R Giuliany, A Heravi-Moussavi, J Rosner, D Lai, I Birol, R Varhol, A Tam, N Dhalla, T Zeng, K Ma, S K Chan, M Griffith, A Moradian, S W Cheng, G B Morin, P Watson, K Gelmon, S Chia, S F Chin, C Curtis, O M Rueda, P D Pharoah, S Damaraju, J Mackey, K Hoon, T Harkins, V Tadigotla, M Sigaroudinia, P Gascard, T Tlsty, J F Costello, I M Meyer, C J Eaves, W W Wasserman, S Jones, D Huntsman, M Hirst, C Caldas, M A Marra, and S Aparicio. The clonal and mutational evolution spectrum of primary triple-negative breast cancers. Nature, 486(7403): , Jun [4] Sohrab P Shah, Xiang Xuan, Ron J DeLeeuw, Mehrnoush Khojasteh, Wan L Lam, Raymond Ng, and Kevin P Murphy. Integrating copy number polymorphisms into array cgh analysis using a robust hmm. Bioinformatics, 22(14):e431 9, Jul
Package HMMcopy. September 6, 2011
 Package HMMcopy September 6, 2011 Type Package Title Copy number prediction with correction for GC and mappability bias for HTS data Version 0.1.0 Date 2011-09-06 Author Maintainer
Package HMMcopy September 6, 2011 Type Package Title Copy number prediction with correction for GC and mappability bias for HTS data Version 0.1.0 Date 2011-09-06 Author Maintainer
Shrinkage of logarithmic fold changes
 Shrinkage of logarithmic fold changes Michael Love August 9, 2014 1 Comparing the posterior distribution for two genes First, we run a DE analysis on the Bottomly et al. dataset, once with shrunken LFCs
Shrinkage of logarithmic fold changes Michael Love August 9, 2014 1 Comparing the posterior distribution for two genes First, we run a DE analysis on the Bottomly et al. dataset, once with shrunken LFCs
How to Use pkgdeptools
 How to Use pkgdeptools Seth Falcon February 10, 2018 1 Introduction The pkgdeptools package provides tools for computing and analyzing dependency relationships among R packages. With it, you can build
How to Use pkgdeptools Seth Falcon February 10, 2018 1 Introduction The pkgdeptools package provides tools for computing and analyzing dependency relationships among R packages. With it, you can build
Triform: peak finding in ChIP-Seq enrichment profiles for transcription factors
 Triform: peak finding in ChIP-Seq enrichment profiles for transcription factors Karl Kornacker * and Tony Håndstad October 30, 2018 A guide for using the Triform algorithm to predict transcription factor
Triform: peak finding in ChIP-Seq enrichment profiles for transcription factors Karl Kornacker * and Tony Håndstad October 30, 2018 A guide for using the Triform algorithm to predict transcription factor
How to use cghmcr. October 30, 2017
 How to use cghmcr Jianhua Zhang Bin Feng October 30, 2017 1 Overview Copy number data (arraycgh or SNP) can be used to identify genomic regions (Regions Of Interest or ROI) showing gains or losses that
How to use cghmcr Jianhua Zhang Bin Feng October 30, 2017 1 Overview Copy number data (arraycgh or SNP) can be used to identify genomic regions (Regions Of Interest or ROI) showing gains or losses that
Image Analysis with beadarray
 Mike Smith October 30, 2017 Introduction From version 2.0 beadarray provides more flexibility in the processing of array images and the extraction of bead intensities than its predecessor. In the past
Mike Smith October 30, 2017 Introduction From version 2.0 beadarray provides more flexibility in the processing of array images and the extraction of bead intensities than its predecessor. In the past
Count outlier detection using Cook s distance
 Count outlier detection using Cook s distance Michael Love August 9, 2014 1 Run DE analysis with and without outlier removal The following vignette produces the Supplemental Figure of the effect of replacing
Count outlier detection using Cook s distance Michael Love August 9, 2014 1 Run DE analysis with and without outlier removal The following vignette produces the Supplemental Figure of the effect of replacing
Bioconductor: Annotation Package Overview
 Bioconductor: Annotation Package Overview April 30, 2018 1 Overview In its current state the basic purpose of annotate is to supply interface routines that support user actions that rely on the different
Bioconductor: Annotation Package Overview April 30, 2018 1 Overview In its current state the basic purpose of annotate is to supply interface routines that support user actions that rely on the different
How to use CNTools. Overview. Algorithms. Jianhua Zhang. April 14, 2011
 How to use CNTools Jianhua Zhang April 14, 2011 Overview Studies have shown that genomic alterations measured as DNA copy number variations invariably occur across chromosomal regions that span over several
How to use CNTools Jianhua Zhang April 14, 2011 Overview Studies have shown that genomic alterations measured as DNA copy number variations invariably occur across chromosomal regions that span over several
segmentseq: methods for detecting methylation loci and differential methylation
 segmentseq: methods for detecting methylation loci and differential methylation Thomas J. Hardcastle October 30, 2018 1 Introduction This vignette introduces analysis methods for data from high-throughput
segmentseq: methods for detecting methylation loci and differential methylation Thomas J. Hardcastle October 30, 2018 1 Introduction This vignette introduces analysis methods for data from high-throughput
How To Use GOstats and Category to do Hypergeometric testing with unsupported model organisms
 How To Use GOstats and Category to do Hypergeometric testing with unsupported model organisms M. Carlson October 30, 2017 1 Introduction This vignette is meant as an extension of what already exists in
How To Use GOstats and Category to do Hypergeometric testing with unsupported model organisms M. Carlson October 30, 2017 1 Introduction This vignette is meant as an extension of what already exists in
CQN (Conditional Quantile Normalization)
 CQN (Conditional Quantile Normalization) Kasper Daniel Hansen khansen@jhsph.edu Zhijin Wu zhijin_wu@brown.edu Modified: August 8, 2012. Compiled: April 30, 2018 Introduction This package contains the CQN
