Jointly Organised by: Presenter: Certificates endorsed by the National. Training Grant is available under HRDF SBL Scheme
|
|
|
- Gillian Robinson
- 6 years ago
- Views:
Transcription
1 The course training program consists of 9 principal modules and 2 additional modules, each of 3 days duration. These modules cover the essential principles of Good Manufacturing Practice (GMP). Participants are expected to gain an understanding of current requirements and future international trends within the pharmaceutical industry. Each participant will be assessed on their level of participation within classroom discussion, assignments and their level of competence in achieving the course objectives. Assignments will be case studies based on actual events that have occurred in the pharmaceutical industry. Training Grant is available under HRDF SBL Scheme Aims and Objectives The aim of the course is to provide an in-depth understanding of international GMP and the knowledge and know-how to be able to implement Good Manufacturing in the work place. Trainers This course has been developed by SeerPharma Pty Ltd and trainers are provided by SeerPharma Pty Ltd, Australia or SeerPharma (Singapore) Pte Ltd. All SeerPharma trainers hold higher education degrees with a minimum of a Bachelor s degree and have a number of years of industry experience in Quality Management or Production Management roles in major and multinational companies. They have experience in all international regulatory standards including FDA, EU, PIC/S, TGA and ISO. The trainer for each module will have specific expertise in that subject matter. SeerPharma is Australia s and Asia Pacific s premier training & consulting group offering integrated consulting, training and technical services to Australia and the Asia Pacific region to meet all international regulatory standards. Who Should Attend Key Personnel in any Aspect of GMP & Quality Management, Managers, Engineers, Executives, Quality Practitioners and any member of a pharmaceutical factory who is from Research and Development, Quality and Production will find this program relevant and beneficial to their job function. Jointly Organised by: Presenter: Certificates endorsed by the National Malaysian Organisation of Pharmaceutical Industries National Pharmaceutical Control Bureau, MOH Pharmaceutical Control Bureau, Ministry of Health, Malaysia will be awarded to participants upon successful completion of each module. For further details please visit
2 COURSE OUTLINE Module 1 International Good Manufacturing, Quality Management Systems and GMP for Pharmaceutical Operations Day 1 QA Principles & International GMPs Quality Management, Quality Assurance & Quality Control Day2 Key Quality Assurance Systems and GMP Responsibilities for Managers & Supervisors GMP Principles for Manufacturing Operations Day 3 GMP Principles for Packaging Operations Equipment Management Module 2 Validation Principles and Day 1 Validation Principles & International Regulations Validation Master Plans and Validation Documents Equipment Qualification and Commissioning Day 2 Developing Validation Specifications Compiling Functional/Design Specifications and Checking against User Requirement Specifications Preparing DQ, IQ, OQ and PQ protocols Day 3 Creating Protocols and Protocol Execution Deviation Management Final Summary Report Module 3 Contamination Control Day 1 Introduction to Contamination Control Microbiological Aspects of Manufacturing Day 2 Cleaning and Sanitation HVAC and Controlled Environments control & qualification Day 3 Environmental Monitoring Programs Control of Water Systems Module 4 Risk Management in Pharmaceutical Operations (ICH Q9) Day 1 Principles of Risk Management and ICH Q9 Risk Management Techniques - FMEA, FTA, HACCP Day 2 Risk Management to Compliance and Quality Assurance Management Applying Risk Management in Compliance Day 3 Risk Management Principles in Validation Programs Applying Risk Management in Validation Module 5 - Good (Quality Control) Laboratory (GLPs) Day 1 Introduction to Good (Quality Control) Laboratory (GLPs) Qualification and Calibration of Laboratory Equipment Day 2 Analytical Method Validation Biological assays Validation and Control Basic Statistics for Quality Control Laboratories Day 3 Pharmaceutical Sampling Plan Pharmaceutical Stability Programs Module 7 - Solid Dose Manufacture Principles and Day 1 QA/GMPs Over Finished Dose Forms (FDF) Granulation Technology & Control Day 2 Blending and Milling Technology & Control Powder Transfer Technology and Containment System Day 3 Compression, Encapsulation and Control Coating Technology & Control Module 8 Liquid and Cream Manufacture Principles and Day 1 Encapsulation Technology & Control Packaging and Labeling of Oral Dose Forms Day 2 Introduction to Oral Liquid Preparation Formulation Introduction to Topical Preparation Formulation Day 3 Manufacture and Packaging of Cream and Ointments Case Study Validation Plans for a new Topical Products Module 9 GxP and Quality Auditing Day 1 Critical role of quality audit in GxP compliance & improvement GxP audit schedule, managing regulatory audits in an effective manner, what to expect from GMP licensing audits Day 2 The role of supplier audits for actives, excipients and components in Vendor management Documents, records & data for effective audits. Day 3 Four fundamental steps of auditing explained in detail, tips on how to manage & facilitate audits in a constructive manner Utilisation of risk management in relation to prioritising audits Introduction to Computer Validation: Principles and Practice Day 1 Regulations and GAMP. System Development Life Cycle and Risk Assessment Day 2 Network Qualification and Planning Phases. Pre Development Phases Day 3 Development, Testing, Qualification and Use 21 CFR Part 11 Good Laboratory (for Non-Clinical Laboratories) Day 1 Review of FDA Regulations and Controls Principles of Good Laboratory (GLP's) in a Regulated Environment Day 2 Review of FDA CFR 58 Regulations Day 3 GLPs and Documentation Compliance Protocols, Records and Reports Module 6 Compliance with Good Manufacturing for the Pharmaceutical Engineer Day 1 Facility Layout and Design Principles Design and Construction of Critical Utilities: inc. Water, Gases and Steam Day 2 Water Systems: Design, Control & Validation HVAC: Design, Control & Validation Day 3 Qualification of Processing Equipment Planned Preventative Maintenance and Calibration Data Base
