Research Team Personnel
|
|
|
- Griffin Dennis
- 5 years ago
- Views:
Transcription
1 Research Team Personnel Individuals involved in the following activities should be named on the protocol application: Recruitment Obtaining consent Collecting data Analyzing data If there is uncertainty as to the role of a person and whether or not the person needs to be identified/named on the protocol application, please contact the Office of Research Compliance for guidance. Uncc irb@uncc.edu or or There are different procedures for adding individuals affiliated with UNC Charlotte and adding individuals not affiliated with UNC Charlotte. Project Personnel are added in the General Information section of the protocol application. Personnel are added one by one and a role assigned to each individual. Choose Click here to add personnel link. The Investigator must add him/herself to the protocol applicatin. 1
2 This will result in a personnel table. This initial step is followed for all personnel, including those not affiliated with UNC Charlotte. NOTE: DO NOT MANUALLY ADD INDIVIDUAL INFORMATION TO THE TABLE FROM THIS SCREEN 2
3 For individuals affiliated with UNC Charlotte: Click the magnifying glass icon or put the cursor in the text box to activate the search option to search UNC Charlotte directories (data are populated daily from Banner). Search by Last Name or PID if this number is known to you. Choose the correct UNC Charlotte individual from the resulting search results. 3
4 The person s information will automatically populate the initial personnel table. Choose the appropriate Role for the person and then Save the personnel record to complete the procedure for adding this person to the protocol application. As you add personnel, you will need to designate a Liaison. This person serves as the point of contact for questions related to the study or study site for multi site studies. This may be someone other than the Principal Investigator. 4
5 For individuals NOT affiliated with UNC Charlotte: Independent Investigators Persons who will collaborate with limited involvement in the study, the study is not a multi site study, and/or the person is involved in the research in an unaffiliated capacity (not an institutional capacity) may be may be named on the protocol as Independent Investigators. Click the magnifying glass icon or put the cursor in the text box to activate the search option to search UNC Charlotte directories (data are populated daily from Banner). Click the Person Not Listed link to generate the appropriate personnel table to manually add unaffiliated persons. 5
6 Manually enter the person s name and contact information. The person s affiliation may also be manually entered in the Department Name field and NOT in the External Institution field (i.e., leave the External Institution field empty/blank). Click the Submit button. 6
7 The manually entered information will populate the initial personnel screen. Confirm that the Independent Investigator box is checked and the person s Role on the research team is identified. Save the personnel listing. The saved personnel listing will appear in the protocol as an Independent Investigator. 7
8 For individuals NOT affiliated with UNC Charlotte: External Institutions ONLY add External Institutions if this is a multi site study and the external institutions will rely on the UNC Charlotte IRB for review and oversight of the study at all institutions (multi site). ONLY use the External Institutions search (magnifying glass) to add a collaborator if UNC Charlotte will enter into an IRB Authorization Agreement/Reliance Agreement with the collaborator s IRB. If there is any question as to applicability of such an agreement Office of Research Compliance for guidance. Uncc irb@uncc.edu or or NOTE: If an External Institution will be named on the protocol, the Screening Question #4 must be a YES response. IRBIS will prompt you to update this Screening Question if you attempt to add personnel as using External Institutions. 8
9 You may manually add persons. Use the External Institution search (magnifying glass) to add a collaborator IF UNC Charlotte will enter into an IRB Authorization Agreement/Reliance Agreement with the collaborator s IRB. This may be applicable if this is a multi site study. The External Institution Listing search will activate so that you may search for the collaborating institution. 9
10 Select the appropriate institution to populate the External Institution field. The available Role for the unaffiliated person is limited to External Site PI. Complete the remainder of the table and Submit. The manually added External Institution and PI will populate the initial personnel screen. A Liaison for the External Institution must be designated. Confirm that the Liaison item is completed. Save the personnel listing. 10
Buck-IRB Amendment User Guide
 Office of Research Buck-IRB Amendment User Guide Office of Responsible Research Practices 1960 Kenny Road, Columbus, OH 43210-1016 Institutional Review Board General Guidance for Amendments: The Start
Office of Research Buck-IRB Amendment User Guide Office of Responsible Research Practices 1960 Kenny Road, Columbus, OH 43210-1016 Institutional Review Board General Guidance for Amendments: The Start
IRBManager Quick Start Guide AMENDMENT SUBMISSION - CHANGE IN PERSONNEL
 Page 1 of 12 IRBManager Quick Start Guide AMENDMENT SUBMISSION - CHANGE IN PERSONNEL NOTE: This Quick Start Guide provides instructions for removing and/or adding personnel, other than the Principal Investigator,
Page 1 of 12 IRBManager Quick Start Guide AMENDMENT SUBMISSION - CHANGE IN PERSONNEL NOTE: This Quick Start Guide provides instructions for removing and/or adding personnel, other than the Principal Investigator,
Cayuse IRB. Submitting a New Protocol. Office of Research Compliance (657)
 Cayuse IRB Submitting a New Protocol Office of Research Compliance irb@fullerton.edu (657) 278-7719 How to use this tutorial This tutorial is written from the point of view of a Primary Investigator (PI),
Cayuse IRB Submitting a New Protocol Office of Research Compliance irb@fullerton.edu (657) 278-7719 How to use this tutorial This tutorial is written from the point of view of a Primary Investigator (PI),
Online Reliance System FAQs
