Title: Management of Hypertension and Multiple Risk Factors to Enhance Cardiovascular Health - A Feasibility Study in Singapore Polyclinics
|
|
|
- William Burns
- 6 years ago
- Views:
Transcription
1 Author s response to reviews Title: Management of Hypertension and Multiple Risk Factors to Enhance Cardiovascular Health - A Feasibility Study in Singapore Polyclinics Authors: Tazeen Jafar (tazeen.jafar@duke-nus.edu.sg) Ngiap Tan (tan.ngiap.chuan@singhealth.com.sg) John Allen (john.allen@duke-nus.edu.sg) Shreyasee Pradhan (shreyasee.pradhan@duke-nus.edu.sg) Paul Goh (paul.goh@singhealth.com.sg) Saeideh Tavajoh (saeideh.tavajoh@duke-nus.edu.sg) Fong Keng (fong.mew.keng@singhealth.com.sg) Jason Chan (jason.chan.m.h@singhealth.com.sg) Version: 2 Date: 23 Jun 2016 Author s response to reviews: Dear Editor-in-chief, We appreciate your thorough review of our manuscript (ref BHSR-D R1) entitled Management of Hypertension and Multiple Risk Factors to Enhance Cardiovascular Health - A Feasibility Study in Singapore Polyclinics has been potentially accepted for publication by BMC Health Services Research. We thank the reviewers for their valuable feedback. We have incorporated all amendments in the revised manuscript Enclosed is the revised original article, as approved by the co-authors, for consideration in your journal. Below is a summary of our point by point responses (in red color) to the comments:
2 1. Please include the registration date of the trial with the other trial information, and also state if it was retrospectively registered (registered after the 1st participant was recruited). Thank you for the suggestion. ClinicalTrials.gov registration was dated December 28, 2014 that is before the 1st participant was recruited in September Please also update the abstract on the submission system to include the trial registration details. Noted with thanks. Done. 2. We request that all supplementary information be uploaded as additional files, so please upload the appendices included in the manuscript as separate supplementary files. We uploaded appendices as in supplementary document listed in the file inventory 3. Along with submitting supplementary files, we would appreciate if you added a Œ Supplementary files¹ section after References that lists the following information for these files: File name, Title of data, Description of data. Thank you for your suggestion. We inserted the information for Supplementary files 4. We require all sections listed under Declarations in our submission guidelines ( be provided in the manuscript, so please add Œ Consent for publication¹ to the declarations. You can simply state ³Not applicable² in this section. Noted and done. 5. Please clarify the type of consent received from participants (written or verbal).
3 It was written informed consent, and has been mentioned clearly in the manuscript. 6. Please remove the figure titles/captions from within the figure files. These files should contain the image graphic only. Please place any titles/captions as text after the References. We have placed caption after the References 7. Upon uploading your revisions, please remove any tracked changes or highlighting and include only a single clean copy of the manuscript. Noted with thanksdear Editor-in-chief, We appreciate your thorough review of our manuscript (ref BHSR-D R1) entitled Management of Hypertension and Multiple Risk Factors to Enhance Cardiovascular Health - A Feasibility Study in Singapore Polyclinics has been potentially accepted for publication by BMC Health Services Research. We thank the reviewers for their valuable feedback. We have incorporated all amendments in the revised manuscript Enclosed is the revised original article, as approved by the co-authors, for consideration in your journal. Below is a summary of our point by point responses (in red color) to the comments: 1. Please include the registration date of the trial with the other trial information, and also state if it was retrospectively registered (registered after the 1st participant was recruited). Thank you for the suggestion. ClinicalTrials.gov registration was dated December 28, 2014 that is before the 1st participant was recruited in September Please also update the abstract on the submission system to include the trial registration details.