3 METHODOLOGY: Lectures, workshops, case studies and group activities. ASSESSMENT: A variety of assessment strategies will be used and may include assignments, classroom engagement, projects and presentations. Participants will be informed of the assessment method, date of assessment and percentage contribution at the start of the module. Course Dates * : Module 1 International Good Manufacturing Quality Management Systems and GMP for Pharmaceutical Operations 8 10 March 2010 (Mon Weds) at (Eastin Hotel) Module 2 Validation Principles and April 2010 (Mon Weds) at (Eastin Hotel) Introduction to Computer System Validation: Principles and 4 6 May 2010 (Tues Thur) at (Eastin Hotel) Module 3 Contamination Control May 2010 (Mon Weds) at (Boulevard Hotel) Module 4 Risk Management in Pharmaceutical Operations (ICHQ9) June 2010 (Mon Weds) at (Boulevard Hotel) Good Laboratory (for Non-Clinical Laboratories) 5 7 July 2010 (Mon Weds) at (Boulevard Hotel) Module 5 Good Quality Control Laboratory July 2010 (Mon Weds) at (Boulevard Hotel) Module 6 Compliance with Good Manufacturing for the Pharmaceutical Engineer September 2010 (Tues Thurs) at (Boulevard Hotel) Module 7 Solid Dose Manufacture Principles and October 2010 (Mon Weds) at (Boulevard Hotel) Module 8 Liquid and Cream Manufacture Principles and November 2010 (Mon Weds) at (Boulevard Hotel) Module 9 GxP and Quality Auditing December 2010 (Mon Weds) at (Boulevard Hotel) Fee per participant per course: (The fee includes course materials, lunch and refreshments) MOPI Member 30 days before commencement of course RM2, days before commencement of course RM2, days before commencement of course RM2,700,00 Non-MOPI Member 30 days before commencement of course RM2, days before commencement of course RM days before commencement of course RM Foreign Participant 30 days before commencement of course USD $1, days before commencement of course USD $1, days before commencement of course USD $1, Time: 9.00 am 5.00 pm Venue: Eastin Hotel, 13, Jalan 16/11, Pusat Dagang Seksyen 16, Petaling Jaya, Selangor Darul Ehsan Tel: Hotel Room Rates: Single without breakfast RM210++ Single with breakfast RM232++ Twin Sharing with breakfast RM254++ Venue: Boulevard Hotel, Mid Valley City, Lingkaran Syed Putra, Kuala Lumpur, Malaysia Tel: Hotel Room Rates: Single with breakfast RM280 nett Twin Sharing with breakfast RM295 nett Alternative Hotel Room at Cititel Hotel Single with breakfast RM250 nett Twin Sharing with breakfast RM225 Nett * Dates and Instructors are subject to change depending on attendance feedbacks and instructor availability. In case of a change, updated dates and instructor profile will be advised to the organizer and the attendees prior to the start of each course. BOOK YOUR SEAT NOW!!! For further enquiries, please contact: Mike/Janet, MOPI 1st Floor,Wisma Yan, No 17 & 19, Jalan Selangor, Petaling Jaya,Selangor, West Malaysia Tel: , Fax: mike@mopi.org.my and admin@mopi.org.my
4 ADMINISTRATION DETAILS: Important Notice: Payment is required with registration and must be received 2 weeks prior to the start of the relevant module to guarantee your place. Walkin participants will only be admitted on the basis of space availability at the course and with immediate full payment by company cheque in favour of the Malaysian Organisation of Pharmaceutical Industries. Registration will be treated as confirmed only upon receipt of payment in full. CANCELLATIONS & TRANSFERS: If a registrant is unable to attend, a substitute candidate is welcome at no extra charge. Please provide the name and the title of the substitute participant at least 2 working days prior to the relevant course. Notice of cancellation by fax/ is required 14 working days prior to commencement of each module and refund less RM500 as administration charge will be made. However a complete set of documentation will be sent to you. Regrettably, no refund can be made for cancellations received less than 10 working days prior to the commencement of each module. However a complete set of documentation will be sent to you. MOPI / SeerPharma reserves the right to cancel or reschedule the training modules. All efforts will be taken to inform participants of any change. MOPI /SeerPharma however will not be held liable for reimbursement of any claims or expenses should cancellation or rescheduling occur. REGISTRATION FORM Subject to Administration Dear Sir Subject to Administration details Please register the following participant(s) for the above program. (To be completed in BLOCK LETTERS) 1 Name 2 Name MOPI Member Non-Member Foreign 3 Name 4 Name (If space insufficient, please attach a separate list) Enclosed cheque/bank draft No for RM being payment for participant(s) made in favour of the Malaysian Organisation of Pharmaceutical Industries. Please tick accordingly: Submitted by: Module 1 International Good Manufacturing Quality Management Systems and GMP for Pharmaceutical Operations 8 10 March 2010 (Mon Weds) at (Eastin Hotel) Module 2 Validation Principles and April 2010 (Mon Weds) at (Eastin Hotel) Introduction to Computer System Validation: Principles and 4 6 May 2010 (Tues Thur) at (Eastin Hotel) Module 3 Contamination Control May 2010 (Mon Weds) at (Boulevard Hotel) Module 4 Risk Management in Pharmaceutical Operations (ICHQ9) June 2010 (Mon Weds) at (Boulevard Hotel) Good Laboratory (for Non-Clinical Laboratories) 5 7 July 2010 (Mon Weds) at (Boulevard Hotel) Module 5 Good Quality Control Laboratory July 2010 (Mon Weds) at (Boulevard Hotel) Module 6 Compliance with Good Manufacturing for the Pharmaceutical Engineer September 2010 (Tues Thurs) at (Boulevard Hotel) Module 7 Solid Dose Manufacture Principles and October 2010 (Mon Weds) at (Boulevard Hotel) Module 8 Liquid and Cream Manufacture Principles and November 2010 (Mon Weds) at (Boulevard Hotel) Module 9 GxP and Quality Auditing December 2010 (Mon Weds) at (Boulevard Hotel) Name Designation Company Stamp (with Address, Telephone & Fax Number) Office Use Only Registration Accepted on. Payment Accepted on..
5 Hotel Map Eastin Hotel, 13, Jalan 16/11, Pusat Dagang Seksyen 16, Petaling Jaya,Selangor Darul Ehsan Boulevard Hotel, Mid Valley City, Lingkaran Syed Putra, Kuala Lumpur, Malaysia Parking at Zone C, Mid Valley, to Ground Floor North Court lift, Use the exit door next to the lift to Boulevard Hotel
YEAR Endorsed by: Training Grant is available under HRDF SBL Scheme
 YEAR 2018 The course training program consists of 9 principal modules and 6 additional modules. These modules cover the essential principles of Good Manufacturing Practice (GMP). Participants are expected
YEAR 2018 The course training program consists of 9 principal modules and 6 additional modules. These modules cover the essential principles of Good Manufacturing Practice (GMP). Participants are expected
In-Vitro Diagnostic Directive (IVDD) and IVDD Technical File Preparation Workshop. 3-4 July 2014 TÜV SÜD
 In-Vitro Diagnostic Directive (IVDD) and IVDD Technical File Preparation Workshop 3-4 July 2014 TÜV SÜD Introduction The In-Vitro Diagnostic Directive (IVDD) 98/79/EC applies to all In-Vitro Diagnostic
In-Vitro Diagnostic Directive (IVDD) and IVDD Technical File Preparation Workshop 3-4 July 2014 TÜV SÜD Introduction The In-Vitro Diagnostic Directive (IVDD) 98/79/EC applies to all In-Vitro Diagnostic
FMM Institute. Centre for Professional Development
 FMM Institute Centre for Professional Development In-House Training Available Course Description Participants will learn how to create and edit bullet slides, use PowerPoint s drawing tools, incorporate
FMM Institute Centre for Professional Development In-House Training Available Course Description Participants will learn how to create and edit bullet slides, use PowerPoint s drawing tools, incorporate
FMM Institute. Centre for Professional Development
 FMM Institute Centre for Professional Development In-House Training Available Course Description PivotTables are powerful data analysis tools, yet most Excel users do not use them to their fullest potential.
FMM Institute Centre for Professional Development In-House Training Available Course Description PivotTables are powerful data analysis tools, yet most Excel users do not use them to their fullest potential.