 Online Reliance System FAQs Table of Contents Online Reliance System FAQs... 1 When should the Online Reliance System be used?...3 Who can use the Online Reliance System?...3 Do I have to use the Online
Online Reliance System FAQs Table of Contents Online Reliance System FAQs... 1 When should the Online Reliance System be used?...3 Who can use the Online Reliance System?...3 Do I have to use the Online
Mentor eirb Researcher User Manual
 Ascension Wisconsin IRB Mentor eirb Researcher User Manual Contents 1 Introduction 1.1 Purpose of this Manual 1.2 User Support 2 Mentor eirb & User Accounts 2.1 About Mentor 2.2 User Roles 2.3 Requesting
Ascension Wisconsin IRB Mentor eirb Researcher User Manual Contents 1 Introduction 1.1 Purpose of this Manual 1.2 User Support 2 Mentor eirb & User Accounts 2.1 About Mentor 2.2 User Roles 2.3 Requesting
Creating a distribution group
 Creating a distribution group You can group a number of selected contacts together, for example all of your team. This is known as a distribution group or list. Using a distribution group, you can email
Creating a distribution group You can group a number of selected contacts together, for example all of your team. This is known as a distribution group or list. Using a distribution group, you can email
STUDY ASSISTANT. Study Assistant. Version 10.03
 STUDY ASSISTANT Study Assistant Version 10.03 Contents Introduction... 3 Add a New Study... 3 My Studies... 3 Find a Study... 9 Find a Study for All Users... 12 imedris Data Corporation 2 Study Assistant
STUDY ASSISTANT Study Assistant Version 10.03 Contents Introduction... 3 Add a New Study... 3 My Studies... 3 Find a Study... 9 Find a Study for All Users... 12 imedris Data Corporation 2 Study Assistant
UNIVERSITY OF NORTH FLORIDA. Institutional Review Board (IRB) Guide to IRBNet:
 UNIVERSITY OF NORTH FLORIDA Institutional Review Board (IRB) Guide to IRBNet: Version 2 11-15-2013 Table of Contents i Introduction 2 IRBNet User Registration 2 IRBNet ID Number 2 Training and Credentials
UNIVERSITY OF NORTH FLORIDA Institutional Review Board (IRB) Guide to IRBNet: Version 2 11-15-2013 Table of Contents i Introduction 2 IRBNet User Registration 2 IRBNet ID Number 2 Training and Credentials
SUBMITTING A NEW IBC PROJECT APPLICATION (INITIAL)
 SUBMITTING A After you have established and activated your username and password, you can begin to create an IBC application in IRBNet. To start building a new IBC application, you must first CREATE A
SUBMITTING A After you have established and activated your username and password, you can begin to create an IBC application in IRBNet. To start building a new IBC application, you must first CREATE A
PCORI Online: Awardee User Guide Research Awards
 PCORI Online: Awardee User Guide Research Awards Updated as of 1/31/18 1 Table of Contents Section 1: Introduction to PCORI Online... 3 1.1 Getting Started - Tips for Using PCORI Online... 4 1.2 Logging
PCORI Online: Awardee User Guide Research Awards Updated as of 1/31/18 1 Table of Contents Section 1: Introduction to PCORI Online... 3 1.1 Getting Started - Tips for Using PCORI Online... 4 1.2 Logging
New Protocol - NoN-humaN subjects research training guide
 New Protocol - NoN-humaN subjects research training guide 2015, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Non-Human Subjects Research 1 table of contents Table of Contents
New Protocol - NoN-humaN subjects research training guide 2015, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Non-Human Subjects Research 1 table of contents Table of Contents
COEUS LITE IRB COEUS 4.5.1_P3 USER GUIDE
 COEUS LITE IRB COEUS 4.5.1_P3 USER GUIDE Version: August 24, 2016 1 TABLE OF CONTENTS A note about this guide... 4 Getting access to Coeus Lite... 4 Preparing to enter a protocol... 4 Getting Started:
COEUS LITE IRB COEUS 4.5.1_P3 USER GUIDE Version: August 24, 2016 1 TABLE OF CONTENTS A note about this guide... 4 Getting access to Coeus Lite... 4 Preparing to enter a protocol... 4 Getting Started:
The Center for Translational Medicine at UT Southwestern Medical Center. The Research Launchpad: Your One-Stop Shop to CTSA Resources and Services
 The Center for Translational Medicine at UT Southwestern Medical Center The Research Launchpad: Your One-Stop Shop to CTSA Resources and Services Users Guide Table of Contents a. Introduction b. Where
The Center for Translational Medicine at UT Southwestern Medical Center The Research Launchpad: Your One-Stop Shop to CTSA Resources and Services Users Guide Table of Contents a. Introduction b. Where
Institutional Review Board (IRB)
 IRBNet User s Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org If you have any questions regarding the submission of an IRB protocol contact:
IRBNet User s Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org If you have any questions regarding the submission of an IRB protocol contact:
IRBManager Faculty Adviser Role
 IRBManager Faculty Adviser Role Human Subjects Research Institutional Review Board irb@okstate.edu 405-744-3377 Page 1 Table of Contents ABOUT... 3 NOTIFICATION OF REVIEW... 3 LOGGING INTO YOUR IRBMANAGER
IRBManager Faculty Adviser Role Human Subjects Research Institutional Review Board irb@okstate.edu 405-744-3377 Page 1 Table of Contents ABOUT... 3 NOTIFICATION OF REVIEW... 3 LOGGING INTO YOUR IRBMANAGER
the address book training guide
 the address book training guide 2015, The Trustees of Indiana University 1 table of Contents Table of Contents 2 table of Contents 3 about this document 4 how to access the address book 5 address book
the address book training guide 2015, The Trustees of Indiana University 1 table of Contents Table of Contents 2 table of Contents 3 about this document 4 how to access the address book 5 address book
IRBManager Quick Start Guide INITIAL APPLICATION - OVERVIEW
 Page 1 of 16 GENERAL INFORMATION IRBManager Quick Start Guide INITIAL APPLICATION - OVERVIEW Initial Application Types: The IRBManager initial application form (xform) is available for specific types of
Page 1 of 16 GENERAL INFORMATION IRBManager Quick Start Guide INITIAL APPLICATION - OVERVIEW Initial Application Types: The IRBManager initial application form (xform) is available for specific types of
Princeton University COLLABORATIVE INSTITUTIONAL TRAINING INITIATIVE (CITI) RESEARCH. Institutional Review Board