CQN (Conditional Quantile Normalization) Kasper Daniel Hansen khansen@jhsph.edu Zhijin Wu zhijin_wu@brown.edu Modified: August 8, 2012. Compiled: April 30, 2018 Introduction This package contains the CQN
The analysis of rtpcr data
 The analysis of rtpcr data Jitao David Zhang, Markus Ruschhaupt October 30, 2017 With the help of this document, an analysis of rtpcr data can be performed. For this, the user has to specify several parameters
The analysis of rtpcr data Jitao David Zhang, Markus Ruschhaupt October 30, 2017 With the help of this document, an analysis of rtpcr data can be performed. For this, the user has to specify several parameters
segmentseq: methods for detecting methylation loci and differential methylation
 segmentseq: methods for detecting methylation loci and differential methylation Thomas J. Hardcastle October 13, 2015 1 Introduction This vignette introduces analysis methods for data from high-throughput
segmentseq: methods for detecting methylation loci and differential methylation Thomas J. Hardcastle October 13, 2015 1 Introduction This vignette introduces analysis methods for data from high-throughput
Introduction to the Codelink package
 Introduction to the Codelink package Diego Diez October 30, 2018 1 Introduction This package implements methods to facilitate the preprocessing and analysis of Codelink microarrays. Codelink is a microarray
Introduction to the Codelink package Diego Diez October 30, 2018 1 Introduction This package implements methods to facilitate the preprocessing and analysis of Codelink microarrays. Codelink is a microarray
riboseqr Introduction Getting Data Workflow Example Thomas J. Hardcastle, Betty Y.W. Chung October 30, 2017
 riboseqr Thomas J. Hardcastle, Betty Y.W. Chung October 30, 2017 Introduction Ribosome profiling extracts those parts of a coding sequence currently bound by a ribosome (and thus, are likely to be undergoing
riboseqr Thomas J. Hardcastle, Betty Y.W. Chung October 30, 2017 Introduction Ribosome profiling extracts those parts of a coding sequence currently bound by a ribosome (and thus, are likely to be undergoing
The bumphunter user s guide
 The bumphunter user s guide Kasper Daniel Hansen khansen@jhsph.edu Martin Aryee aryee.martin@mgh.harvard.edu Rafael A. Irizarry rafa@jhu.edu Modified: November 23, 2012. Compiled: April 30, 2018 Introduction
The bumphunter user s guide Kasper Daniel Hansen khansen@jhsph.edu Martin Aryee aryee.martin@mgh.harvard.edu Rafael A. Irizarry rafa@jhu.edu Modified: November 23, 2012. Compiled: April 30, 2018 Introduction
Working with aligned nucleotides (WORK- IN-PROGRESS!)
 Working with aligned nucleotides (WORK- IN-PROGRESS!) Hervé Pagès Last modified: January 2014; Compiled: November 17, 2017 Contents 1 Introduction.............................. 1 2 Load the aligned reads
Working with aligned nucleotides (WORK- IN-PROGRESS!) Hervé Pagès Last modified: January 2014; Compiled: November 17, 2017 Contents 1 Introduction.............................. 1 2 Load the aligned reads
HowTo: Querying online Data
 HowTo: Querying online Data Jeff Gentry and Robert Gentleman November 12, 2017 1 Overview This article demonstrates how you can make use of the tools that have been provided for on-line querying of data
HowTo: Querying online Data Jeff Gentry and Robert Gentleman November 12, 2017 1 Overview This article demonstrates how you can make use of the tools that have been provided for on-line querying of data
Analyse RT PCR data with ddct
 Analyse RT PCR data with ddct Jitao David Zhang, Rudolf Biczok and Markus Ruschhaupt October 30, 2017 Abstract Quantitative real time PCR (qrt PCR or RT PCR for short) is a laboratory technique based on
Analyse RT PCR data with ddct Jitao David Zhang, Rudolf Biczok and Markus Ruschhaupt October 30, 2017 Abstract Quantitative real time PCR (qrt PCR or RT PCR for short) is a laboratory technique based on
MIGSA: Getting pbcmc datasets
 MIGSA: Getting pbcmc datasets Juan C Rodriguez Universidad Católica de Córdoba Universidad Nacional de Córdoba Cristóbal Fresno Instituto Nacional de Medicina Genómica Andrea S Llera Fundación Instituto
MIGSA: Getting pbcmc datasets Juan C Rodriguez Universidad Católica de Córdoba Universidad Nacional de Córdoba Cristóbal Fresno Instituto Nacional de Medicina Genómica Andrea S Llera Fundación Instituto
survsnp: Power and Sample Size Calculations for SNP Association Studies with Censored Time to Event Outcomes
 survsnp: Power and Sample Size Calculations for SNP Association Studies with Censored Time to Event Outcomes Kouros Owzar Zhiguo Li Nancy Cox Sin-Ho Jung Chanhee Yi June 29, 2016 1 Introduction This vignette
survsnp: Power and Sample Size Calculations for SNP Association Studies with Censored Time to Event Outcomes Kouros Owzar Zhiguo Li Nancy Cox Sin-Ho Jung Chanhee Yi June 29, 2016 1 Introduction This vignette
An Overview of the S4Vectors package
 Patrick Aboyoun, Michael Lawrence, Hervé Pagès Edited: February 2018; Compiled: June 7, 2018 Contents 1 Introduction.............................. 1 2 Vector-like and list-like objects...................
Patrick Aboyoun, Michael Lawrence, Hervé Pagès Edited: February 2018; Compiled: June 7, 2018 Contents 1 Introduction.............................. 1 2 Vector-like and list-like objects...................
Advanced analysis using bayseq; generic distribution definitions
 Advanced analysis using bayseq; generic distribution definitions Thomas J Hardcastle October 30, 2017 1 Generic Prior Distributions bayseq now offers complete user-specification of underlying distributions
Advanced analysis using bayseq; generic distribution definitions Thomas J Hardcastle October 30, 2017 1 Generic Prior Distributions bayseq now offers complete user-specification of underlying distributions
Analysis of two-way cell-based assays
 Analysis of two-way cell-based assays Lígia Brás, Michael Boutros and Wolfgang Huber April 16, 2015 Contents 1 Introduction 1 2 Assembling the data 2 2.1 Reading the raw intensity files..................
Analysis of two-way cell-based assays Lígia Brás, Michael Boutros and Wolfgang Huber April 16, 2015 Contents 1 Introduction 1 2 Assembling the data 2 2.1 Reading the raw intensity files..................