Configuration, Installation, Commissioning, Troubleshooting, Operation & Maintenance
 ISO 9001:2008 Certified ISO 29990:2010 Certified (Certificate No:1007049195) ISO 29990 (Certificate No: 1078694951) Foundation Fieldbus Troubleshooting, Operation & Maintenance 29 May - 02 June 2016 Muscat
ISO 9001:2008 Certified ISO 29990:2010 Certified (Certificate No:1007049195) ISO 29990 (Certificate No: 1078694951) Foundation Fieldbus Troubleshooting, Operation & Maintenance 29 May - 02 June 2016 Muscat
FMM Institute. Centre for Professional Development
 FMM Institute Centre for Professional Development In-House Training Available Course Description This course introduce participants to the more advanced features in Microsoft Access 2003. Tasks include
FMM Institute Centre for Professional Development In-House Training Available Course Description This course introduce participants to the more advanced features in Microsoft Access 2003. Tasks include
FMM Institute. Centre for Professional Development
 FMM Institute Centre for Professional Development In-House Training Available Course Description This course introduce participants to the more advanced features in Microsoft Access 2003. Tasks include
FMM Institute Centre for Professional Development In-House Training Available Course Description This course introduce participants to the more advanced features in Microsoft Access 2003. Tasks include
IT TRAINING COURSE - JANUARY TO DECEMBER 2019
 IT TRAINING COURSE JANUARY TO DECEMBER In Collaboration with All courses are HRDF claimable is Microsoft WORD Microsoft Word (Fundamental & Discover The Techniques to A Professional Word Report 07 07 2
IT TRAINING COURSE JANUARY TO DECEMBER In Collaboration with All courses are HRDF claimable is Microsoft WORD Microsoft Word (Fundamental & Discover The Techniques to A Professional Word Report 07 07 2
Facilitator. TIME SERIES ECONOMETRICS FOR THE PRACTITIONER (E-VIEWS) Time Series Econometrics- I
 TIME SERIES ECONOMETRICS FOR THE PRACTITIONER (E-VIEWS) Econometrics- I 11 12 April 2014 Econometrics II 18 19 April 2014 AND PANEL DATA ECONOMETRICS (STATA) Econometrics I 2 3 May 2014 Econometrics II
TIME SERIES ECONOMETRICS FOR THE PRACTITIONER (E-VIEWS) Econometrics- I 11 12 April 2014 Econometrics II 18 19 April 2014 AND PANEL DATA ECONOMETRICS (STATA) Econometrics I 2 3 May 2014 Econometrics II
Global Impact for a Safe and Sustainable Future
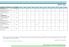 TRAINING CALENDAR FOR YEAR 2018 ISO 9001:2015 Quality Management System Foundation ISO 9001:2015 Quality Management System Internal Auditor ISO 9001:2015 QMS Auditor/Lead Auditor (IRCA Course Certification
TRAINING CALENDAR FOR YEAR 2018 ISO 9001:2015 Quality Management System Foundation ISO 9001:2015 Quality Management System Internal Auditor ISO 9001:2015 QMS Auditor/Lead Auditor (IRCA Course Certification
IT Audit Essentials. Date: 10 th 12 th March 2015 Time: 9 am to 5.30 pm Venue: Iverson Associates, Center Point Bandar Utama, Kuala Lumpur
 IT Audit Essentials Date: 10 th 12 th March 2015 Time: 9 am to 5.30 pm Venue: Iverson Associates, Center Point Bandar Utama, Kuala Lumpur IT Audit Essentials Workshop Overview ISACA Malaysia Chapter is
IT Audit Essentials Date: 10 th 12 th March 2015 Time: 9 am to 5.30 pm Venue: Iverson Associates, Center Point Bandar Utama, Kuala Lumpur IT Audit Essentials Workshop Overview ISACA Malaysia Chapter is
GMP/GDP auditor training course
 GMP/GDP auditor training course Frankfurt, November 20 th and 21 st 2018 INTRODUCTION Asociación Forum Auditorías (AFA) is providing 3 rd party audit services since 2005 to pharmaceutical companies. Since
GMP/GDP auditor training course Frankfurt, November 20 th and 21 st 2018 INTRODUCTION Asociación Forum Auditorías (AFA) is providing 3 rd party audit services since 2005 to pharmaceutical companies. Since
FMM Institute. Centre for Professional Development
 FMM Institute Centre for Professional Development In-House Training Available Course Description This course will cover in depth on VBA such as create userform application, design own toolbar, shortcut
FMM Institute Centre for Professional Development In-House Training Available Course Description This course will cover in depth on VBA such as create userform application, design own toolbar, shortcut
Tunku Abdul Rahman University College is conducting CIMA Certificate in Business Accounting (Cert BA).
 PUBLIC COURSE Tunku Abdul Rahman University College is conducting CIMA Certificate in Business Accounting (Cert BA). The CIMA Cert BA elevates people and businesses to success by developing skills and
PUBLIC COURSE Tunku Abdul Rahman University College is conducting CIMA Certificate in Business Accounting (Cert BA). The CIMA Cert BA elevates people and businesses to success by developing skills and
TÜV Rheinland Functional Safety Program
 IICA Functional Safety Engineer Training Safety Instrumented Systems TÜV Rheinland Functional Safety Program IICA Functional Safety Engineer SIS Training TÜV Rheinland Functional Safety Program The IICA
IICA Functional Safety Engineer Training Safety Instrumented Systems TÜV Rheinland Functional Safety Program IICA Functional Safety Engineer SIS Training TÜV Rheinland Functional Safety Program The IICA
HVAC DESIGN COURSE LEVEL 1 : ESSENTIAL TOOLS FOR HIGH PERFORMANCE BUILDING DESIGNERS
 HVAC DESIGN COURSE LEVEL 1 : ESSENTIAL TOOLS FOR HIGH PERFORMANCE BUILDING DESIGNERS DATE TIME VENUE : 4th to 6th August 2014 (Monday to Wednesday) : 9.00 am to 6.00pm (Registration at 8.15am) : Atlanta
HVAC DESIGN COURSE LEVEL 1 : ESSENTIAL TOOLS FOR HIGH PERFORMANCE BUILDING DESIGNERS DATE TIME VENUE : 4th to 6th August 2014 (Monday to Wednesday) : 9.00 am to 6.00pm (Registration at 8.15am) : Atlanta
ISPE THAILAND AND TIPA 2017
 Venue: 1 st Floor, Convention A-B Room, Ambassador Hotel Bangkok, 171 Sukhumvit Soi 11, Bangkok 10110, Thailand (BTS Nana Station Exit 3), http://www.amtel.co.th/ (Special room rates are available for
Venue: 1 st Floor, Convention A-B Room, Ambassador Hotel Bangkok, 171 Sukhumvit Soi 11, Bangkok 10110, Thailand (BTS Nana Station Exit 3), http://www.amtel.co.th/ (Special room rates are available for
Pre-registration Exam workshop 2017
 Pre-registration Exam workshop 2017 Programme TIME ITEM PRESENTER 09:00 09:30 Welcome and SAPC presentation SAPC (30 min) 09:30 10:30 Examination Paper 1 Facilitator (60 min) 10:30-10:45 SHORT BREAK 15
Pre-registration Exam workshop 2017 Programme TIME ITEM PRESENTER 09:00 09:30 Welcome and SAPC presentation SAPC (30 min) 09:30 10:30 Examination Paper 1 Facilitator (60 min) 10:30-10:45 SHORT BREAK 15
MY PUBLIC ACCOUNTANT PRACTICE PROGRAM Preparation for practice success
 MY PUBLIC ACCOUNTANT PRACTICE PROGRAM 2017 Preparation for practice success tools for success The My Public Accountant (MPA) Practice Program is a fully interactive workshop which provides you with the
MY PUBLIC ACCOUNTANT PRACTICE PROGRAM 2017 Preparation for practice success tools for success The My Public Accountant (MPA) Practice Program is a fully interactive workshop which provides you with the
Advances in Well Testing and Wireline Formation Testing
 Advances in Well Testing and Wireline Formation Testing 8 9 December 2014 Kuala Lumpur Convention Center, Kuala Lumpur, Malaysia Why should you attend? To review and appreciate recent advances in the subject
Advances in Well Testing and Wireline Formation Testing 8 9 December 2014 Kuala Lumpur Convention Center, Kuala Lumpur, Malaysia Why should you attend? To review and appreciate recent advances in the subject
MEMBERS CIRCULAR 12 February 2015 Ref 003/02/15
 1 MEMBERS CIRCULAR 12 February 2015 Ref 003/02/15 Dear Members CERTIFICATION COURSE IN SHOPPING MALL MANAGEMENT: Marketing and Leasing Administration Operations and Maintenance Greetings! PPK Malaysia
1 MEMBERS CIRCULAR 12 February 2015 Ref 003/02/15 Dear Members CERTIFICATION COURSE IN SHOPPING MALL MANAGEMENT: Marketing and Leasing Administration Operations and Maintenance Greetings! PPK Malaysia
The Project Management Professional (PMP) Examination Preparatory Course
 An ISO 9001:2015 & ISO 29990:2010 Certified Company This course is registered with the Project Management Institute (PMI) R.E.P. Program* The Project Management Professional (PMP) 17-21 Sep 2017 Dubai,