 RESEARCH Research Integrity and Assurance Princeton University Institutional Review Board COLLABORATIVE INSTITUTIONAL TRAINING INITIATIVE (CITI) Human Subjects Training Instructions for CITI Research Integrity
RESEARCH Research Integrity and Assurance Princeton University Institutional Review Board COLLABORATIVE INSTITUTIONAL TRAINING INITIATIVE (CITI) Human Subjects Training Instructions for CITI Research Integrity
IRB News : Addition of a new application type for submitting reliance agreements
 In alignment with the NIH mandate for single IRB review that goes into effect in January 2018, HSERA is being updated to include a new reliance agreement specific application. This abbreviated online form
In alignment with the NIH mandate for single IRB review that goes into effect in January 2018, HSERA is being updated to include a new reliance agreement specific application. This abbreviated online form
FIELDS REQUIRED TO SAVE A PROTOCOL
 Human Subject Research Protocols If conducting human subject research, a protocol must be submitted in WVU+kc to the WVU Institutional Review Board for review. Click on the Create IRB Protocol link located
Human Subject Research Protocols If conducting human subject research, a protocol must be submitted in WVU+kc to the WVU Institutional Review Board for review. Click on the Create IRB Protocol link located
HCM Instructions. Tip: Use the Magnifying Glass to search for values. Click the Add button to start a new request. 2/5/16 Page 1 of 8
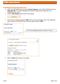 Creating Security Request Form Log in to the My BGSU portal and select Security Request under MISC SERVICES within the Employees tab. You will be redirected to the Security Request launch page within the
Creating Security Request Form Log in to the My BGSU portal and select Security Request under MISC SERVICES within the Employees tab. You will be redirected to the Security Request launch page within the
Table of Contents IRBNet Actions Designer Submission Types Revisions Reportable Events Amendments Contact Information
 FLORIDA AGRICULTURAL AND MECHANICAL UNIVERSITY Institutional Review Board (IRB) Guide to IRBNet: Table of Contents Introduction 2 IRBNet User Registration 2 IRBNet ID Number 2 Training and Credentials
FLORIDA AGRICULTURAL AND MECHANICAL UNIVERSITY Institutional Review Board (IRB) Guide to IRBNet: Table of Contents Introduction 2 IRBNet User Registration 2 IRBNet ID Number 2 Training and Credentials
NEW PROTOCOL - HUMANITARIAN USE DEVICE (HUD) TRAINING GUIDE
 NEW PROTOCOL - HUMANITARIAN USE DEVICE (HUD) TRAINING GUIDE 2014, The Trustees of Indiana University KC IRB Training Guide: New Protocol Humanitarian Use Device (HUD) 1 Table of Contents Table of Contents
NEW PROTOCOL - HUMANITARIAN USE DEVICE (HUD) TRAINING GUIDE 2014, The Trustees of Indiana University KC IRB Training Guide: New Protocol Humanitarian Use Device (HUD) 1 Table of Contents Table of Contents
LabVantage User Manual: Requests (Sample Withdrawal)
 LabVantage User Manual: Requests (Sample Withdrawal) Disclaimer: Printed copies are not the official document. Please see the online PDF file for the most up-to-date version. http://htrc.uchicago.edu 8/1/17
LabVantage User Manual: Requests (Sample Withdrawal) Disclaimer: Printed copies are not the official document. Please see the online PDF file for the most up-to-date version. http://htrc.uchicago.edu 8/1/17
Protocol Management System (PMS) Investigator User Guide. Harris Health System Administrative Approval
 Protocol Management System (PMS) Investigator User Guide Harris Health System Administrative Approval November 2013 eprotocol - Investigator User Guide 2 Table of Contents 1. INTRODUCTION 3 2. STARTING
Protocol Management System (PMS) Investigator User Guide Harris Health System Administrative Approval November 2013 eprotocol - Investigator User Guide 2 Table of Contents 1. INTRODUCTION 3 2. STARTING
Top Ten Questions About IRBNet.org
 Top Ten Questions About IRBNet.org 1. How do I register? Registration is easy. To register, simply navigate your web browser www.irbnet.org. The login area for IRBNet is in the upper right corner of the
Top Ten Questions About IRBNet.org 1. How do I register? Registration is easy. To register, simply navigate your web browser www.irbnet.org. The login area for IRBNet is in the upper right corner of the
University of the Sciences. IRBNet User s Guide
 University of the Sciences IRBNet User s Guide Instructions for On-line Submission of Research Protocols to the Research Committees - IRB, IACUC or IBC. www.irbnet.org If you have any questions regarding
University of the Sciences IRBNet User s Guide Instructions for On-line Submission of Research Protocols to the Research Committees - IRB, IACUC or IBC. www.irbnet.org If you have any questions regarding
University of Pennsylvania Institutional Review Board
 Reliance Agreement Guidance Creating Consent Templates This document provides step by step guidance on how to convert the Penn IRB approved consent form into a consent form template that can be shared
Reliance Agreement Guidance Creating Consent Templates This document provides step by step guidance on how to convert the Penn IRB approved consent form into a consent form template that can be shared
CITY UNIVERSITY OF NEW YORK. i. Visit:
 CITY UNIVERSITY OF NEW YORK I. ACCESSING IRB NET (New Registration) i. Visit: https://www.irbnet.org/release/index.html ii. New users: Click on New Registration in the top right corner iii. Fill-out the
CITY UNIVERSITY OF NEW YORK I. ACCESSING IRB NET (New Registration) i. Visit: https://www.irbnet.org/release/index.html ii. New users: Click on New Registration in the top right corner iii. Fill-out the
Amend A Protocol training guide
 Amend A Protocol training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 About this guide 4 login 5 Before you Begin Hyperlinks The Table of
Amend A Protocol training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 About this guide 4 login 5 Before you Begin Hyperlinks The Table of
retrieving study documents training guide
 retrieving study documents training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 about this document 4 retrieving study documents 5 current
retrieving study documents training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 about this document 4 retrieving study documents 5 current