4 Noted with thanks. Done. 2. We request that all supplementary information be uploaded as additional files, so please upload the appendices included in the manuscript as separate supplementary files. We uploaded appendices as in supplementary document listed in the file inventory 3. Along with submitting supplementary files, we would appreciate if you added a Œ Supplementary files¹ section after References that lists the following information for these files: File name, Title of data, Description of data. Thank you for your suggestion. We inserted the information for Supplementary files 4. We require all sections listed under Declarations in our submission guidelines ( be provided in the manuscript, so please add Œ Consent for publication¹ to the declarations. You can simply state ³Not applicable² in this section. Noted and done. 5. Please clarify the type of consent received from participants (written or verbal). It was written informed consent, and has been mentioned clearly in the manuscript. 6. Please remove the figure titles/captions from within the figure files. These files should contain the image graphic only. Please place any titles/captions as text after the References. We have placed caption after the References
5 7. Upon uploading your revisions, please remove any tracked changes or highlighting and include only a single clean copy of the manuscript. Noted with thanks
Author s response to reviews
 Author s response to reviews Title: Cleavage of the Urokinase Receptor (upar) on Oral Cancer Cells: Regulation by Transforming Growth Factor - β1 (TGF-β1) and Potential Effects on Migration and Invasion
Author s response to reviews Title: Cleavage of the Urokinase Receptor (upar) on Oral Cancer Cells: Regulation by Transforming Growth Factor - β1 (TGF-β1) and Potential Effects on Migration and Invasion
Guide to the Distributor Zone
 Guide to the Distributor Zone Registering for a Distributor account Logging into the Distributor Zone Updating your profile Registering for a Challenge Registering a Participant Connecting a Participant
Guide to the Distributor Zone Registering for a Distributor account Logging into the Distributor Zone Updating your profile Registering for a Challenge Registering a Participant Connecting a Participant
A Step-By-Step Guide for Submitting and Accessing Manuscripts Via ejmanager
 1 A Step-By-Step Guide for Submitting and Accessing Manuscripts Via ejmanager 2 Contents of the Guide 1. Accessing the ejmanager... 3 2. Registering for a new account... 4 3. Submitting a manuscript to
1 A Step-By-Step Guide for Submitting and Accessing Manuscripts Via ejmanager 2 Contents of the Guide 1. Accessing the ejmanager... 3 2. Registering for a new account... 4 3. Submitting a manuscript to
If you log into your account directly, please go to your personal startpage. You find the submission to review under the status Active.
 1. Submission of a review If you received an email with the request to review an article, you can click on the link in the email to enter the page dedicated to reviewers (please see the next page of these
1. Submission of a review If you received an email with the request to review an article, you can click on the link in the email to enter the page dedicated to reviewers (please see the next page of these
WPSAR Online Journal System. User Guide for Reviewers
 WPSAR Online Journal System User Guide for Reviewers Table of contents Invitation to review..... Accessing the online journal system.... The review process. Step 1: Accepting the review... Step 2: Reviewer
WPSAR Online Journal System User Guide for Reviewers Table of contents Invitation to review..... Accessing the online journal system.... The review process. Step 1: Accepting the review... Step 2: Reviewer
Chapter 8. Reviewers. Overview. Review Home Page. Submissions. Figure 8.1. Reviewer Home. Figure 8.2. Active Submissions
 Chapter 8. Reviewers Overview The Reviewer is invited by email to review a submission, which includes its title and abstract, as well as the journal's URL and a username and password for the Reviewer to
Chapter 8. Reviewers Overview The Reviewer is invited by email to review a submission, which includes its title and abstract, as well as the journal's URL and a username and password for the Reviewer to
Engaged Management ReView Author Guidelines V.1.0. Friday, March 11, 2015
 Engaged Management ReView Author Guidelines V.1.0 Friday, March 11, 2015 Table of Contents Engaged Management ReView (EMR) Guidelines for Authors... 3 SECTION 1: Author How to Submit an Article... 3 SECTION