An ISO 9001:2015 & ISO 29990:2010 Certified Company This course is registered with the Project Management Institute (PMI) R.E.P. Program* The Project Management Professional (PMP) 17-21 Sep 2017 Dubai,
Sample Site Selection Locations for RABS and Isolators
 Microrite, Inc. brings you this unique learning experience in Sample Site Selection Locations for RABS and Isolator; Part of Microrite s step-by-step webinar series. Sample Site Selection Locations for
Microrite, Inc. brings you this unique learning experience in Sample Site Selection Locations for RABS and Isolator; Part of Microrite s step-by-step webinar series. Sample Site Selection Locations for
Industry Guidelines for Computerized Systems Validation (GAMP, PDA Technical Reports)
 Training Course Computerized System Validation in the Pharmaceutical Industry Istanbul, 16-17 January 2003 Industry Guidelines for Computerized Systems Validation (GAMP, PDA Technical Reports) Wolfgang
Training Course Computerized System Validation in the Pharmaceutical Industry Istanbul, 16-17 January 2003 Industry Guidelines for Computerized Systems Validation (GAMP, PDA Technical Reports) Wolfgang
Non-Viable Particle Monitoring of Cleanrooms - Periodic and Continuous Monitoring
 Microrite, Inc. brings you this unique learning experience in Non-Viable Particle Monitoring of Cleanrooms - Periodic and Continuous Monitoring; Part of Microrite s step-by-step webinar series. Non-Viable
Microrite, Inc. brings you this unique learning experience in Non-Viable Particle Monitoring of Cleanrooms - Periodic and Continuous Monitoring; Part of Microrite s step-by-step webinar series. Non-Viable
PMP Exam Preparation Training in Dubai. The Project Management Professional (PMP) September 2018 Dubai, United Arab Emirates
 An ISO 9001:2015 & ISO 29990:2010 Certified Company 17-SEP-17 The PMI Registered Education Provider logo and PMP are registered marks of the Project Management Institute, Inc. PMP Exam Preparation The
An ISO 9001:2015 & ISO 29990:2010 Certified Company 17-SEP-17 The PMI Registered Education Provider logo and PMP are registered marks of the Project Management Institute, Inc. PMP Exam Preparation The
Global Impact for a Safe and Sustainable Future
 ISO 9001:2015 Quality Management System Auditor Transition (IRCA) 2 800 13-14 - 7-8 - 6-7 - ISO 9001:2015 Quality Management System Foundation 2 700-11-12 5-6 - 5-6 ISO 9001:2015 Quality Management System
ISO 9001:2015 Quality Management System Auditor Transition (IRCA) 2 800 13-14 - 7-8 - 6-7 - ISO 9001:2015 Quality Management System Foundation 2 700-11-12 5-6 - 5-6 ISO 9001:2015 Quality Management System
Workshop on Design for Compliance to IEC (3rd Edition) and EMC Standards & Requirements. 14 th - 15 th May 2015 TÜV SÜD
 Workshop on Design for Compliance to IEC 61010-1 (3rd Edition) and EMC Standards & Requirements 14 th - 15 th May 2015 TÜV SÜD Introduction The introduction of risk assessment to IEC 61010-1 (3rd Edition)
Workshop on Design for Compliance to IEC 61010-1 (3rd Edition) and EMC Standards & Requirements 14 th - 15 th May 2015 TÜV SÜD Introduction The introduction of risk assessment to IEC 61010-1 (3rd Edition)
Program Management Professionals (PgMP)
 An ISO 9001:2015 & ISO 29990:2010 Certified Company PgMP and PMI are registered marks of the Project of the Project Management Institute Inc. Program Management Examination Preparatory Course 02-06 December
An ISO 9001:2015 & ISO 29990:2010 Certified Company PgMP and PMI are registered marks of the Project of the Project Management Institute Inc. Program Management Examination Preparatory Course 02-06 December
October 21-23, 2013 Holly Springs, NC
 NEBB s Cleanroom Performance Testing Certified Professional Seminar and Exam October 21-23, 2013 Holly Springs, NC Introduction: NEBB, in continuance of its policy to enhance education and training within
NEBB s Cleanroom Performance Testing Certified Professional Seminar and Exam October 21-23, 2013 Holly Springs, NC Introduction: NEBB, in continuance of its policy to enhance education and training within
FMM Institute. Centre for Professional Development
 FMM Institute Centre for Professional Development In-House Training Available This course provides participants with the concepts and skills to use Adobe Photoshop CS effectively. Participants will get
FMM Institute Centre for Professional Development In-House Training Available This course provides participants with the concepts and skills to use Adobe Photoshop CS effectively. Participants will get
Advanced Systems, Inc. Registration Form. (Please complete all information)
 Registration Form (Please complete all information) To register by email, send a note to Advanced Systems, Inc., (mmoore@advancedsys.com) and indicate that you would like to register for the Field Sampling
Registration Form (Please complete all information) To register by email, send a note to Advanced Systems, Inc., (mmoore@advancedsys.com) and indicate that you would like to register for the Field Sampling
G7 Expert Certification (Sheetfed Offset Printing) (Location: Shanghai)
 G7 Expert Certification (Sheetfed Offset Printing) (Location: Shanghai) G7 Expert: G7 Expert Individual or independent technical consultants, or technical professional in field of color management, process
G7 Expert Certification (Sheetfed Offset Printing) (Location: Shanghai) G7 Expert: G7 Expert Individual or independent technical consultants, or technical professional in field of color management, process
Centre Approval Application (International Qualifications)
 Centre Approval Application (International Qualifications) This form must be completed by: Centres seeking approval from EDI to offer LCCI international qualifications and examinations Existing centres
Centre Approval Application (International Qualifications) This form must be completed by: Centres seeking approval from EDI to offer LCCI international qualifications and examinations Existing centres
EMC & RF Workshop. 9 th November 2017 TÜV SÜD
 EMC & RF Workshop 9 th November 2017 TÜV SÜD Introduction Dealing with electromagnetic interference (EMI) can be one of the most problematic challenges in the design and development of electrical and electronic
EMC & RF Workshop 9 th November 2017 TÜV SÜD Introduction Dealing with electromagnetic interference (EMI) can be one of the most problematic challenges in the design and development of electrical and electronic
IN-DEPTH UNDERSTANDING OF HVAC EQUIPMENT & SYSTEM SPECIFICATIONS AND PERFORMANCE 11 & 12 JULY 2016 WISMA IEM
 IEM TRAINING CENTRE SDN. BHD. (127273-K) No. 33-1A (1 st Floor), Jalan 52/18, P.O. Box 224 (Jalan Sultan P.O.) 46720 Petaling Jaya, Selangor Darul Ehsan (GST ID No. 001525395456) Telephone No. 03-79586851
IEM TRAINING CENTRE SDN. BHD. (127273-K) No. 33-1A (1 st Floor), Jalan 52/18, P.O. Box 224 (Jalan Sultan P.O.) 46720 Petaling Jaya, Selangor Darul Ehsan (GST ID No. 001525395456) Telephone No. 03-79586851
SAVE THE DATE MARCH Basics of St. Charles, MO CLICK HERE to register online!
 SAVE THE DATE MARCH 22-24 2016 St. Charles, MO 63304 Basics of TABLET MANUFACTURING & TROUBLESHOOTING By attending this course you will enhance your knowledge of maintenance and troubleshooting procedures
SAVE THE DATE MARCH 22-24 2016 St. Charles, MO 63304 Basics of TABLET MANUFACTURING & TROUBLESHOOTING By attending this course you will enhance your knowledge of maintenance and troubleshooting procedures
Scout Association of Hong Kong
 JUNE 2018 Scout Association of Hong Kong Venue Booking https://booking.scout.org.hk/ Index Content Page 1. Register Account 2 2. Apply Venue Booking 10 3. View Booking Application 22 4. Confirm Booking
JUNE 2018 Scout Association of Hong Kong Venue Booking https://booking.scout.org.hk/ Index Content Page 1. Register Account 2 2. Apply Venue Booking 10 3. View Booking Application 22 4. Confirm Booking
Danfoss Turbocor Learning Center
 ue 2011 Danfoss Turbocor Learning Center TRAINING CATALOG 2 Table of Contents 1. Introduction... 4 2. Danfoss Turbocor Learning Center... 4 3. Danfoss Turbocor Instructors Profile... 5 4. Registration...
ue 2011 Danfoss Turbocor Learning Center TRAINING CATALOG 2 Table of Contents 1. Introduction... 4 2. Danfoss Turbocor Learning Center... 4 3. Danfoss Turbocor Instructors Profile... 5 4. Registration...