Revised: February 28, Investigator/Coordinator Guide
 2014 Revised: February 28, 2014 Table of Contents Getting Started... 3 Navigational Tips... 4 Adding a New Application (Form 1)... 6 Routing Form... 11 Routing for Signatures... 15 Workflow Tracking and
2014 Revised: February 28, 2014 Table of Contents Getting Started... 3 Navigational Tips... 4 Adding a New Application (Form 1)... 6 Routing Form... 11 Routing for Signatures... 15 Workflow Tracking and
NEW PROTOCOL - NON-HUMAN SUBJECTS RESEARCH TRAINING GUIDE
 NEW PROTOCOL - NON-HUMAN SUBJECTS RESEARCH TRAINING GUIDE 2014, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Non-Human Subjects Research 1 Table of Contents Table of Contents
NEW PROTOCOL - NON-HUMAN SUBJECTS RESEARCH TRAINING GUIDE 2014, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Non-Human Subjects Research 1 Table of Contents Table of Contents
Institutional Review Board (IRB)
 IRBNet User s Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org If you have any questions regarding the submission of an IRB protocol, contact:
IRBNet User s Instructions for On-line Submission of Research Protocols to the Institutional Review Board (IRB) www.irbnet.org If you have any questions regarding the submission of an IRB protocol, contact:
WITH MODIFICATIONS ), S, PHONE NUMBERS OR OTHER FORMS, PLEASE VISIT THE IRB S WEBSITE AT:
 Modification Request Dominican College Institutional Review Board (IRB) IRB #: Date Received: IRB Approval Name & Signature: FOR MORE IRB INSTRUCTIONS, APPLICATIONS (including the RENEWAL FORM that should
Modification Request Dominican College Institutional Review Board (IRB) IRB #: Date Received: IRB Approval Name & Signature: FOR MORE IRB INSTRUCTIONS, APPLICATIONS (including the RENEWAL FORM that should
***** ***** June
 SLU eirb Investigator Guide Saint Louis University ***** eirb Investigator Submitter Guide ***** Institutional Review Board June 2011 http://eirb.slu.edu Institutional Review Board Saint Louis University
SLU eirb Investigator Guide Saint Louis University ***** eirb Investigator Submitter Guide ***** Institutional Review Board June 2011 http://eirb.slu.edu Institutional Review Board Saint Louis University
IRBNet User Manual. University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB)
 University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB) IRBNet User Manual Office of Research Integrity and Education Mary Reed Building 222 INTRODUCTION The Office
University of Denver Human Research Protection Program (HRPP) Institutional Review Board (IRB) IRBNet User Manual Office of Research Integrity and Education Mary Reed Building 222 INTRODUCTION The Office
STREAMLYNE GUIDE FOR STUDENTS/PRINCIPAL INVESTIGATORS
 STREAMLYNE GUIDE FOR STUDENTS/PRINCIPAL INVESTIGATORS Rev: 01/2017 In This Document Logging In... 1 Creating a New Protocol... 2 Revising a Returned Protocol... 7 Submitting an Amendment or Renewal Application...
STREAMLYNE GUIDE FOR STUDENTS/PRINCIPAL INVESTIGATORS Rev: 01/2017 In This Document Logging In... 1 Creating a New Protocol... 2 Revising a Returned Protocol... 7 Submitting an Amendment or Renewal Application...
You may also scroll through the Table of Contents and click on the topic or question of interest.
 We have posted our IRB process related FAQs in a searchable PDF format. You may search by using the CTRL/F key combination. Just type the search word in the box that appears. You may also scroll through
We have posted our IRB process related FAQs in a searchable PDF format. You may search by using the CTRL/F key combination. Just type the search word in the box that appears. You may also scroll through
How to Submit a New UIW IRB Application
 How to Submit a New UIW IRB Application Step Log In Visit https://uiw.forms.ethicalreviewmanager.com/ click on at the top right corner of the page. New Users: Click on New User Fill in the applicable information
How to Submit a New UIW IRB Application Step Log In Visit https://uiw.forms.ethicalreviewmanager.com/ click on at the top right corner of the page. New Users: Click on New User Fill in the applicable information
TIP SHEET GETTING STARTED: HOW TO REQUEST A MTA (MATERIAL TRANSFER AGREEMENT)
 WHAT IS IT? TIP SHEET A Material Transfer Agreement (MTA) is executed when proprietary materials are transferred in or out of Northwestern. An MTA documents the material being transferred and terms and
WHAT IS IT? TIP SHEET A Material Transfer Agreement (MTA) is executed when proprietary materials are transferred in or out of Northwestern. An MTA documents the material being transferred and terms and
ARC IRB Chair s Manual
 ARC IRB Chair s Manual This guide serves to aid an IRB Chair in becoming familiar with the basic functions of the ARC system and how to review and approve an application. ARC IRB Chairs Manual Page 2 Table
ARC IRB Chair s Manual This guide serves to aid an IRB Chair in becoming familiar with the basic functions of the ARC system and how to review and approve an application. ARC IRB Chairs Manual Page 2 Table
NAU New Project Submission. Guidance for creating a new project
 NAU New Project Submission Guidance for creating a new project Go to IRBNet Website address: https://www.irbnet.org/release/index.html Login to your account Enter your Username and Password and select
NAU New Project Submission Guidance for creating a new project Go to IRBNet Website address: https://www.irbnet.org/release/index.html Login to your account Enter your Username and Password and select
renew & Amend A Protocol training guide
 renew & Amend A Protocol training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 About this guide 4 login 5 Before You Begin Hyperlinks The Table
renew & Amend A Protocol training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 About this guide 4 login 5 Before You Begin Hyperlinks The Table
STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR / STUDENT
 Rev: 06/2017 STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR / STUDENT In This Document Protocol Application (Exempt and Expedited/Full Board Review)...2 NIH Certificate (Required)... 2 Logging In...
Rev: 06/2017 STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR / STUDENT In This Document Protocol Application (Exempt and Expedited/Full Board Review)...2 NIH Certificate (Required)... 2 Logging In...