Engaged Management ReView Author Guidelines V.1.0 Friday, March 11, 2015 Table of Contents Engaged Management ReView (EMR) Guidelines for Authors... 3 SECTION 1: Author How to Submit an Article... 3 SECTION
Overview. Section 1. Reviewer Home Page. Module 3: Reviewing a Manuscript for Ethnicity & Disease
 Module 3: Reviewing a Manuscript for Ethnicity & Disease Overview The information in this document provides step-by-step instructions for conducting a review for Ethn Dis. The manuscript tracking system
Module 3: Reviewing a Manuscript for Ethnicity & Disease Overview The information in this document provides step-by-step instructions for conducting a review for Ethn Dis. The manuscript tracking system
Buck-IRB Amendment User Guide
 Office of Research Buck-IRB Amendment User Guide Office of Responsible Research Practices 1960 Kenny Road, Columbus, OH 43210-1016 Institutional Review Board General Guidance for Amendments: The Start
Office of Research Buck-IRB Amendment User Guide Office of Responsible Research Practices 1960 Kenny Road, Columbus, OH 43210-1016 Institutional Review Board General Guidance for Amendments: The Start
AUTHOR S GUIDELINES FOR MANUSCRIPT SUBMISSION
 AUTHOR S GUIDELINES FOR MANUSCRIPT SUBMISSION Registration and Login Details Registration 1. All first time users are required to Register within the system. 2. Once you are registered, you will receive
AUTHOR S GUIDELINES FOR MANUSCRIPT SUBMISSION Registration and Login Details Registration 1. All first time users are required to Register within the system. 2. Once you are registered, you will receive
JAFES Online Journal System User Guide for Authors
 JAFES Online Journal System User Guide for Authors 2017 Version 1.0 THE JOURNAL The Journal of the ASEAN Federation of Endocrine Societies (JAFES) is an OPEN ACCESS, internationally peer-reviewed, English
JAFES Online Journal System User Guide for Authors 2017 Version 1.0 THE JOURNAL The Journal of the ASEAN Federation of Endocrine Societies (JAFES) is an OPEN ACCESS, internationally peer-reviewed, English
Standard submission of a manuscript through the online OAPL system Simple Straightforward Self-explanatory
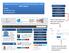 Standard submission of a manuscript through the online OAPL system Simple Straightforward Self-explanatory Choose this button and enter your username and password Choose this button and retrieve your username
Standard submission of a manuscript through the online OAPL system Simple Straightforward Self-explanatory Choose this button and enter your username and password Choose this button and retrieve your username
Manual. User Registration and Submission of Articles to the Thermal Science. Editorial Board of Thermal Science
 Manual User Registration and Submission of Articles to the Thermal Science Editorial Board of Thermal Science User registration: The Thermal Science accepts submissions of articles through on-line platform
Manual User Registration and Submission of Articles to the Thermal Science Editorial Board of Thermal Science User registration: The Thermal Science accepts submissions of articles through on-line platform
Instructions for Authors Canadian Journal of Pathology (CJP)
 Instructions for Authors Canadian Journal of Pathology (CJP) Canadian Journal of Pathology is a quarterly, peer-reviewed publication. It is published by Clockwork Communications as the agent for Canadian
Instructions for Authors Canadian Journal of Pathology (CJP) Canadian Journal of Pathology is a quarterly, peer-reviewed publication. It is published by Clockwork Communications as the agent for Canadian
This is a quick guide on how to register with the Interbull Bulletin and then submit your paper via the new Open Journal System.
 - This is a quick guide on how to register with the Interbull Bulletin and then submit your paper via the new Open Journal System. Please read through the Author Guidelines on the Bulletin website, this
- This is a quick guide on how to register with the Interbull Bulletin and then submit your paper via the new Open Journal System. Please read through the Author Guidelines on the Bulletin website, this
LOGGING INTO THE MTN WEBSITE:
 MTN PEER REVIEW WALK THROUGH FOR REVIEWERS OF THE DOCUMENT SUBMISSION PROCESS: LOGGING INTO THE MTN WEBSITE: 1) Click the login link in the top right corner of the MTN website home page (http://www.mtnstopshiv.org/).
MTN PEER REVIEW WALK THROUGH FOR REVIEWERS OF THE DOCUMENT SUBMISSION PROCESS: LOGGING INTO THE MTN WEBSITE: 1) Click the login link in the top right corner of the MTN website home page (http://www.mtnstopshiv.org/).