An Introduction to ShortRead
 Martin Morgan Modified: 21 October, 2013. Compiled: April 30, 2018 > library("shortread") The ShortRead package provides functionality for working with FASTQ files from high throughput sequence analysis.
Martin Morgan Modified: 21 October, 2013. Compiled: April 30, 2018 > library("shortread") The ShortRead package provides functionality for working with FASTQ files from high throughput sequence analysis.
Visualisation, transformations and arithmetic operations for grouped genomic intervals
 ## Warning: replacing previous import ggplot2::position by BiocGenerics::Position when loading soggi Visualisation, transformations and arithmetic operations for grouped genomic intervals Thomas Carroll
## Warning: replacing previous import ggplot2::position by BiocGenerics::Position when loading soggi Visualisation, transformations and arithmetic operations for grouped genomic intervals Thomas Carroll
Simulation of molecular regulatory networks with graphical models
 Simulation of molecular regulatory networks with graphical models Inma Tur 1 inma.tur@upf.edu Alberto Roverato 2 alberto.roverato@unibo.it Robert Castelo 1 robert.castelo@upf.edu 1 Universitat Pompeu Fabra,
Simulation of molecular regulatory networks with graphical models Inma Tur 1 inma.tur@upf.edu Alberto Roverato 2 alberto.roverato@unibo.it Robert Castelo 1 robert.castelo@upf.edu 1 Universitat Pompeu Fabra,
RAPIDR. Kitty Lo. November 20, Intended use of RAPIDR 1. 2 Create binned counts file from BAMs Masking... 1
 RAPIDR Kitty Lo November 20, 2014 Contents 1 Intended use of RAPIDR 1 2 Create binned counts file from BAMs 1 2.1 Masking.................................................... 1 3 Build the reference 2 3.1
RAPIDR Kitty Lo November 20, 2014 Contents 1 Intended use of RAPIDR 1 2 Create binned counts file from BAMs 1 2.1 Masking.................................................... 1 3 Build the reference 2 3.1
SimBindProfiles: Similar Binding Profiles, identifies common and unique regions in array genome tiling array data
 SimBindProfiles: Similar Binding Profiles, identifies common and unique regions in array genome tiling array data Bettina Fischer October 30, 2018 Contents 1 Introduction 1 2 Reading data and normalisation
SimBindProfiles: Similar Binding Profiles, identifies common and unique regions in array genome tiling array data Bettina Fischer October 30, 2018 Contents 1 Introduction 1 2 Reading data and normalisation
Basic4Cseq: an R/Bioconductor package for the analysis of 4C-seq data
 Basic4Cseq: an R/Bioconductor package for the analysis of 4C-seq data Carolin Walter October 30, 2017 Contents 1 Introduction 1 1.1 Loading the package...................................... 2 1.2 Provided
Basic4Cseq: an R/Bioconductor package for the analysis of 4C-seq data Carolin Walter October 30, 2017 Contents 1 Introduction 1 1.1 Loading the package...................................... 2 1.2 Provided
Creating IGV HTML reports with tracktables
 Thomas Carroll 1 [1em] 1 Bioinformatics Core, MRC Clinical Sciences Centre; thomas.carroll (at)imperial.ac.uk June 13, 2018 Contents 1 The tracktables package.................... 1 2 Creating IGV sessions
Thomas Carroll 1 [1em] 1 Bioinformatics Core, MRC Clinical Sciences Centre; thomas.carroll (at)imperial.ac.uk June 13, 2018 Contents 1 The tracktables package.................... 1 2 Creating IGV sessions
MultipleAlignment Objects
 MultipleAlignment Objects Marc Carlson Bioconductor Core Team Fred Hutchinson Cancer Research Center Seattle, WA April 30, 2018 Contents 1 Introduction 1 2 Creation and masking 1 3 Analytic utilities 7
MultipleAlignment Objects Marc Carlson Bioconductor Core Team Fred Hutchinson Cancer Research Center Seattle, WA April 30, 2018 Contents 1 Introduction 1 2 Creation and masking 1 3 Analytic utilities 7
NacoStringQCPro. Dorothee Nickles, Thomas Sandmann, Robert Ziman, Richard Bourgon. Modified: April 10, Compiled: April 24, 2017.
 NacoStringQCPro Dorothee Nickles, Thomas Sandmann, Robert Ziman, Richard Bourgon Modified: April 10, 2017. Compiled: April 24, 2017 Contents 1 Scope 1 2 Quick start 1 3 RccSet loaded with NanoString data
NacoStringQCPro Dorothee Nickles, Thomas Sandmann, Robert Ziman, Richard Bourgon Modified: April 10, 2017. Compiled: April 24, 2017 Contents 1 Scope 1 2 Quick start 1 3 RccSet loaded with NanoString data
Basic4Cseq: an R/Bioconductor package for the analysis of 4C-seq data
 Basic4Cseq: an R/Bioconductor package for the analysis of 4C-seq data Carolin Walter April 16, 2015 Contents 1 Introduction 1 1.1 Loading the package...................................... 2 1.2 Provided
Basic4Cseq: an R/Bioconductor package for the analysis of 4C-seq data Carolin Walter April 16, 2015 Contents 1 Introduction 1 1.1 Loading the package...................................... 2 1.2 Provided
An introduction to gaucho
 An introduction to gaucho Alex Murison Alexander.Murison@icr.ac.uk Christopher P Wardell Christopher.Wardell@icr.ac.uk October 30, 2017 This vignette serves as an introduction to the R package gaucho.
An introduction to gaucho Alex Murison Alexander.Murison@icr.ac.uk Christopher P Wardell Christopher.Wardell@icr.ac.uk October 30, 2017 This vignette serves as an introduction to the R package gaucho.