Danfoss Turbocor Learning Center
 Danfoss Turbocor Learning Center TRAINING CATALOG A subsidiary of Danfoss A/S 2 Table of Contents 1. Introduction... 5 2. Danfoss Turbocor Learning Center... 5 3. Danfoss Turbocor Instructors Profile...
Danfoss Turbocor Learning Center TRAINING CATALOG A subsidiary of Danfoss A/S 2 Table of Contents 1. Introduction... 5 2. Danfoss Turbocor Learning Center... 5 3. Danfoss Turbocor Instructors Profile...
The forum will cover the key legislative amendments in the Companies (Amendment) Act 2014 and the practical applications and key filing requirements.
 The forum will cover the key legislative amendments in the Companies (Amendment) Act 2014 and the practical applications and key filing requirements. PROGRAMME AT A GLANCE Presentation on Key Legislative
The forum will cover the key legislative amendments in the Companies (Amendment) Act 2014 and the practical applications and key filing requirements. PROGRAMME AT A GLANCE Presentation on Key Legislative
Medical Device Seminar Bringing your medical products to United States of America (USA)
 Medical Device Seminar Bringing your medical products to United States of America (USA) 4 th June 2015 TÜV SÜD Introduction Healthcare improvements rely on an innovative and diverse medical device sector
Medical Device Seminar Bringing your medical products to United States of America (USA) 4 th June 2015 TÜV SÜD Introduction Healthcare improvements rely on an innovative and diverse medical device sector
Cleanroom Performance
 Cleanroom Performance Testing (CPT) Seminar March 4-6, 2019 Vancouver, WA Location: This Seminar is Designed for: This seminar is intended for individuals with a minimum of two years of cleanroom testing
Cleanroom Performance Testing (CPT) Seminar March 4-6, 2019 Vancouver, WA Location: This Seminar is Designed for: This seminar is intended for individuals with a minimum of two years of cleanroom testing
What Disinfectants Do You Need - FDA or EPA Registered
 Microrite, Inc. brings you this unique learning experience in Understanding What Disinfectants Do You Need-FDA or EPA Registered; Part of Microrite s step-by-step webinar series. What Disinfectants Do
Microrite, Inc. brings you this unique learning experience in Understanding What Disinfectants Do You Need-FDA or EPA Registered; Part of Microrite s step-by-step webinar series. What Disinfectants Do
The definitive guide to communication design
 The definitive guide to communication design DURATION 5 months, 35 Lessons DATES 4 December 2018 30 April 2019 TIME VENUE Tuesdays & Thursdays 7.00 PM 9.00 PM Level 10, Block 2, VSQ @ PJ City Centre, Jalan
The definitive guide to communication design DURATION 5 months, 35 Lessons DATES 4 December 2018 30 April 2019 TIME VENUE Tuesdays & Thursdays 7.00 PM 9.00 PM Level 10, Block 2, VSQ @ PJ City Centre, Jalan
FMM Institute. Centre for Professional Development
 FMM Institute Centre for Professional Development In-House Training Available This course provides participants with the concepts and skills to use Adobe Photoshop CS effectively. Participants will get
FMM Institute Centre for Professional Development In-House Training Available This course provides participants with the concepts and skills to use Adobe Photoshop CS effectively. Participants will get
MEMBERS CIRCULAR 13 June 2016 Ref 0028/06/16
 1 MEMBERS CIRCULAR 13 June 2016 Ref 0028/06/16 Dear Members CERTIFICATION COURSE IN SHOPPING MALL MANAGEMENT: Marketing and Leasing Operations and Maintenance Greetings! PPK Malaysia is pleased to announce
1 MEMBERS CIRCULAR 13 June 2016 Ref 0028/06/16 Dear Members CERTIFICATION COURSE IN SHOPPING MALL MANAGEMENT: Marketing and Leasing Operations and Maintenance Greetings! PPK Malaysia is pleased to announce
PROFIBUS TRAINING COURSE REGISTRATION FORMS
 PROFIBUS TRAINING COURSE REGISTRATION FORMS PLEASE FAX COMPLETED REGISTRATION FORMS TO +27 31 767-2627 Instructions: 1. Read the information regarding course starting times, venues, food and accommodation
PROFIBUS TRAINING COURSE REGISTRATION FORMS PLEASE FAX COMPLETED REGISTRATION FORMS TO +27 31 767-2627 Instructions: 1. Read the information regarding course starting times, venues, food and accommodation
Touchstone Technologies, Inc. Course Catalog February 2017
 Touchstone Technologies, Inc. Course Catalog February 2017 Angela Bazigos ANGELA BAZIGOS 1 HR Courses Course Duration Audience Speaker 1. Basics of Project Management Webinar 90 mins Management Bazigos
Touchstone Technologies, Inc. Course Catalog February 2017 Angela Bazigos ANGELA BAZIGOS 1 HR Courses Course Duration Audience Speaker 1. Basics of Project Management Webinar 90 mins Management Bazigos
GUIDELINES TO HDC LICENSING PROGRAM
 Version 2.0 GUIDELINES TO HDC LICENSING PROGRAM Copyright HDC 2015 TABLE OF CONTENT INTRODUCTION HDC HALAL TRAINING LICENSING PROGRAM LICENSING REQUIREMENT LICENSING:ROLE AND RESPONSIBILITY LICENSING FEE
Version 2.0 GUIDELINES TO HDC LICENSING PROGRAM Copyright HDC 2015 TABLE OF CONTENT INTRODUCTION HDC HALAL TRAINING LICENSING PROGRAM LICENSING REQUIREMENT LICENSING:ROLE AND RESPONSIBILITY LICENSING FEE
PMP Certification Preparatory Course
 PMP Certification Preparatory Course Why Project Management Professional (PMP ) Certification? In today's flexible organization, the boundaries between functions are becoming less defined. You are most
PMP Certification Preparatory Course Why Project Management Professional (PMP ) Certification? In today's flexible organization, the boundaries between functions are becoming less defined. You are most
Program Management Professionals (PgMP)
 An ISO 9001:2015 & ISO 29990:2010 Certified Company Program Management 10-14 Dec 2017 Dubai, United Arab Emirates 27-JUN-17 Program Management Why Choose this Training Course? Program management is the
An ISO 9001:2015 & ISO 29990:2010 Certified Company Program Management 10-14 Dec 2017 Dubai, United Arab Emirates 27-JUN-17 Program Management Why Choose this Training Course? Program management is the
October 9-11, 2012 Lenexa, KS