Appendix: Data Availability Policies & Replication Policies Time of Evaluation: January 2012
 Appendix: Data Availability Policies & Replication Policies Time of Evaluation: January 2012 Table of Contents: Data Availability Policies:... 2 1) American Economic Review:... 2 2) Journal of Political
Appendix: Data Availability Policies & Replication Policies Time of Evaluation: January 2012 Table of Contents: Data Availability Policies:... 2 1) American Economic Review:... 2 2) Journal of Political
Student Guide. Click here to log in. A: Log in
 Middlesex Online Research Ethics (MORE) Applicant Guidance Notes (Student & Staff) Note: PhD ethics applications must be submitted as Student Applications. Creating, completing and submitting a Research
Middlesex Online Research Ethics (MORE) Applicant Guidance Notes (Student & Staff) Note: PhD ethics applications must be submitted as Student Applications. Creating, completing and submitting a Research
LOUGHBOROUGH UNIVERSITY RESEARCH OFFICE STANDARD OPERATING PROCEDURE. Loughborough University (LU) Research Office SOP 1027 LU
 LOUGHBOROUGH UNIVERSITY RESEARCH OFFICE STANDARD OPERATING PROCEDURE Loughborough University (LU) Research Office SOP 1027 LU Process for Writing Study Protocols for NHS Research Sponsored by Loughborough
LOUGHBOROUGH UNIVERSITY RESEARCH OFFICE STANDARD OPERATING PROCEDURE Loughborough University (LU) Research Office SOP 1027 LU Process for Writing Study Protocols for NHS Research Sponsored by Loughborough
Uploading Files to Project MUSE May 2018
 Uploading Files to Project MUSE May 2018 Project MUSE publishes journal articles in four categories: Complete Issues Preprint Publishing Continuous Publishing Digital Only Publishing. Instructions for
Uploading Files to Project MUSE May 2018 Project MUSE publishes journal articles in four categories: Complete Issues Preprint Publishing Continuous Publishing Digital Only Publishing. Instructions for
BRANDMAN UNIVERSITY INSTITUTIONAL REVIEW BOARD Continuing Review Request/Closure Report
 BRANDMAN UNIVERSITY INSTITUTIONAL REVIEW BOARD Continuing Review Request/Closure Report Instructions: Please complete all sections of this form and submit with attached documents, forms, and/or explanations
BRANDMAN UNIVERSITY INSTITUTIONAL REVIEW BOARD Continuing Review Request/Closure Report Instructions: Please complete all sections of this form and submit with attached documents, forms, and/or explanations
Instructions on how to submit a paper in Editorial Manager
 Instructions on how to submit a paper in Editorial Manager 1. Registering your profile If you do not have a profile at the IWA congress Editorial Manager * (EM) website, you will need to create one. To
Instructions on how to submit a paper in Editorial Manager 1. Registering your profile If you do not have a profile at the IWA congress Editorial Manager * (EM) website, you will need to create one. To
Submission Guide.