New Protocol - expedited & full board
 New Protocol - expedited & full board training guide 2015, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Expedited & Full Board 1 table of contents Table of Contents 2 table
New Protocol - expedited & full board training guide 2015, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Expedited & Full Board 1 table of contents Table of Contents 2 table
OnCore Enterprise Research. Subject Administration Full Study
 OnCore Enterprise Research Subject Administration Full Study Principal Investigator Clinical Research Coordinator June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Table of Contents What
OnCore Enterprise Research Subject Administration Full Study Principal Investigator Clinical Research Coordinator June 2017 P a g e 1 This page is intentionally blank. P a g e 2 Table of Contents What
Last revised: September 30, e-protocol User Guide 1
 e-protocol User Guide Last revised: September 30, 2015 e-protocol User Guide 1 e-protocol is an electronic system for submitting and monitoring the status of Institutional Review Board (IRB) submissions.
e-protocol User Guide Last revised: September 30, 2015 e-protocol User Guide 1 e-protocol is an electronic system for submitting and monitoring the status of Institutional Review Board (IRB) submissions.
The IACUC is charged with the responsibility of ensuring that the Animal Welfare Act policies and procedures are followed.
 Protis is an online application system in support of research activities that require approval by the Institutional Animal Care and Use Committee (IACUC) or the Institutional Review Board (IRB). The IACUC
Protis is an online application system in support of research activities that require approval by the Institutional Animal Care and Use Committee (IACUC) or the Institutional Review Board (IRB). The IACUC
Maximo Self Service Center
 Maximo Self Service Center Once you have received an email regarding your registration approval, go to the following web address: https://maximo.mysodexo.com Log in to the Self Service Center: Your User
Maximo Self Service Center Once you have received an email regarding your registration approval, go to the following web address: https://maximo.mysodexo.com Log in to the Self Service Center: Your User
Module 6: Validating Decisions
 Table of Contents Module 6: Validating Decisions Module 6 Objective: Covers how Core Staff members process the decisions of the reviewers on each eresearch submission through a decision validation. Refer
Table of Contents Module 6: Validating Decisions Module 6 Objective: Covers how Core Staff members process the decisions of the reviewers on each eresearch submission through a decision validation. Refer
Kuali Research User Guide: Create an IRB Protocol
 Kuali Research User Guide: Create an IRB Protocol Version.0: November 06 Purpose: To create an IRB Protocol document to be used for tracking new Human Subjects Research requests. Trigger / Timing / Frequency:
Kuali Research User Guide: Create an IRB Protocol Version.0: November 06 Purpose: To create an IRB Protocol document to be used for tracking new Human Subjects Research requests. Trigger / Timing / Frequency:
NAU IRBNet Application
 NAU IRBNet Application Guidance for NAU Research Application Revised 17-0202 Go to IRBNet Website address: irbnet.org/release/index.html Login to your account - Enter your Username and Password and select
NAU IRBNet Application Guidance for NAU Research Application Revised 17-0202 Go to IRBNet Website address: irbnet.org/release/index.html Login to your account - Enter your Username and Password and select
SMART JOURNAL MANUAL 9.2
 SMART JOURNAL MANUAL 9.2 Table of Contents Smart Journal Entry... 3 Smart ERP Gadgets... 7 2 Smart Journal Entry Introduction Smart Journal is a tool that facilitates the cash transfer process for those
SMART JOURNAL MANUAL 9.2 Table of Contents Smart Journal Entry... 3 Smart ERP Gadgets... 7 2 Smart Journal Entry Introduction Smart Journal is a tool that facilitates the cash transfer process for those
University of North Florida
 Last edited by: Kayla Champaigne Last edited on: December 5, 2012 University of North Florida Institutional Review Board IRB Protocol Please note, this is a sample of the North Florida - IRB Protocol output.
Last edited by: Kayla Champaigne Last edited on: December 5, 2012 University of North Florida Institutional Review Board IRB Protocol Please note, this is a sample of the North Florida - IRB Protocol output.
Informed Consent and the Consent Form
 Informed Consent and the Consent Form What is informed consent? What does the process look like? Who can obtain consent? Where can I find more information? Consent Form Informed Consent They are NOT the
Informed Consent and the Consent Form What is informed consent? What does the process look like? Who can obtain consent? Where can I find more information? Consent Form Informed Consent They are NOT the
DATA USE AGREEMENT INVESTIGATORS: PLEASE PDF and TO:
 1730 Minor Ave STE 1900 (206) 342-1692 (206) 342-1688 DATA USE AGREEMENT INVESTIGATORS: PLEASE PDF and email TO: webhelpiaslc@crab.org TO: Participating Institution: Principal Investigator: This Data Agreement
1730 Minor Ave STE 1900 (206) 342-1692 (206) 342-1688 DATA USE AGREEMENT INVESTIGATORS: PLEASE PDF and email TO: webhelpiaslc@crab.org TO: Participating Institution: Principal Investigator: This Data Agreement
CITY UNIVERSITY OF NEW YORK. Creating a New Project in IRBNet. i. After logging in, click Create New Project on left side of the page.
 CITY UNIVERSITY OF NEW YORK Creating a New Project in IRBNet i. After logging in, click Create New Project on left side of the page. ii. Enter the title of the project, the principle investigator s (PI)
CITY UNIVERSITY OF NEW YORK Creating a New Project in IRBNet i. After logging in, click Create New Project on left side of the page. ii. Enter the title of the project, the principle investigator s (PI)
Questions and Answers. Converting Existing Protocols into CHeRP IRB
 Questions and Answers Converting Existing Protocols into CHeRP IRB Questions: (hold ctrl and click on the question to jump directly to that answer) 1. Do I need to convert my existing protocol applications
Questions and Answers Converting Existing Protocols into CHeRP IRB Questions: (hold ctrl and click on the question to jump directly to that answer) 1. Do I need to convert my existing protocol applications
Guide for Researchers: Online Human Ethics Application Form
 Ethics & Integrity Research Office HUMAN RESEARCH ETHICS ONLINE APPLICATION October 2016/V1.03 Guide for Researchers: Online Human Ethics Application Form ENQUIRIES Senior Human Ethics Officer University
Ethics & Integrity Research Office HUMAN RESEARCH ETHICS ONLINE APPLICATION October 2016/V1.03 Guide for Researchers: Online Human Ethics Application Form ENQUIRIES Senior Human Ethics Officer University
Responding to REB Feedback
 Responding to REB Feedback A ROMEO Hasty Handbook Presented by the CHEO Research Institute Table of Contents: What is REB Feedback? The REB Feedback Letter The Investigator Response Letter Finding the
Responding to REB Feedback A ROMEO Hasty Handbook Presented by the CHEO Research Institute Table of Contents: What is REB Feedback? The REB Feedback Letter The Investigator Response Letter Finding the
era(infoed) Electronic Submission Instructions and Tips: Initial Application
 era(infoed) Electronic Submission Instructions and Tips: Initial Application This instructional document introduces users to submitting NEW research protocols electronically through the era(infoed) system.