 NEBB s Cleanroom Performance Testing Certified Professional Seminar and Exam October 9-11, 2012 Lenexa, KS Introduction: NEBB, in continuance of its policy to enhance education and training within the
NEBB s Cleanroom Performance Testing Certified Professional Seminar and Exam October 9-11, 2012 Lenexa, KS Introduction: NEBB, in continuance of its policy to enhance education and training within the
Excel VBA for Absolute Beginners
 Applied Technology Group Sdn Bhd (1012178-W) W-5-3, Subang Square Business Centre, Jalan SS15/4G, 47500 Subang Jaya, Selangor, Malaysia. Tel: (+603) 5634 7905 Fax: (+603) 5637 9945 Email: admin@apptechgroups.net
Applied Technology Group Sdn Bhd (1012178-W) W-5-3, Subang Square Business Centre, Jalan SS15/4G, 47500 Subang Jaya, Selangor, Malaysia. Tel: (+603) 5634 7905 Fax: (+603) 5637 9945 Email: admin@apptechgroups.net
GUIDELINES FOR REGISTRATION AS PROFESSIONAL ENGINEER Section 24A ( 1 ) of the Registration of Engineers Act 1967 (Revised 2015)
 GUIDELINES FOR REGISTRATION AS PROFESSIONAL ENGINEER Section 24A ( 1 ) of the Registration of Engineers Act 1967 (Revised 2015) No person shall employ a person, sole proprietorship, partnership or body
GUIDELINES FOR REGISTRATION AS PROFESSIONAL ENGINEER Section 24A ( 1 ) of the Registration of Engineers Act 1967 (Revised 2015) No person shall employ a person, sole proprietorship, partnership or body
FORM A4 APPLICATION FOR REGISTRATION AS A PROFESSIONAL ENGINEER Section 24A (1) of the Registration of Engineers Act 1967 (Revised 2015)
 Rev. No.: 1 FORM A4 APPLICATION FOR REGISTRATION AS A PROFESSIONAL ENGINEER Section 24A (1) of the Registration of Engineers Act 1967 (Revised 2015) (To be completed by the Applicant in BLOCK LETTERS)
Rev. No.: 1 FORM A4 APPLICATION FOR REGISTRATION AS A PROFESSIONAL ENGINEER Section 24A (1) of the Registration of Engineers Act 1967 (Revised 2015) (To be completed by the Applicant in BLOCK LETTERS)
CERTIFICATE IN LUXEMBOURG COMPANY SECRETARIAL & GOVERNANCE PRACTICE
 CERTIFICATE IN LUXEMBOURG COMPANY SECRETARIAL & GOVERNANCE PRACTICE POLICY ILA asbl 19, rue de Bitbourg L-1273 Luxembourg TABLE OF CONTENTS Program Entry 3 Eligibility criteria 3 Training program 4 Application
CERTIFICATE IN LUXEMBOURG COMPANY SECRETARIAL & GOVERNANCE PRACTICE POLICY ILA asbl 19, rue de Bitbourg L-1273 Luxembourg TABLE OF CONTENTS Program Entry 3 Eligibility criteria 3 Training program 4 Application
Cleanroom Performance
 Cleanroom Performance Testing (CPT) Seminar October 7-9, 2019 Gaithersburg, MD Location: This Seminar is Designed for: This seminar is intended for individuals with a minimum of two years of cleanroom
Cleanroom Performance Testing (CPT) Seminar October 7-9, 2019 Gaithersburg, MD Location: This Seminar is Designed for: This seminar is intended for individuals with a minimum of two years of cleanroom
Green Building/LEED AP Building Design & Construction Exam Preparation
 Green Building/LEED AP Building Design & Construction Exam Preparation Be among the first to master the new green building principles in the region February 16 17 and May 11 12, 2011 American University
Green Building/LEED AP Building Design & Construction Exam Preparation Be among the first to master the new green building principles in the region February 16 17 and May 11 12, 2011 American University
Cleanroom Microbiology for Non-Microbiologists
 Microrite, Inc. brings you this unique learning experience in understanding cleanroom microbiology concepts and techniques Cleanroom Microbiology for Non-Microbiologists A course designed specifically
Microrite, Inc. brings you this unique learning experience in understanding cleanroom microbiology concepts and techniques Cleanroom Microbiology for Non-Microbiologists A course designed specifically
POST MASTER S CERTIFICATE IN VALIDATION SCIENCE
 POST MASTER S CERTIFICATE IN VALIDATION SCIENCE BACKGROUND Temple University s School of Pharmacy continues to be the leader in providing outstanding graduate-level courses in Regulatory Affairs and Quality
POST MASTER S CERTIFICATE IN VALIDATION SCIENCE BACKGROUND Temple University s School of Pharmacy continues to be the leader in providing outstanding graduate-level courses in Regulatory Affairs and Quality
ICA CERTIFICATE IN COMPLIANCE. Leading Excellence in Banking. Wilmington Risk & Compliance
 ICA CERTIFICATE IN COMPLIANCE Leading Excellence in Banking Wilmington Risk & Compliance Global Reach BIBF plays a vital role in the training and development of human capital in the Middle East and North
ICA CERTIFICATE IN COMPLIANCE Leading Excellence in Banking Wilmington Risk & Compliance Global Reach BIBF plays a vital role in the training and development of human capital in the Middle East and North
Process Safety Management Training
 IHS OPERATIONAL EXCELLENCE & RISK MANAGEMENT Process Safety Management Training Leverage state-of-the-art resources and practical knowhow to learn about proven management system concepts and techniques.
IHS OPERATIONAL EXCELLENCE & RISK MANAGEMENT Process Safety Management Training Leverage state-of-the-art resources and practical knowhow to learn about proven management system concepts and techniques.
TRUSTEE EXPENSE REIMBURSEMENT FORM
 5/7/2018 Task Authorization Mobile Support Sign Out ROCKY VIEW SCHOOLS Expense Reimbursement Authorization Review all sections of the form and then complete the last section at the bottom of the page.
5/7/2018 Task Authorization Mobile Support Sign Out ROCKY VIEW SCHOOLS Expense Reimbursement Authorization Review all sections of the form and then complete the last section at the bottom of the page.
Professional Evaluation and Certification Board Frequently Asked Questions
 Professional Evaluation and Certification Board Frequently Asked Questions 1. About PECB... 2 2. General... 2 3. PECB Official Training Courses... 4 4. Course Registration... 5 5. Certification... 5 6.
Professional Evaluation and Certification Board Frequently Asked Questions 1. About PECB... 2 2. General... 2 3. PECB Official Training Courses... 4 4. Course Registration... 5 5. Certification... 5 6.