Fitting Baselines to Spectra
 Fitting Baselines to Spectra Claudia Beleites DIA Raman Spectroscopy Group, University of Trieste/Italy (2005 2008) Spectroscopy Imaging, IPHT, Jena/Germany (2008 2016) ÖPV, JKI,
Fitting Baselines to Spectra Claudia Beleites DIA Raman Spectroscopy Group, University of Trieste/Italy (2005 2008) Spectroscopy Imaging, IPHT, Jena/Germany (2008 2016) ÖPV, JKI,
Wave correction for arrays
 Wave correction for arrays Eitan Halper-Stromberg and Robert B. Scharpf April 30, 2018 Abstract ArrayTV is a GC correction algorithm for microarray data designed to mitigate the appearance of waves. This
Wave correction for arrays Eitan Halper-Stromberg and Robert B. Scharpf April 30, 2018 Abstract ArrayTV is a GC correction algorithm for microarray data designed to mitigate the appearance of waves. This
Package TitanCNA. June 30, 2018
 Type Package Package TitanCNA June 30, 2018 Title Subclonal copy number and LOH prediction from whole genome sequencing of tumours Version 1.18.0 Date 2017-05-10 Author Gavin Ha, Sohrab P Shah Maintainer
Type Package Package TitanCNA June 30, 2018 Title Subclonal copy number and LOH prediction from whole genome sequencing of tumours Version 1.18.0 Date 2017-05-10 Author Gavin Ha, Sohrab P Shah Maintainer
INSPEcT - INference of Synthesis, Processing and degradation rates in Time-course analysis
 INSPEcT - INference of Synthesis, Processing and degradation rates in Time-course analysis Stefano de Pretis April 7, 20 Contents 1 Introduction 1 2 Quantification of Exon and Intron features 3 3 Time-course
INSPEcT - INference of Synthesis, Processing and degradation rates in Time-course analysis Stefano de Pretis April 7, 20 Contents 1 Introduction 1 2 Quantification of Exon and Intron features 3 3 Time-course
A complete analysis of peptide microarray binding data using the pepstat framework
 A complete analysis of peptide microarray binding data using the pepstat framework Greg Imholte, Renan Sauteraud, Mike Jiang and Raphael Gottardo April 30, 2018 This document present a full analysis, from
A complete analysis of peptide microarray binding data using the pepstat framework Greg Imholte, Renan Sauteraud, Mike Jiang and Raphael Gottardo April 30, 2018 This document present a full analysis, from
MMDiff2: Statistical Testing for ChIP-Seq Data Sets
 MMDiff2: Statistical Testing for ChIP-Seq Data Sets Gabriele Schwikert 1 and David Kuo 2 [1em] 1 The Informatics Forum, University of Edinburgh and The Wellcome
MMDiff2: Statistical Testing for ChIP-Seq Data Sets Gabriele Schwikert 1 and David Kuo 2 [1em] 1 The Informatics Forum, University of Edinburgh and The Wellcome
Introduction to QDNAseq
 Ilari Scheinin October 30, 2017 Contents 1 Running QDNAseq.......................... 1 1.1 Bin annotations.......................... 1 1.2 Processing bam files....................... 2 1.3 Downstream analyses......................
Ilari Scheinin October 30, 2017 Contents 1 Running QDNAseq.......................... 1 1.1 Bin annotations.......................... 1 1.2 Processing bam files....................... 2 1.3 Downstream analyses......................
Creating a New Annotation Package using SQLForge
 Creating a New Annotation Package using SQLForge Marc Carlson, HervÃľ PagÃĺs, Nianhua Li April 30, 2018 1 Introduction The AnnotationForge package provides a series of functions that can be used to build
Creating a New Annotation Package using SQLForge Marc Carlson, HervÃľ PagÃĺs, Nianhua Li April 30, 2018 1 Introduction The AnnotationForge package provides a series of functions that can be used to build
DupChecker: a bioconductor package for checking high-throughput genomic data redundancy in meta-analysis
 DupChecker: a bioconductor package for checking high-throughput genomic data redundancy in meta-analysis Quanhu Sheng, Yu Shyr, Xi Chen Center for Quantitative Sciences, Vanderbilt University, Nashville,
DupChecker: a bioconductor package for checking high-throughput genomic data redundancy in meta-analysis Quanhu Sheng, Yu Shyr, Xi Chen Center for Quantitative Sciences, Vanderbilt University, Nashville,
An Introduction to the methylumi package
 An Introduction to the methylumi package Sean Davis and Sven Bilke October 13, 2014 1 Introduction Gene expression patterns are very important in understanding any biologic system. The regulation of gene
An Introduction to the methylumi package Sean Davis and Sven Bilke October 13, 2014 1 Introduction Gene expression patterns are very important in understanding any biologic system. The regulation of gene
bayseq: Empirical Bayesian analysis of patterns of differential expression in count data
 bayseq: Empirical Bayesian analysis of patterns of differential expression in count data Thomas J. Hardcastle October 30, 2017 1 Introduction This vignette is intended to give a rapid introduction to the
bayseq: Empirical Bayesian analysis of patterns of differential expression in count data Thomas J. Hardcastle October 30, 2017 1 Introduction This vignette is intended to give a rapid introduction to the
Preliminary Figures for Renormalizing Illumina SNP Cell Line Data
 Preliminary Figures for Renormalizing Illumina SNP Cell Line Data Kevin R. Coombes 17 March 2011 Contents 1 Executive Summary 1 1.1 Introduction......................................... 1 1.1.1 Aims/Objectives..................................
Preliminary Figures for Renormalizing Illumina SNP Cell Line Data Kevin R. Coombes 17 March 2011 Contents 1 Executive Summary 1 1.1 Introduction......................................... 1 1.1.1 Aims/Objectives..................................
Creating a New Annotation Package using SQLForge
 Creating a New Annotation Package using SQLForge Marc Carlson, Herve Pages, Nianhua Li November 19, 2013 1 Introduction The AnnotationForge package provides a series of functions that can be used to build
Creating a New Annotation Package using SQLForge Marc Carlson, Herve Pages, Nianhua Li November 19, 2013 1 Introduction The AnnotationForge package provides a series of functions that can be used to build
Overview of ensemblvep
 Overview of ensemblvep Lori Shepherd and Valerie Obenchain October 30, 2017 Contents 1 Introduction 1 2 Results as R objects 1 3 Write results to a file 4 4 Configuring runtime options 5 5 sessioninfo()
Overview of ensemblvep Lori Shepherd and Valerie Obenchain October 30, 2017 Contents 1 Introduction 1 2 Results as R objects 1 3 Write results to a file 4 4 Configuring runtime options 5 5 sessioninfo()
mocluster: Integrative clustering using multiple
 mocluster: Integrative clustering using multiple omics data Chen Meng Modified: June 03, 2015. Compiled: October 30, 2018. Contents 1 mocluster overview......................... 1 2 Run mocluster............................
mocluster: Integrative clustering using multiple omics data Chen Meng Modified: June 03, 2015. Compiled: October 30, 2018. Contents 1 mocluster overview......................... 1 2 Run mocluster............................