era(infoed) Electronic Submission Instructions and Tips: Initial Application This instructional document introduces users to submitting NEW research protocols electronically through the era(infoed) system.
Click. IRB Study Submission Guide
 Click IRB Study Submission Guide October 2016 Table of Contents Logging In... 3 Creating a New Study... 4 Finding More Information... 5 Editing a Study... 5 Checking the Study for Errors... 6 Submitting
Click IRB Study Submission Guide October 2016 Table of Contents Logging In... 3 Creating a New Study... 4 Finding More Information... 5 Editing a Study... 5 Checking the Study for Errors... 6 Submitting
NEW PROTOCOL - REQUEST TO DEFER IRB REVIEW
 NEW PROTOCOL - REQUEST TO DEFER IRB REVIEW TRAINING GUIDE 2014, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Request to Defer IRB Review 1 Table of Contents Table of Contents
NEW PROTOCOL - REQUEST TO DEFER IRB REVIEW TRAINING GUIDE 2014, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Request to Defer IRB Review 1 Table of Contents Table of Contents
New Protocol - emergency use training guide
 New Protocol - emergency use training guide 2015, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Emergency Use 1 table of contents Table of Contents 2 table of contents 3 about
New Protocol - emergency use training guide 2015, The Trustees of Indiana University KC IRB Training Guide: New Protocol - Emergency Use 1 table of contents Table of Contents 2 table of contents 3 about
irb.unm.edu
 IRBNet Submission Instructions The purpose of this document is to provide IRBNet submission instructions for UNM researchers using IRBNet. Please contact the Office of the IRB for assistance: irb.unm.edu
IRBNet Submission Instructions The purpose of this document is to provide IRBNet submission instructions for UNM researchers using IRBNet. Please contact the Office of the IRB for assistance: irb.unm.edu
UVMClick IRB Study Submission Guide
 UVMClick IRB Study Submission Guide September 2018 Table of Contents How to Login 3 How to Create a New Study 4 Find More Information 5 How to Edit a Study 6 Check the Study for Errors 7 Submit the Study
UVMClick IRB Study Submission Guide September 2018 Table of Contents How to Login 3 How to Create a New Study 4 Find More Information 5 How to Edit a Study 6 Check the Study for Errors 7 Submit the Study
STUDY ASSISTANT. Adding a New Study & Submitting to the Review Board. Version 10.03
 STUDY ASSISTANT Adding a New Study & Submitting to the Review Board Version 10.03 Contents Introduction... 3 Add a Study... 3 Selecting an Application... 3 1.0 General Information... 3 2.0 Add Department(s)...
STUDY ASSISTANT Adding a New Study & Submitting to the Review Board Version 10.03 Contents Introduction... 3 Add a Study... 3 Selecting an Application... 3 1.0 General Information... 3 2.0 Add Department(s)...
ELECTRONIC RESEARCH STUDY APPLICATION (ERSA) INITIAL APPLICATION GUIDEBOOK. (Version date 03/03/2006)
 ELECTRONIC RESEARCH STUDY APPLICATION (ERSA) INITIAL APPLICATION GUIDEBOOK (Version date 03/03/2006) Table of Contents ERSA Home Page and Login 3 CRC Desktop 4 Creating a: New Study Application 5 Amendment
ELECTRONIC RESEARCH STUDY APPLICATION (ERSA) INITIAL APPLICATION GUIDEBOOK (Version date 03/03/2006) Table of Contents ERSA Home Page and Login 3 CRC Desktop 4 Creating a: New Study Application 5 Amendment
Welcome to the Guest Access Portal
 Welcome to the Guest Access Portal The Guest Access Portal allows students to grant a guest, such as a parent or spouse, access to view their education records in Banner Self-Service. Only students may
Welcome to the Guest Access Portal The Guest Access Portal allows students to grant a guest, such as a parent or spouse, access to view their education records in Banner Self-Service. Only students may
Using the e Version of the Protocol Summary. University of Utah IRB Version: January 2012
 Using the e Version of the Protocol Summary University of Utah IRB Version: January 2012 What is the e Version of the Protocol Summary? Beginning January 2012, ERICA will create an up todate e version
Using the e Version of the Protocol Summary University of Utah IRB Version: January 2012 What is the e Version of the Protocol Summary? Beginning January 2012, ERICA will create an up todate e version
Job Aid. Account Distribution, LABOR
 Job Aid Account Distribution, LABOR Account Distribution, LABOR Account Distribution, LABOR is the Approval Category used to make changes to the index and account that the employee s job is charged to.
Job Aid Account Distribution, LABOR Account Distribution, LABOR Account Distribution, LABOR is the Approval Category used to make changes to the index and account that the employee s job is charged to.
How to Create and Submit an Amendment in INSPIR II
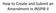 How to Create and Submit an Amendment in INSPIR II Login into INSPIR and from your Home page, click on My Studies to list all the studies that you have access to Locate the study and click on the Open
How to Create and Submit an Amendment in INSPIR II Login into INSPIR and from your Home page, click on My Studies to list all the studies that you have access to Locate the study and click on the Open
STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018. I. Protocol Application & IRB Certification Tutorial...2
 STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018 I. Protocol Application & IRB Certification Tutorial...2 II. Logging In (Password)... 2 III. Creating a New Protocol Record / Menu Bar...
STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018 I. Protocol Application & IRB Certification Tutorial...2 II. Logging In (Password)... 2 III. Creating a New Protocol Record / Menu Bar...
Drexel University. Page 1 of 48. Version November agf
 Drexel University Page 1 of 48 TABLE OF CONTENTS Getting Started. 3 Creating a New Protocol.. 4 General Information... 7 Organization Information... 9 Investigators/Study Personnel.. 10 Correspondents
Drexel University Page 1 of 48 TABLE OF CONTENTS Getting Started. 3 Creating a New Protocol.. 4 General Information... 7 Organization Information... 9 Investigators/Study Personnel.. 10 Correspondents
Submitting a Response to Modifications Required by the IRB to Secure Approval
 Submitting a Response to Modifications Required by the IRB to Secure Approval If you have submitted an item to the IRB, and the IRB has responded with a Modifications Required to Secure Approval memorandum,
Submitting a Response to Modifications Required by the IRB to Secure Approval If you have submitted an item to the IRB, and the IRB has responded with a Modifications Required to Secure Approval memorandum,
What is A web-based protocol submission system for IACUC and IRB protocols
 What is Tick@Lab? A web-based protocol submission system for IACUC and IRB protocols The system offers: o Available in one place 24/7 from anywhere o Smart forms available to complete your desired request
What is Tick@Lab? A web-based protocol submission system for IACUC and IRB protocols The system offers: o Available in one place 24/7 from anywhere o Smart forms available to complete your desired request
Module 6: Validating Decisions
 Table of Contents Module 6: Validating Decisions Module 6 Objective: Covers how Core Staff members process the decisions of the reviewers on each eresearch submission through a decision validation. Refer
Table of Contents Module 6: Validating Decisions Module 6 Objective: Covers how Core Staff members process the decisions of the reviewers on each eresearch submission through a decision validation. Refer
Version 2.1 June 12, 2018
 Version 2.1 June 12, 2018 MAESTRO Review System login: https://maestro.research.nmsu.edu MAESTRO Help & Training: http://maestrohelp.research.nmsu.edu MAESTRO & IRB Questions and Helpdesk Support: Call
Version 2.1 June 12, 2018 MAESTRO Review System login: https://maestro.research.nmsu.edu MAESTRO Help & Training: http://maestrohelp.research.nmsu.edu MAESTRO & IRB Questions and Helpdesk Support: Call
Adding/Removing Internal Investigators from an Already Approved Study (Section 3.0)
 Adding/Removing Internal Investigators from an Already Approved Study (Section 3.0) Internal Investigators These are BU/BMC faculty and staff Boston Public Health Commission and Boston Healthnet Community
Adding/Removing Internal Investigators from an Already Approved Study (Section 3.0) Internal Investigators These are BU/BMC faculty and staff Boston Public Health Commission and Boston Healthnet Community
BOULDER IRB era InfoEd Amendments
 BOULDER IRB era InfoEd Amendments Last Update: 2017/12/06 Preface: This guide explains how to submit an Amendment to modify an approved study. If you already have a submission pending review do not submit
BOULDER IRB era InfoEd Amendments Last Update: 2017/12/06 Preface: This guide explains how to submit an Amendment to modify an approved study. If you already have a submission pending review do not submit
Drexel University. Version March Page 1 of 58 Version 01 March 2017 agf
 Drexel University Version March 2017 Page 1 of 58 TABLE OF CONTENTS Getting Started 3 Connecting through the VPN.. 4 Electronic Questionnaires and Applications...10 Welcome to eirb 11 Creating a New Protocol.13
Drexel University Version March 2017 Page 1 of 58 TABLE OF CONTENTS Getting Started 3 Connecting through the VPN.. 4 Electronic Questionnaires and Applications...10 Welcome to eirb 11 Creating a New Protocol.13
Initiating a Security Request Form
 Initiating a Security Request Form You may request security for employees with an online form. This form allows you to initiate the request with the level of desired access and then it will be routed through
Initiating a Security Request Form You may request security for employees with an online form. This form allows you to initiate the request with the level of desired access and then it will be routed through
copying a protocol in Kc irb training guide
 copying a protocol in Kc irb training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 about this guide 4 copying a protocol 5 protocol actions
copying a protocol in Kc irb training guide 2015, The Trustees of Indiana University 1 table of contents Table of Contents 2 table of contents 3 about this guide 4 copying a protocol 5 protocol actions
To allow Investigators to complete the required Certification for a proposal, you must complete the steps below:
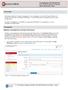 Investigator Certifications Non-NIH/PHS Sponsor Proposals Quick Reference Card Overview: Individuals listed as Principal Investigator or Co-Investigator on the Key Persons/Personnel screen are required
Investigator Certifications Non-NIH/PHS Sponsor Proposals Quick Reference Card Overview: Individuals listed as Principal Investigator or Co-Investigator on the Key Persons/Personnel screen are required
Human Subjects Development Guide
 Research Integrity & Compliance Institutional Review Board Human Subjects Development Guide ERA Version 15, Nov. 2016 Table of Contents Logging In... 3 Adding a Delegate... 4 Viewing Items as a Delegate...
Research Integrity & Compliance Institutional Review Board Human Subjects Development Guide ERA Version 15, Nov. 2016 Table of Contents Logging In... 3 Adding a Delegate... 4 Viewing Items as a Delegate...
Quick Tip Sheet. Log into your Web Portal at with your user name and password
 To Add Badge Holder: 3. Select: (+) located at the top right of the page 4. Place your cursor in the field for Badge / Sticker # 5. Scan in the 10-digit Badge number by placing the card over your desktop
To Add Badge Holder: 3. Select: (+) located at the top right of the page 4. Place your cursor in the field for Badge / Sticker # 5. Scan in the 10-digit Badge number by placing the card over your desktop
The most current protocol information should be entered. This includes incorporation of all past amendments and modifications.