 Submission Guide www.jstatsoft.org Abstract The Journal of Statistical Software (JSS) is an open-source and open-access scientific journal by the statistical software community for everybody interested
Submission Guide www.jstatsoft.org Abstract The Journal of Statistical Software (JSS) is an open-source and open-access scientific journal by the statistical software community for everybody interested
Authors Guide for the submission of articles through OJS
 REVISTA MEXICANA DE ECONOMÍA Y FINANZAS ISSN online: 2448-6795 ISSN print: 1665-5346 Authors Guide for the submission of articles through OJS Objective The purpose of this guide is to inform the authors
REVISTA MEXICANA DE ECONOMÍA Y FINANZAS ISSN online: 2448-6795 ISSN print: 1665-5346 Authors Guide for the submission of articles through OJS Objective The purpose of this guide is to inform the authors
Online Journal Submission Guide
 Online Journal Submission Guide Any manuscript submissions (Full paper for either Peer Review or Non-Peer for the traditional Journal or Special Edition; Perspective; Commentaries; or Letters to the Editor)
Online Journal Submission Guide Any manuscript submissions (Full paper for either Peer Review or Non-Peer for the traditional Journal or Special Edition; Perspective; Commentaries; or Letters to the Editor)
BRILL s Editorial Manager (EM) Manual for Authors Contents
 BRILL s Editorial Manager (EM) Manual for Authors Contents 1. Introduction... 2 2. Getting Started: Creating an Account... 2 2.1 Creating an Account Using Your ORCID Record... 3 3. Logging into EM... 4
BRILL s Editorial Manager (EM) Manual for Authors Contents 1. Introduction... 2 2. Getting Started: Creating an Account... 2 2.1 Creating an Account Using Your ORCID Record... 3 3. Logging into EM... 4
Getting Started: Log On
 Dear AJCH Authors, I would like to formally introduce and invite all our contributors to ScholarOne Manuscripts! ScholarOne Manuscripts is our new system to expedite the entire manuscript submission process.
Dear AJCH Authors, I would like to formally introduce and invite all our contributors to ScholarOne Manuscripts! ScholarOne Manuscripts is our new system to expedite the entire manuscript submission process.
Ethical Review Online Application System: User Guide
 Ethical Review Online Application System: User Guide 1 Contents Purpose of this document... 3 Accessing the system... 3 Setting internet browsers to check spelling... 4 Creating a new Application... 5
Ethical Review Online Application System: User Guide 1 Contents Purpose of this document... 3 Accessing the system... 3 Setting internet browsers to check spelling... 4 Creating a new Application... 5
Instructions for Online Manuscript Submission. Download the Author Quick Guide from
 Public Health Action International Union Against Tuberculosis and Lung Disease Health solutions for the poor Instructions for Online Manuscript Submission Go to Manuscript Central at http://mc.manuscriptcentral.com/pha
Public Health Action International Union Against Tuberculosis and Lung Disease Health solutions for the poor Instructions for Online Manuscript Submission Go to Manuscript Central at http://mc.manuscriptcentral.com/pha
Data management for funding applications. RDCS training, 17 th March, 2014
 Data management for funding applications RDCS training, 17 th March, 2014 Overview of this session: The data cycle in a clinical study What is data management Data management procedures Data management
Data management for funding applications RDCS training, 17 th March, 2014 Overview of this session: The data cycle in a clinical study What is data management Data management procedures Data management
INTRODUCTION. This guidebook provides step-by-step information on the process of submission on the new website.
 0 INTRODUCTION On March 14, 2018, Makara Human Behavior Studies in Asia (Makara Hubs- Asia) starts using a new website: http://hubsasia.ui.ac.id. Every submitted manuscript after March 14, 2018 is processed
0 INTRODUCTION On March 14, 2018, Makara Human Behavior Studies in Asia (Makara Hubs- Asia) starts using a new website: http://hubsasia.ui.ac.id. Every submitted manuscript after March 14, 2018 is processed
How to Submit a New UIW IRB Application
 How to Submit a New UIW IRB Application Step Log In Visit https://uiw.forms.ethicalreviewmanager.com/ click on at the top right corner of the page. New Users: Click on New User Fill in the applicable information
How to Submit a New UIW IRB Application Step Log In Visit https://uiw.forms.ethicalreviewmanager.com/ click on at the top right corner of the page. New Users: Click on New User Fill in the applicable information
Section.Editor s Visual Guide to Editorial Manager
 Section.Editor s Visual Guide to Editorial Manager ***Some of the Screenshots have been taken from the Editorial Manager Trial Site, so as to prevent identity of the manuscripts or the Author names from
Section.Editor s Visual Guide to Editorial Manager ***Some of the Screenshots have been taken from the Editorial Manager Trial Site, so as to prevent identity of the manuscripts or the Author names from
As an Author, your tasks include submission; submitting revised copy; copyediting; and proofreading.