RDAVIDWebService: a versatile R interface to DAVID
 RDAVIDWebService: a versatile R interface to DAVID Cristóbal Fresno 1,2 and Elmer A. Fernández 1,2 1 Bio-science Data Mining Group, Catholic University of Córdoba, Córdoba, Argentina. 2 CONICET, Argentina
RDAVIDWebService: a versatile R interface to DAVID Cristóbal Fresno 1,2 and Elmer A. Fernández 1,2 1 Bio-science Data Mining Group, Catholic University of Córdoba, Córdoba, Argentina. 2 CONICET, Argentina
Classification of Breast Cancer Clinical Stage with Gene Expression Data
 Classification of Breast Cancer Clinical Stage with Gene Expression Data Zhu Wang Connecticut Children s Medical Center University of Connecticut School of Medicine zwang@connecticutchildrens.org July
Classification of Breast Cancer Clinical Stage with Gene Expression Data Zhu Wang Connecticut Children s Medical Center University of Connecticut School of Medicine zwang@connecticutchildrens.org July
The rtracklayer package
 The rtracklayer package Michael Lawrence January 22, 2018 Contents 1 Introduction 2 2 Gene expression and microrna target sites 2 2.1 Creating a target site track..................... 2 2.1.1 Constructing
The rtracklayer package Michael Lawrence January 22, 2018 Contents 1 Introduction 2 2 Gene expression and microrna target sites 2 2.1 Creating a target site track..................... 2 2.1.1 Constructing
INSPEcT - INference of Synthesis, Processing and degradation rates in Time-course analysis
 INSPEcT - INference of Synthesis, Processing and degradation rates in Time-course analysis Stefano de Pretis October 30, 2017 Contents 1 Introduction.............................. 1 2 Quantification of
INSPEcT - INference of Synthesis, Processing and degradation rates in Time-course analysis Stefano de Pretis October 30, 2017 Contents 1 Introduction.............................. 1 2 Quantification of
Analysis of screens with enhancer and suppressor controls
 Analysis of screens with enhancer and suppressor controls Lígia Brás, Michael Boutros and Wolfgang Huber April 16, 2015 Contents 1 Introduction 1 2 Assembling the data 2 2.1 Reading the raw intensity files..................
Analysis of screens with enhancer and suppressor controls Lígia Brás, Michael Boutros and Wolfgang Huber April 16, 2015 Contents 1 Introduction 1 2 Assembling the data 2 2.1 Reading the raw intensity files..................
Introduction to the TSSi package: Identification of Transcription Start Sites
 Introduction to the TSSi package: Identification of Transcription Start Sites Julian Gehring, Clemens Kreutz October 30, 2017 Along with the advances in high-throughput sequencing, the detection of transcription
Introduction to the TSSi package: Identification of Transcription Start Sites Julian Gehring, Clemens Kreutz October 30, 2017 Along with the advances in high-throughput sequencing, the detection of transcription
RMassBank for XCMS. Erik Müller. January 4, Introduction 2. 2 Input files LC/MS data Additional Workflow-Methods 2
 RMassBank for XCMS Erik Müller January 4, 2019 Contents 1 Introduction 2 2 Input files 2 2.1 LC/MS data........................... 2 3 Additional Workflow-Methods 2 3.1 Options..............................
RMassBank for XCMS Erik Müller January 4, 2019 Contents 1 Introduction 2 2 Input files 2 2.1 LC/MS data........................... 2 3 Additional Workflow-Methods 2 3.1 Options..............................
Overview of ensemblvep
 Overview of ensemblvep Lori Shepherd and Valerie Obenchain October 30, 2018 Contents 1 Introduction 1 2 Results as R objects 1 3 Write results to a file 4 4 Configuring runtime options 5 5 sessioninfo()
Overview of ensemblvep Lori Shepherd and Valerie Obenchain October 30, 2018 Contents 1 Introduction 1 2 Results as R objects 1 3 Write results to a file 4 4 Configuring runtime options 5 5 sessioninfo()
SIBER User Manual. Pan Tong and Kevin R Coombes. May 27, Introduction 1
 SIBER User Manual Pan Tong and Kevin R Coombes May 27, 2015 Contents 1 Introduction 1 2 Using SIBER 1 2.1 A Quick Example........................................... 1 2.2 Dealing With RNAseq Normalization................................
SIBER User Manual Pan Tong and Kevin R Coombes May 27, 2015 Contents 1 Introduction 1 2 Using SIBER 1 2.1 A Quick Example........................................... 1 2.2 Dealing With RNAseq Normalization................................
Introduction: microarray quality assessment with arrayqualitymetrics
 Introduction: microarray quality assessment with arrayqualitymetrics Audrey Kauffmann, Wolfgang Huber April 4, 2013 Contents 1 Basic use 3 1.1 Affymetrix data - before preprocessing.......................