Risk Based EBRS Implementation using GAMP 5
 Risk Based EBRS Implementation using GAMP 5 Gilberto Rossi 1 ETIF Argentina Risk-based EBRS Implementation using GAMP 5 Risk management - why do we need it? Taking risk managing risk and mismanaging risk
Risk Based EBRS Implementation using GAMP 5 Gilberto Rossi 1 ETIF Argentina Risk-based EBRS Implementation using GAMP 5 Risk management - why do we need it? Taking risk managing risk and mismanaging risk
NEWS ISSUE # 26
 PRESIDENT S MESSAGE Dear Members, Time really flies and soon 2012 will be over and we will be welcoming the New Year 2013. ISACA Malaysia Chapter has kick started the year end celebration by having the
PRESIDENT S MESSAGE Dear Members, Time really flies and soon 2012 will be over and we will be welcoming the New Year 2013. ISACA Malaysia Chapter has kick started the year end celebration by having the
BCS, The Chartered Institute for IT. Exam Cancellation and Rescheduling Policy. September 2017
 BCS, The Chartered Institute for IT Exam Cancellation and Rescheduling Policy September 2017 CONTENTS 1. SCOPE... 2 2. EXAMINATION CANCELLATION POLICY... 2 3. RESCHEDULING EXAMINATIONS... 2 4. RESCHEDULING
BCS, The Chartered Institute for IT Exam Cancellation and Rescheduling Policy September 2017 CONTENTS 1. SCOPE... 2 2. EXAMINATION CANCELLATION POLICY... 2 3. RESCHEDULING EXAMINATIONS... 2 4. RESCHEDULING
Oil & Gas Industry Quality Management System Auditor/ Lead Auditor Training
 An Intensive 2-Week Training Course Oil & Gas Industry Quality Management System Auditor/ Lead Auditor Training 27 Oct - 07 Nov 2019, Dubai 24-OCT-18 This course is Designed, Developed, and will be Delivered
An Intensive 2-Week Training Course Oil & Gas Industry Quality Management System Auditor/ Lead Auditor Training 27 Oct - 07 Nov 2019, Dubai 24-OCT-18 This course is Designed, Developed, and will be Delivered
30 September - 3 October 2018* Kempinski Hotel, Mall of the Emirates, Dubai, UAE
 30 September - 3 October 2018* OFFER 32 FOR Save up to $1000 by booking early! www.institutelm.com Who Should Attend? This cutting-edge course has been tailor made for professionals in the Middle East
30 September - 3 October 2018* OFFER 32 FOR Save up to $1000 by booking early! www.institutelm.com Who Should Attend? This cutting-edge course has been tailor made for professionals in the Middle East
September 28 th -30 th, 2016 Kuala Lumpur, Malaysia
 Certification in ISO 31000 (3 day masterclasses) Become a certified ISO 31000 risk management professional September 28 th -30 th, 2016 Kuala Lumpur, Malaysia Contact: Ng Tien Seong, Email: ngseannts@gmail.com
Certification in ISO 31000 (3 day masterclasses) Become a certified ISO 31000 risk management professional September 28 th -30 th, 2016 Kuala Lumpur, Malaysia Contact: Ng Tien Seong, Email: ngseannts@gmail.com
Financial Planning Institute of Southern Africa SETTING THE STANDARD. Continuous Professional Development (Cpd) Policy
 FPI FPI Financial Planning Institute of Southern Africa SETTING THE STANDARD Continuous Professional Development (Cpd) Policy Table of Contents Definitions 3-4 Introduction 4 Primary Responsibility 5 Mandatory
FPI FPI Financial Planning Institute of Southern Africa SETTING THE STANDARD Continuous Professional Development (Cpd) Policy Table of Contents Definitions 3-4 Introduction 4 Primary Responsibility 5 Mandatory
Training Programmes Sri Lanka Accreditation Board for Conformity Assessment
 Training Programmes -2018 Sri Lanka Accreditation Board for Conformity Assessment General Instructions: All interested parties are requested to fill separate reservation form for each and submit to the
Training Programmes -2018 Sri Lanka Accreditation Board for Conformity Assessment General Instructions: All interested parties are requested to fill separate reservation form for each and submit to the
2019 Educational Courses Catalog
 2019 Educational Courses Catalog (STULZ USA),, U.S.A. Phone: +888.529.1266 Email: STULZTraining@stulz-ats.com www.stulz.com TRG-C2019 Rev. A 10/27/17 Important Information These training sessions alone
2019 Educational Courses Catalog (STULZ USA),, U.S.A. Phone: +888.529.1266 Email: STULZTraining@stulz-ats.com www.stulz.com TRG-C2019 Rev. A 10/27/17 Important Information These training sessions alone
ISPE Thailand 3 rd Seminar 2018
 ISPE Thailand 3 rd Seminar 2018 Contamination Risk Assessment in Pharmaceutical Cleanrooms & How to use Airflow Visualization (Smoke Studies) to troubleshoot Cleanroom Contamination issues 29 th 30 th
ISPE Thailand 3 rd Seminar 2018 Contamination Risk Assessment in Pharmaceutical Cleanrooms & How to use Airflow Visualization (Smoke Studies) to troubleshoot Cleanroom Contamination issues 29 th 30 th
Regulatory Compliance (Insurance)
 Regulatory Compliance (Insurance) Regulatory Compliance (Insurance) There is no denying that experience matched with the right training & education will help you achieve your goals and advance your career.
Regulatory Compliance (Insurance) Regulatory Compliance (Insurance) There is no denying that experience matched with the right training & education will help you achieve your goals and advance your career.
Advanced Systems, Inc. Course. Registration Form. (Please complete all information)
 MARCH 9 TO 11, 2015 Registration Form (Please complete all information) To register on-line for the Environmental Laboratory Assessments Basic Assessor Training. Course offered in Olathe, Kansas on March
MARCH 9 TO 11, 2015 Registration Form (Please complete all information) To register on-line for the Environmental Laboratory Assessments Basic Assessor Training. Course offered in Olathe, Kansas on March
Be PCI compliant. Secure your customers credit card information.
 Be PCI compliant. Secure your customers credit card information. Risks in e-business Today In this generation, many proprietors are moving on to e-commerce and venturing into selling (or buying) electronically.
Be PCI compliant. Secure your customers credit card information. Risks in e-business Today In this generation, many proprietors are moving on to e-commerce and venturing into selling (or buying) electronically.
WASTEWATER COLLECTION LEVEL 1 TRAINING AND CERTIFICATION EXAM FOR WASTEWATER OPERATORS
 NATIONAL TRIBAL WATER AND WASTEWATER OPERATOR CERTIFICATION PROGRAM TRAINING ANNOUNCEMENT WASTEWATER COLLECTION LEVEL 1 TRAINING AND CERTIFICATION EXAM FOR WASTEWATER OPERATORS Hosted by The University
NATIONAL TRIBAL WATER AND WASTEWATER OPERATOR CERTIFICATION PROGRAM TRAINING ANNOUNCEMENT WASTEWATER COLLECTION LEVEL 1 TRAINING AND CERTIFICATION EXAM FOR WASTEWATER OPERATORS Hosted by The University
NEBB FUME HOOD PERFORMANCE TESTING SEMINAR JUNE 5 & 6, 2017
 NEBB FUME HOOD PERFORMANCE TESTING SEMINAR JUNE 5 & 6, 2017 FHT SEMINAR DATES MONDAY & TUESDAY, JUNE 5 & 6, 2017 SEMINAR REGISTRATION DEADLINE MONDAY, MAY 22, 2017 FHT CP PAPER-BASED EXAM DAY WEDNESDAY,
NEBB FUME HOOD PERFORMANCE TESTING SEMINAR JUNE 5 & 6, 2017 FHT SEMINAR DATES MONDAY & TUESDAY, JUNE 5 & 6, 2017 SEMINAR REGISTRATION DEADLINE MONDAY, MAY 22, 2017 FHT CP PAPER-BASED EXAM DAY WEDNESDAY,
Water Distribution Level 1 January 8 12, 2018
 Water Distribution Level 1 Operator Training and Certi ication Exam January 8 12, 2018 Hosted by: Lac Courte Oreilles Public Works Lac Courte Oreilles Ojibwa Community College Hayward, Wisconsin Includes:
Water Distribution Level 1 Operator Training and Certi ication Exam January 8 12, 2018 Hosted by: Lac Courte Oreilles Public Works Lac Courte Oreilles Ojibwa Community College Hayward, Wisconsin Includes:
Makai Lay Level 1 Training Course Darlington, UK August 2014
 Darlington, UK August 2014 Ocean-Tec Systems Ltd, Office Suites 6 & 7, Lakeland Estates Business Park, Faverdale North Darlington, England, DL3 0PX Tel: +44 (0) 845 094 6035 Fax: +44 (0) 845 094 6039 Email:
Darlington, UK August 2014 Ocean-Tec Systems Ltd, Office Suites 6 & 7, Lakeland Estates Business Park, Faverdale North Darlington, England, DL3 0PX Tel: +44 (0) 845 094 6035 Fax: +44 (0) 845 094 6039 Email:
Master the implementation and management of a Cybersecurity Program based on ISO/IEC 27032