 Chapter 5. Authors Overview OJS exists to serve Authors as well as journals. Not only does OJS provide an easy-to-use submission process, it can collect and disseminate key information about Authors and
Chapter 5. Authors Overview OJS exists to serve Authors as well as journals. Not only does OJS provide an easy-to-use submission process, it can collect and disseminate key information about Authors and
CIRP Life Cycle Engineering 2019 Paper Submission Process (Step-by-step with screenshot)
 CIRP Life Cycle Engineering 2019 Paper Submission Process (SIMPLIFIED VERSION) 1. Article Type: Please select SI: LCE 2019 from the dropdown menu. 2. Section/Category: Please again select SI: LCE 2019
CIRP Life Cycle Engineering 2019 Paper Submission Process (SIMPLIFIED VERSION) 1. Article Type: Please select SI: LCE 2019 from the dropdown menu. 2. Section/Category: Please again select SI: LCE 2019
MANUAL FOR Airiti ONLINE SUBMISSION AND PEER REVIEW SYSTEM
 MANUAL FOR Airiti ONLINE SUBMISSION AND PEER REVIEW SYSTEM (AUTHORS AND REVIEWERS ONLY) 2010/9 1 INDEX 1 How to Use... 4 1.1 Author... 4 1.2 Reviewer... 5 2 Application... 6 2.1 Account established by
MANUAL FOR Airiti ONLINE SUBMISSION AND PEER REVIEW SYSTEM (AUTHORS AND REVIEWERS ONLY) 2010/9 1 INDEX 1 How to Use... 4 1.1 Author... 4 1.2 Reviewer... 5 2 Application... 6 2.1 Account established by
Create a new Human Research Ethics Application in InfoEd
 Create a new Human Research Ethics Application in InfoEd IMPORTANT! A Co-Investigator or Student can create, complete and submit new human research ethics applications in InfoEd on behalf of the Chief
Create a new Human Research Ethics Application in InfoEd IMPORTANT! A Co-Investigator or Student can create, complete and submit new human research ethics applications in InfoEd on behalf of the Chief
Research Management Information System (RIMS) Human Ethics - User Guide
 Research Management Information System (RIMS) Human Ethics - User Guide Contents How to create an Initial Application to the HREC... 2 Step 1: Create a new protocol... 2 Step 2: Title... 3 Step 3: Chief
Research Management Information System (RIMS) Human Ethics - User Guide Contents How to create an Initial Application to the HREC... 2 Step 1: Create a new protocol... 2 Step 2: Title... 3 Step 3: Chief
Edit an Existing Submission in InfoEd
 Edit an Existing Submission in InfoEd 1. Locate the project in the InfoEd Chief Investigator on the project: Log into InfoEd, In the Home screen, go to My Items tab and find the record. Once the record
Edit an Existing Submission in InfoEd 1. Locate the project in the InfoEd Chief Investigator on the project: Log into InfoEd, In the Home screen, go to My Items tab and find the record. Once the record
GUIDE TO CERTIFICATION
 GUIDE TO CERTIFICATION December 2017 *Note this document is temporary, and the content will soon appear on peer.gbci.org, at the latest November 30, 2015.* CONGRATULATIONS ON YOUR DECISION TO PURSUE PEER
GUIDE TO CERTIFICATION December 2017 *Note this document is temporary, and the content will soon appear on peer.gbci.org, at the latest November 30, 2015.* CONGRATULATIONS ON YOUR DECISION TO PURSUE PEER
eirb Electronic Institutional Review Board User Guide For more information contact: Research Administration (803)
 eirb Electronic Institutional Review Board User Guide For more information contact: Research Administration (803) 434 4899 Developed August 2008 by Research Development University of South Carolina 1 Introduction
eirb Electronic Institutional Review Board User Guide For more information contact: Research Administration (803) 434 4899 Developed August 2008 by Research Development University of South Carolina 1 Introduction
DIGITAL CLASSICS ONLINE
 DIGITAL CLASSICS ONLINE Online Submission Instructions Link: http://journals.ub.uni-heidelberg.de/index.php/dco / The individual steps for filing, correspondence if necessary, and re-submission of a revised
DIGITAL CLASSICS ONLINE Online Submission Instructions Link: http://journals.ub.uni-heidelberg.de/index.php/dco / The individual steps for filing, correspondence if necessary, and re-submission of a revised