Introduction: microarray quality assessment with arrayqualitymetrics Audrey Kauffmann, Wolfgang Huber April 4, 2013 Contents 1 Basic use 3 1.1 Affymetrix data - before preprocessing.......................
highscreen: High-Throughput Screening for Plate Based Assays
 highscreen: High-Throughput Screening for Plate Based Assays Ivo D. Shterev Cliburn Chan Gregory D. Sempowski August 22, 2018 1 Introduction This vignette describes the use of the R extension package highscreen
highscreen: High-Throughput Screening for Plate Based Assays Ivo D. Shterev Cliburn Chan Gregory D. Sempowski August 22, 2018 1 Introduction This vignette describes the use of the R extension package highscreen
An Introduction to the methylumi package
 An Introduction to the methylumi package Sean Davis and Sven Bilke April 30, 2018 1 Introduction Gene expression patterns are very important in understanding any biologic system. The regulation of gene
An Introduction to the methylumi package Sean Davis and Sven Bilke April 30, 2018 1 Introduction Gene expression patterns are very important in understanding any biologic system. The regulation of gene
Cardinal design and development
 Kylie A. Bemis October 30, 2017 Contents 1 Introduction.............................. 2 2 Design overview........................... 2 3 iset: high-throughput imaging experiments............ 3 3.1 SImageSet:
Kylie A. Bemis October 30, 2017 Contents 1 Introduction.............................. 2 2 Design overview........................... 2 3 iset: high-throughput imaging experiments............ 3 3.1 SImageSet:
PROPER: PROspective Power Evaluation for RNAseq
 PROPER: PROspective Power Evaluation for RNAseq Hao Wu [1em]Department of Biostatistics and Bioinformatics Emory University Atlanta, GA 303022 [1em] hao.wu@emory.edu October 30, 2017 Contents 1 Introduction..............................
PROPER: PROspective Power Evaluation for RNAseq Hao Wu [1em]Department of Biostatistics and Bioinformatics Emory University Atlanta, GA 303022 [1em] hao.wu@emory.edu October 30, 2017 Contents 1 Introduction..............................
Quality control of array genotyping data with argyle Andrew P Morgan
 Quality control of array genotyping data with argyle Andrew P Morgan 2015-10-08 Introduction Proper quality control of array genotypes is an important prerequisite to further analysis. Genotype quality
Quality control of array genotyping data with argyle Andrew P Morgan 2015-10-08 Introduction Proper quality control of array genotypes is an important prerequisite to further analysis. Genotype quality
Introduction to GenomicFiles
 Valerie Obenchain, Michael Love, Martin Morgan Last modified: October 2014; Compiled: October 30, 2018 Contents 1 Introduction.............................. 1 2 Quick Start..............................
Valerie Obenchain, Michael Love, Martin Morgan Last modified: October 2014; Compiled: October 30, 2018 Contents 1 Introduction.............................. 1 2 Quick Start..............................
Raman Spectra of Chondrocytes in Cartilage: hyperspec s chondro data set
 Raman Spectra of Chondrocytes in Cartilage: hyperspec s chondro data set Claudia Beleites CENMAT and DI3, University of Trieste Spectroscopy Imaging, IPHT Jena e.v. February 13,
Raman Spectra of Chondrocytes in Cartilage: hyperspec s chondro data set Claudia Beleites CENMAT and DI3, University of Trieste Spectroscopy Imaging, IPHT Jena e.v. February 13,
Introduction: microarray quality assessment with arrayqualitymetrics
 : microarray quality assessment with arrayqualitymetrics Audrey Kauffmann, Wolfgang Huber October 30, 2017 Contents 1 Basic use............................... 3 1.1 Affymetrix data - before preprocessing.............
: microarray quality assessment with arrayqualitymetrics Audrey Kauffmann, Wolfgang Huber October 30, 2017 Contents 1 Basic use............................... 3 1.1 Affymetrix data - before preprocessing.............
Getting started with flowstats
 Getting started with flowstats F. Hahne, N. Gopalakrishnan October, 7 Abstract flowstats is a collection of algorithms for the statistical analysis of flow cytometry data. So far, the focus is on automated
Getting started with flowstats F. Hahne, N. Gopalakrishnan October, 7 Abstract flowstats is a collection of algorithms for the statistical analysis of flow cytometry data. So far, the focus is on automated
Analysis of Pairwise Interaction Screens
 Analysis of Pairwise Interaction Screens Bernd Fischer April 11, 2014 Contents 1 Introduction 1 2 Installation of the RNAinteract package 1 3 Creating an RNAinteract object 2 4 Single Perturbation effects
Analysis of Pairwise Interaction Screens Bernd Fischer April 11, 2014 Contents 1 Introduction 1 2 Installation of the RNAinteract package 1 3 Creating an RNAinteract object 2 4 Single Perturbation effects
ssviz: A small RNA-seq visualizer and analysis toolkit
 ssviz: A small RNA-seq visualizer and analysis toolkit Diana HP Low Institute of Molecular and Cell Biology Agency for Science, Technology and Research (A*STAR), Singapore dlow@imcb.a-star.edu.sg August
ssviz: A small RNA-seq visualizer and analysis toolkit Diana HP Low Institute of Molecular and Cell Biology Agency for Science, Technology and Research (A*STAR), Singapore dlow@imcb.a-star.edu.sg August
An Introduction to Bioconductor s ExpressionSet Class
 An Introduction to Bioconductor s ExpressionSet Class Seth Falcon, Martin Morgan, and Robert Gentleman 6 October, 2006; revised 9 February, 2007 1 Introduction Biobase is part of the Bioconductor project,
An Introduction to Bioconductor s ExpressionSet Class Seth Falcon, Martin Morgan, and Robert Gentleman 6 October, 2006; revised 9 February, 2007 1 Introduction Biobase is part of the Bioconductor project,
Using crlmm for copy number estimation and genotype calling with Illumina platforms
 Using crlmm for copy number estimation and genotype calling with Illumina platforms Rob Scharpf November, Abstract This vignette illustrates the steps necessary for obtaining marker-level estimates of
Using crlmm for copy number estimation and genotype calling with Illumina platforms Rob Scharpf November, Abstract This vignette illustrates the steps necessary for obtaining marker-level estimates of
An Introduction to GenomeInfoDb
 Martin Morgan, Hervé Pagès, Marc Carlson, Sonali Arora Modified: 17 January, 2014. Compiled: January 21, 2018 Contents 1 Introduction.............................. 2 2 Functionality for all existing organisms...............
Martin Morgan, Hervé Pagès, Marc Carlson, Sonali Arora Modified: 17 January, 2014. Compiled: January 21, 2018 Contents 1 Introduction.............................. 2 2 Functionality for all existing organisms...............