 ISO/IEC 27032 Lead Manager 23rd - 27th October 2017 Hilton Hotel, Sandton, Johannesburg Master the implementation and management of a Program based on ISO/IEC 27032 Why should you attend? Manager training
ISO/IEC 27032 Lead Manager 23rd - 27th October 2017 Hilton Hotel, Sandton, Johannesburg Master the implementation and management of a Program based on ISO/IEC 27032 Why should you attend? Manager training
PECB Certified ISO/IEC Lead Auditor
 When Recognition Matters Lead Auditor 25-29 March 2018 Master the Audit of Rainbow Towers Hotel, Harare Systems (ISMS) based on $1250.00 Who should attend? - Auditors seeking to perform and lead System
When Recognition Matters Lead Auditor 25-29 March 2018 Master the Audit of Rainbow Towers Hotel, Harare Systems (ISMS) based on $1250.00 Who should attend? - Auditors seeking to perform and lead System
Rouge Formation and Control Strategies in WFI Water
 Microrite, Inc. brings you this unique learning experience in Rouge Formation and Control Strategies in WFI Water; Part of Microrite s step-by-step webinar series. Rouge Formation and Control Strategies
Microrite, Inc. brings you this unique learning experience in Rouge Formation and Control Strategies in WFI Water; Part of Microrite s step-by-step webinar series. Rouge Formation and Control Strategies
Radware. Training Policies V001.70
 Radware Training Policies V001.70 TABLE OF CONTENTS SCOPE & DISCLAIMER...3 QUESTIONS?...3 TRAINING...4 OVERVIEW... 4 DELIVERY OPTIONS... 4 TRAINING ENROLLMENT & REGISTRATION... 5 COURSE EVALUATION... 5
Radware Training Policies V001.70 TABLE OF CONTENTS SCOPE & DISCLAIMER...3 QUESTIONS?...3 TRAINING...4 OVERVIEW... 4 DELIVERY OPTIONS... 4 TRAINING ENROLLMENT & REGISTRATION... 5 COURSE EVALUATION... 5
Vehicle Network Seminar Series
 CAN and Higher Layer Protocols One of our most popular seminars, this course is relevant for passenger cars and light duty trucks. CAN (Controller Area Network) is the worldwide standard for automotive
CAN and Higher Layer Protocols One of our most popular seminars, this course is relevant for passenger cars and light duty trucks. CAN (Controller Area Network) is the worldwide standard for automotive
CFA CHARTER FINANCIAL ANALYST. The time is now to build a better tomorrow. IMA Educational Partner in KSA
 CFA CHARTER FINANCIAL ANALYST The time is now to build a better tomorrow IMA Educational Partner in KSA THE CFA CHARTER The Chartered Financial Analyst (CFA) credential is the professional standard of
CFA CHARTER FINANCIAL ANALYST The time is now to build a better tomorrow IMA Educational Partner in KSA THE CFA CHARTER The Chartered Financial Analyst (CFA) credential is the professional standard of
Training courses 2018
 Training courses 2018 The UK s leading specialist air conditioning and refrigeration training provider Get ahead, develop your skills and knowledge Professional training and practical instruction on latest
Training courses 2018 The UK s leading specialist air conditioning and refrigeration training provider Get ahead, develop your skills and knowledge Professional training and practical instruction on latest
INFORMATION SECURITY MANAGEMENT
 ISMS (ISO/IEC 27001:2005 to ISO/IEC 27001:2013) Transition Training Course (A17700) Two (2) Days It is recommended for ISMS registered Provisional Auditors, Auditors, Lead Auditors, Principal Auditors
ISMS (ISO/IEC 27001:2005 to ISO/IEC 27001:2013) Transition Training Course (A17700) Two (2) Days It is recommended for ISMS registered Provisional Auditors, Auditors, Lead Auditors, Principal Auditors
AROC Online Services (AOS) User Manual
 AROC Online Services (AOS) User Manual Version 5.0 (September 2014) FIM/WeeFIM (Workshops) Contents Introduction... 2 Flow chart of AOS functionality... 2 Computing requirements... 2 Session time-out...
AROC Online Services (AOS) User Manual Version 5.0 (September 2014) FIM/WeeFIM (Workshops) Contents Introduction... 2 Flow chart of AOS functionality... 2 Computing requirements... 2 Session time-out...
TRAINING CALENDAR MARCH 2018
 TRAINING CALENDAR MARCH 2018 Why Boecker? Boecker, one of the world s finest Food Safety training organizations and a registered training centre with the Royal Society for Public Health - UK, and Chartered
TRAINING CALENDAR MARCH 2018 Why Boecker? Boecker, one of the world s finest Food Safety training organizations and a registered training centre with the Royal Society for Public Health - UK, and Chartered
INFORMATION SYSTEMS EXAMINATIONS BOARD
 INFORMATION SYSTEMS EXAMINATIONS BOARD Certificate in Freedom of Information Guidelines for Candidates March 2009 ISEB/Regs&Guide/AccredV1.4 03/09 TABLE OF CONTENTS FOREWORD... 2 1 INTRODUCTION... 2 1.1
INFORMATION SYSTEMS EXAMINATIONS BOARD Certificate in Freedom of Information Guidelines for Candidates March 2009 ISEB/Regs&Guide/AccredV1.4 03/09 TABLE OF CONTENTS FOREWORD... 2 1 INTRODUCTION... 2 1.1
With the successful completion of this course the participant will be able to:
 ISO 13485:2016 INTRODUCTION COURSE COURSE DURATION: 1 DAY Course Summary: The introduction course provides the participant with an oversight on the requirements of ISO 13485:2016 standard. Our course is
ISO 13485:2016 INTRODUCTION COURSE COURSE DURATION: 1 DAY Course Summary: The introduction course provides the participant with an oversight on the requirements of ISO 13485:2016 standard. Our course is
Arctic Chiller Group Training Programs
 Arctic Chiller Group Training Programs The Arctic Chiller Group is proud to provide a training course for technicians with the necessary skills to maintain and trouble-shoot the Arctic Chiller Systems.
Arctic Chiller Group Training Programs The Arctic Chiller Group is proud to provide a training course for technicians with the necessary skills to maintain and trouble-shoot the Arctic Chiller Systems.
Investigating Microbial Data Deviations
 Microrite, Inc. brings you this unique learning experience in Investigating Microbial Data Deviations; Part of Microrite s step-by-step webinar series. Investigating Microbial Data Deviations Since the
Microrite, Inc. brings you this unique learning experience in Investigating Microbial Data Deviations; Part of Microrite s step-by-step webinar series. Investigating Microbial Data Deviations Since the
Arctic Chiller Group Training Program
 Course Description Arctic Chiller Group Training Program Maintenance Training & Certification for Service Providers This 3-day course is for service contractors who will maintain, service and troubleshoot
Course Description Arctic Chiller Group Training Program Maintenance Training & Certification for Service Providers This 3-day course is for service contractors who will maintain, service and troubleshoot
TRAINING CALENDAR FEBRUARY 2017 LEVEL 2 AWARD IN FOOD SAFETY LEVEL 3 HACCP (IN CATERING, MANUFACTURING)
 TRAINING CALENDAR FEBRUARY 2017 WHY BOECKER? Boecker, one of the world s finest Food Safety training organizations and a registered training center with the Royal Society for Public Health - UK, and Chartered
TRAINING CALENDAR FEBRUARY 2017 WHY BOECKER? Boecker, one of the world s finest Food Safety training organizations and a registered training center with the Royal Society for Public Health - UK, and Chartered
Choose only one. If you are registered in both category, PEPC & Accredited Checker, separate FORM H is required for each category.
 FORM H REGISTRATION OF ENGINEERS ACT 1967 (REVISED 2015) APPLICATION FOR RENEWAL OF REGISTRATION FOR YEAR.... Regulation 20 of the Registration of Engineers Regulations 1990 (Revised 2015) A. APPLICATION
FORM H REGISTRATION OF ENGINEERS ACT 1967 (REVISED 2015) APPLICATION FOR RENEWAL OF REGISTRATION FOR YEAR.... Regulation 20 of the Registration of Engineers Regulations 1990 (Revised 2015) A. APPLICATION
Training & Certification Guide
 Training & Certification Guide Pragmatic EA Ltd December 2010 Version 2.0.5 Part of the Pragmatic Family Cutting Architecture To the Bone Contents INTRODUCTION 3 What is PEAF 3 Aim of this document 3 Non-Commercial
Training & Certification Guide Pragmatic EA Ltd December 2010 Version 2.0.5 Part of the Pragmatic Family Cutting Architecture To the Bone Contents INTRODUCTION 3 What is PEAF 3 Aim of this document 3 Non-Commercial