 Georgetown University Institutional Review Board Frequently Asked Questions (FAQs) about eric 1. What is eric? eric stands for Electronic Research and Institutional Compliance. This is an on-line IRB application
Georgetown University Institutional Review Board Frequently Asked Questions (FAQs) about eric 1. What is eric? eric stands for Electronic Research and Institutional Compliance. This is an on-line IRB application
UNIVERSITY OF PENNSYLVANIA
 UNIVERSITY OF PENNSYLVANIA Research Inventory System- Subaward Generator Module (RIS-SGM) August 2013 This document contains a user instruction guide for the creation and submission of subaward requests.
UNIVERSITY OF PENNSYLVANIA Research Inventory System- Subaward Generator Module (RIS-SGM) August 2013 This document contains a user instruction guide for the creation and submission of subaward requests.
Electronic Signature Guide
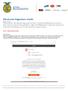 Electronic Signature Guide Created: 07/2017 Please follow the step by step guide on how to electronically sign a Living to Serve grant contract through Nitro. After the FFA Advisor signs the contract,
Electronic Signature Guide Created: 07/2017 Please follow the step by step guide on how to electronically sign a Living to Serve grant contract through Nitro. After the FFA Advisor signs the contract,
eirb User Manual for Research Staff
 eirb User Manual for Research Staff Version 4 1 10.2016 Table of Contents eirb Access... 3 Log-in to eirb... 4 Overview of the eirb Submission and Review Process... 7 Create a New Application and Specify
eirb User Manual for Research Staff Version 4 1 10.2016 Table of Contents eirb Access... 3 Log-in to eirb... 4 Overview of the eirb Submission and Review Process... 7 Create a New Application and Specify
REFERENCE GUIDE FOR MANUAL DATA INPUT v1.1
 REFERENCE GUIDE FOR MANUAL DATA INPUT v. TABLE OF CONTENTS Introduction User Roles Logging in to VIVO Site Administration Menu Navigating VIVO Data Input Overview Profile Fields Mapping a CV to a VIVO
REFERENCE GUIDE FOR MANUAL DATA INPUT v. TABLE OF CONTENTS Introduction User Roles Logging in to VIVO Site Administration Menu Navigating VIVO Data Input Overview Profile Fields Mapping a CV to a VIVO
PI Certification Quick Guide
 PI Certification Quick Guide 1 P age Table of Contents Certifications... 3 PI Self-Certification... 3 PI is Certifying via Link in Email Notification... 3 PI Goes Directly to the Proposal... 6 Certification
PI Certification Quick Guide 1 P age Table of Contents Certifications... 3 PI Self-Certification... 3 PI is Certifying via Link in Email Notification... 3 PI Goes Directly to the Proposal... 6 Certification
DISCOVR-e USER MANUAL. Vanderbilt University Human Research Protection Program
 DISCOVR-e USER MANUAL Vanderbilt University Human Research Protection Program Table of Contents Introduction and Overview... 3 Log into the System... 4 Investigator Dashboard... 5 Submitting a New Study...
DISCOVR-e USER MANUAL Vanderbilt University Human Research Protection Program Table of Contents Introduction and Overview... 3 Log into the System... 4 Investigator Dashboard... 5 Submitting a New Study...
How to Apply for a Teaching Position as an Internal Applicant
 How to Apply for a Teaching Position as an Internal Applicant I. Website Navigation Internal applicants include all GCSD employees who currently hold a position for which certification/licensure is required.
How to Apply for a Teaching Position as an Internal Applicant I. Website Navigation Internal applicants include all GCSD employees who currently hold a position for which certification/licensure is required.
IRB RESEARCH REPOSITORY COMPLIANCE PROGRAM: Repository Protocols and FAQs
 IRB RESEARCH REPOSITORY COMPLIANCE PROGRAM: Repository Protocols and FAQs Compliance Deadline: August 31, 2011 Kathryn Schuff, MD, MCR Andrea Johnson, JD IRB Co-Chair Regulatory Specialist, ORIO Agenda
IRB RESEARCH REPOSITORY COMPLIANCE PROGRAM: Repository Protocols and FAQs Compliance Deadline: August 31, 2011 Kathryn Schuff, MD, MCR Andrea Johnson, JD IRB Co-Chair Regulatory Specialist, ORIO Agenda
Kuali Coeus Implementation: IRB Progress Report. October 7, 2013
 Kuali Coeus Implementation: IRB Progress Report October 7, 2013 IRB Presentation Outline Current State Documentation Future State Process Development KC-IRB24 Protocol Development-Submission Process Process
Kuali Coeus Implementation: IRB Progress Report October 7, 2013 IRB Presentation Outline Current State Documentation Future State Process Development KC-IRB24 Protocol Development-Submission Process Process
IRB Blueprinting Workshop 1
 Kuali Coeus Implementation: IRB Blueprinting Workshop 1 KC IRB 24: Protocol Development Submission KC IRB 4: 118 Designation October 31, 2013 Goals and Outcomes Goals Obtain feedback on the proposed future
Kuali Coeus Implementation: IRB Blueprinting Workshop 1 KC IRB 24: Protocol Development Submission KC IRB 4: 118 Designation October 31, 2013 Goals and Outcomes Goals Obtain feedback on the proposed future
How Does the PI make Pre Review ICF Changes?
 How Does the PI make Pre Review ICF Changes? 1 You, as PI of a study, have received notification from the IRB that Changes [are] required by IRB Staff. These changes include a request for a revision of
How Does the PI make Pre Review ICF Changes? 1 You, as PI of a study, have received notification from the IRB that Changes [are] required by IRB Staff. These changes include a request for a revision of
User Guide for DCP Consortia 2012
 AQuIP On line Accrual Reporting System (OARS) User Guide for DCP Consortia 2012 This User Guide provides an introduction to the AQuIP On line Accrual Reporting System (OARS) for the Division of Cancer
AQuIP On line Accrual Reporting System (OARS) User Guide for DCP Consortia 2012 This User Guide provides an introduction to the AQuIP On line Accrual Reporting System (OARS) for the Division of Cancer