Evaluation of VST algorithm in lumi package
 Evaluation of VST algorithm in lumi package Pan Du 1, Simon Lin 1, Wolfgang Huber 2, Warrren A. Kibbe 1 December 22, 2010 1 Robert H. Lurie Comprehensive Cancer Center Northwestern University, Chicago,
Evaluation of VST algorithm in lumi package Pan Du 1, Simon Lin 1, Wolfgang Huber 2, Warrren A. Kibbe 1 December 22, 2010 1 Robert H. Lurie Comprehensive Cancer Center Northwestern University, Chicago,
ExomeDepth. Vincent Plagnol. May 15, What is new? 1. 4 Load an example dataset 4. 6 CNV calling 5. 9 Visual display 9
 ExomeDepth Vincent Plagnol May 15, 2016 Contents 1 What is new? 1 2 What ExomeDepth does and tips for QC 2 2.1 What ExomeDepth does and does not do................................. 2 2.2 Useful quality
ExomeDepth Vincent Plagnol May 15, 2016 Contents 1 What is new? 1 2 What ExomeDepth does and tips for QC 2 2.1 What ExomeDepth does and does not do................................. 2 2.2 Useful quality
The REDseq user s guide
 The REDseq user s guide Lihua Julie Zhu * October 30, 2017 Contents 1 Introduction 1 2 Examples of using REDseq 2 2.1 Task 1: Build a RE map for a genome..................... 2 2.2 Task 2: Assign mapped
The REDseq user s guide Lihua Julie Zhu * October 30, 2017 Contents 1 Introduction 1 2 Examples of using REDseq 2 2.1 Task 1: Build a RE map for a genome..................... 2 2.2 Task 2: Assign mapped
Seminar III: R/Bioconductor
 Leonardo Collado Torres lcollado@lcg.unam.mx Bachelor in Genomic Sciences www.lcg.unam.mx/~lcollado/ August - December, 2009 1 / 25 Class outline Working with HTS data: a simulated case study Intro R for
Leonardo Collado Torres lcollado@lcg.unam.mx Bachelor in Genomic Sciences www.lcg.unam.mx/~lcollado/ August - December, 2009 1 / 25 Class outline Working with HTS data: a simulated case study Intro R for
Using the qrqc package to gather information about sequence qualities
 Using the qrqc package to gather information about sequence qualities Vince Buffalo Bioinformatics Core UC Davis Genome Center vsbuffalo@ucdavis.edu 2012-02-19 Abstract Many projects in bioinformatics
Using the qrqc package to gather information about sequence qualities Vince Buffalo Bioinformatics Core UC Davis Genome Center vsbuffalo@ucdavis.edu 2012-02-19 Abstract Many projects in bioinformatics
Package copynumber. November 21, 2017
 Package copynumber November 21, 2017 Title Segmentation of single- and multi-track copy number data by penalized least squares regression. Version 1.19.0 Author Gro Nilsen, Knut Liestoel and Ole Christian
Package copynumber November 21, 2017 Title Segmentation of single- and multi-track copy number data by penalized least squares regression. Version 1.19.0 Author Gro Nilsen, Knut Liestoel and Ole Christian
ISA internals. October 18, Speeding up the ISA iteration 2
 ISA internals Gábor Csárdi October 18, 2011 Contents 1 Introduction 1 2 Why two packages? 1 3 Speeding up the ISA iteration 2 4 Running time analysis 3 4.1 The hardware and software.....................
ISA internals Gábor Csárdi October 18, 2011 Contents 1 Introduction 1 2 Why two packages? 1 3 Speeding up the ISA iteration 2 4 Running time analysis 3 4.1 The hardware and software.....................
GxE.scan. October 30, 2018
 GxE.scan October 30, 2018 Overview GxE.scan can process a GWAS scan using the snp.logistic, additive.test, snp.score or snp.matched functions, whereas snp.scan.logistic only calls snp.logistic. GxE.scan
GxE.scan October 30, 2018 Overview GxE.scan can process a GWAS scan using the snp.logistic, additive.test, snp.score or snp.matched functions, whereas snp.scan.logistic only calls snp.logistic. GxE.scan
Introduction to BatchtoolsParam
 Nitesh Turaga 1, Martin Morgan 2 Edited: March 22, 2018; Compiled: January 4, 2019 1 Nitesh.Turaga@ RoswellPark.org 2 Martin.Morgan@ RoswellPark.org Contents 1 Introduction..............................
Nitesh Turaga 1, Martin Morgan 2 Edited: March 22, 2018; Compiled: January 4, 2019 1 Nitesh.Turaga@ RoswellPark.org 2 Martin.Morgan@ RoswellPark.org Contents 1 Introduction..............................
genefu: a package for breast cancer gene expression analysis
 genefu: a package for breast cancer gene expression analysis Benjamin Haibe-Kains 1, Markus Schröder 2, Christos Sotiriou 3, Gianluca Bontempi 4, and John Quackenbush 5,6 1 Bioinformatics and Computational
genefu: a package for breast cancer gene expression analysis Benjamin Haibe-Kains 1, Markus Schröder 2, Christos Sotiriou 3, Gianluca Bontempi 4, and John Quackenbush 5,6 1 Bioinformatics and Computational
SCnorm: robust normalization of single-cell RNA-seq data
 SCnorm: robust normalization of single-cell RNA-seq data Rhonda Bacher and Christina Kendziorski February 18, 2019 Contents 1 Introduction.............................. 1 2 Run SCnorm.............................
SCnorm: robust normalization of single-cell RNA-seq data Rhonda Bacher and Christina Kendziorski February 18, 2019 Contents 1 Introduction.............................. 1 2 Run SCnorm.............................
Package icnv. R topics documented: March 8, Title Integrated Copy Number Variation detection Version Author Zilu Zhou, Nancy Zhang
 Title Integrated Copy Number Variation detection Version 1.2.1 Author Zilu Zhou, Nancy Zhang Package icnv March 8, 2019 Maintainer Zilu Zhou Integrative copy number variation
Title Integrated Copy Number Variation detection Version 1.2.1 Author Zilu Zhou, Nancy Zhang Package icnv March 8, 2019 Maintainer Zilu Zhou Integrative copy number variation
RPHAST: detecting GC-biased gene conversion
 RPHAST: detecting GC-biased gene conversion M. J. Hubisz, K. S. Pollard, and A. Siepel January 30, 2018 1 Introduction This vignette describes some of the basic functions available for detecting GC-biased
RPHAST: detecting GC-biased gene conversion M. J. Hubisz, K. S. Pollard, and A. Siepel January 30, 2018 1 Introduction This vignette describes some of the basic functions available for detecting GC-biased
Getting Started DECIPHERing
 Getting Started DECIPHERing Erik S. Wright April 30, 2018 Contents 1 About DECIPHER 1 2 Design Philosophy 2 2.1 Curators Protect the Originals................................... 2 2.2 Don t Reinvent the
Getting Started DECIPHERing Erik S. Wright April 30, 2018 Contents 1 About DECIPHER 1 2 Design Philosophy 2 2.1 Curators Protect the Originals................................... 2 2.2 Don t Reinvent the
The software comes with 2 installers: (1) SureCall installer (2) GenAligners (contains BWA, BWA- MEM).
 Release Notes Agilent SureCall 4.0 Product Number G4980AA SureCall Client 6-month named license supports installation of one client and server (to host the SureCall database) on one machine. For additional
Release Notes Agilent SureCall 4.0 Product Number G4980AA SureCall Client 6-month named license supports installation of one client and server (to host the SureCall database) on one machine. For additional
How to use MeSH-related Packages
 How to use MeSH-related Packages Koki Tsuyuzaki 1,4, Gota Morota 2, Takeru Nakazato 3 and Itoshi Nikaido 4. October 30, 2017 1 Department of Medical and Life Science, Tokyo University of Science. 2 Department
How to use MeSH-related Packages Koki Tsuyuzaki 1,4, Gota Morota 2, Takeru Nakazato 3 and Itoshi Nikaido 4. October 30, 2017 1 Department of Medical and Life Science, Tokyo University of Science. 2 Department
rhdf5 - HDF5 interface for R
 Bernd Fischer October 30, 2017 Contents 1 Introduction 1 2 Installation of the HDF5 package 2 3 High level R -HDF5 functions 2 31 Creating an HDF5 file and group hierarchy 2 32 Writing and reading objects
Bernd Fischer October 30, 2017 Contents 1 Introduction 1 2 Installation of the HDF5 package 2 3 High level R -HDF5 functions 2 31 Creating an HDF5 file and group hierarchy 2 32 Writing and reading objects
Some Basic ChIP-Seq Data Analysis
 Some Basic ChIP-Seq Data Analysis July 28, 2009 Our goal is to describe the use of Bioconductor software to perform some basic tasks in the analysis of ChIP-Seq data. We will use several functions in the
Some Basic ChIP-Seq Data Analysis July 28, 2009 Our goal is to describe the use of Bioconductor software to perform some basic tasks in the analysis of ChIP-Seq data. We will use several functions in the
M 3 D: Statistical Testing for RRBS Data Sets
 Tom Mayo Modified: 5 September, 2016. Compiled: October 30, 2018 Contents 1 Introduction 1 2 Analysis Pipeline 2 2.1 Reading in Data. 2 2.2 Computing the MMD, and coverage-mmd. 5 2.3 Generating p-values..
Tom Mayo Modified: 5 September, 2016. Compiled: October 30, 2018 Contents 1 Introduction 1 2 Analysis Pipeline 2 2.1 Reading in Data. 2 2.2 Computing the MMD, and coverage-mmd. 5 2.3 Generating p-values..
Flexible Logo plots of symbols and alphanumeric strings using Logolas
 Flexible Logo plots of symbols and alphanumeric strings using Logolas Kushal K Dey Stephens Lab, Dept. of Statistics, The University of Chicago Correspondending Email: kkdey@uchicago.edu April 24, 2017
Flexible Logo plots of symbols and alphanumeric strings using Logolas Kushal K Dey Stephens Lab, Dept. of Statistics, The University of Chicago Correspondending Email: kkdey@uchicago.edu April 24, 2017
Analyzing Genomic Data with NOJAH
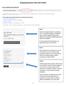 Analyzing Genomic Data with NOJAH TAB A) GENOME WIDE ANALYSIS Step 1: Select the example dataset or upload your own. Two example datasets are available. Genome-Wide TCGA-BRCA Expression datasets and CoMMpass
Analyzing Genomic Data with NOJAH TAB A) GENOME WIDE ANALYSIS Step 1: Select the example dataset or upload your own. Two example datasets are available. Genome-Wide TCGA-BRCA Expression datasets and CoMMpass
Flexible Logo plots of symbols and alphanumeric strings using Logolas
 Flexible Logo plots of symbols and alphanumeric strings using Logolas Kushal K Dey [1em] Stephens Lab, Dept. of Statistics, The University of Chicago Correspondending Email: kkdey@uchicago.edu Abstract
Flexible Logo plots of symbols and alphanumeric strings using Logolas Kushal K Dey [1em] Stephens Lab, Dept. of Statistics, The University of Chicago Correspondending Email: kkdey@uchicago.edu Abstract
R topics documented: 2 R topics documented:
 Package cn.mops March 20, 2019 Maintainer Guenter Klambauer Author Guenter Klambauer License LGPL (>= 2.0) Type Package Title cn.mops - Mixture of Poissons for CNV detection in
Package cn.mops March 20, 2019 Maintainer Guenter Klambauer Author Guenter Klambauer License LGPL (>= 2.0) Type Package Title cn.mops - Mixture of Poissons for CNV detection in
Package conumee. February 17, 2018
 Package conumee February 17, 2018 Title Enhanced copy-number variation analysis using Illumina DNA methylation arrays Version 1.13.0 Author Volker Hovestadt, Marc Zapatka Maintainer Address Division of
Package conumee February 17, 2018 Title Enhanced copy-number variation analysis using Illumina DNA methylation arrays Version 1.13.0 Author Volker Hovestadt, Marc Zapatka Maintainer Address Division of
Biobase development and the new eset
 Biobase development and the new eset Martin T. Morgan * 7 August, 2006 Revised 4 September, 2006 featuredata slot. Revised 20 April 2007 minor wording changes; verbose and other arguments passed through
Biobase development and the new eset Martin T. Morgan * 7 August, 2006 Revised 4 September, 2006 featuredata slot. Revised 20 April 2007 minor wording changes; verbose and other arguments passed through