August Objectives. Agenda. Promoting Your Professional Development with ACMPE. Describe the foundation and value of certification and fellowship
 Promoting Your Professional Development with ACMPE 2015 MGMA-Louisiana Annual Conference August 19-21, 2015 Objectives Describe the foundation and value of certification and fellowship Explain the steps
Promoting Your Professional Development with ACMPE 2015 MGMA-Louisiana Annual Conference August 19-21, 2015 Objectives Describe the foundation and value of certification and fellowship Explain the steps
IOP Conference Series: Materials Science and Engineering
 IOP Conference Series: Materials Science and Engineering The open-access proceedings service for conferences IOP Publishing, Dirac House, Temple Back, Bristol BS1 6BE, UK Tel +44 (0)117 929 7481 Fax +44
IOP Conference Series: Materials Science and Engineering The open-access proceedings service for conferences IOP Publishing, Dirac House, Temple Back, Bristol BS1 6BE, UK Tel +44 (0)117 929 7481 Fax +44
Guide to Supplemental Materials
 Guide to Supplemental Materials October 7, 2017 A submitted manuscript should stand on its own, i.e., it should make a sound presentation of the rationale, research questions, and methods and data analyses,
Guide to Supplemental Materials October 7, 2017 A submitted manuscript should stand on its own, i.e., it should make a sound presentation of the rationale, research questions, and methods and data analyses,
NEWCASTLE CLINICAL TRIALS UNIT STANDARD OPERATING PROCEDURES
 SOP details SOP title: Protocol development SOP number: TM 010 SOP category: Trial Management Version number: 03 Version date: 16 December 2016 Effective date: 16 January 2017 Revision due date: 16 January
SOP details SOP title: Protocol development SOP number: TM 010 SOP category: Trial Management Version number: 03 Version date: 16 December 2016 Effective date: 16 January 2017 Revision due date: 16 January
Redlined changes to BSCP40 for CP1494 Introduction of an objection window for Housekeeping Change Proposals
 Redlined changes to BSCP40 for CP1494 Introduction of an objection window for Housekeeping Change Proposals This CP proposes changes to BSCP40 sections 3.4 and 3.5. There are no impacts on any other sections
Redlined changes to BSCP40 for CP1494 Introduction of an objection window for Housekeeping Change Proposals This CP proposes changes to BSCP40 sections 3.4 and 3.5. There are no impacts on any other sections
Author Guidelines for Endodontic Topics
 1. Submission of Manuscripts Author Guidelines for Endodontic Topics Manuscripts should be submitted electronically via the online submission site http://mc.manuscriptcentral.com/endodontictopics. Complete
1. Submission of Manuscripts Author Guidelines for Endodontic Topics Manuscripts should be submitted electronically via the online submission site http://mc.manuscriptcentral.com/endodontictopics. Complete
Using the Pearson eportfolio System
 Using the Pearson eportfolio System Welcome to the Pearson eportfolio system. This guide provides the following instructions for candidates using the Pearson eportfolio system: Select Your Portfolio Platform
Using the Pearson eportfolio System Welcome to the Pearson eportfolio system. This guide provides the following instructions for candidates using the Pearson eportfolio system: Select Your Portfolio Platform
Submission Guidelines
 Submission Guidelines Clinical Trial Results invites the submission of phase I, II, and III clinical trials for publication in a brief print format, with full trials results online. We encourage the submission
Submission Guidelines Clinical Trial Results invites the submission of phase I, II, and III clinical trials for publication in a brief print format, with full trials results online. We encourage the submission
No more than six tables, pictures or figures can be considered for the paper version, although
 Archaeometry Thank you for your interest in Archaeometry, we look forward to receiving your paper. We are aiming for the printed edition of Archaeometry to publish papers of no more than 15 pages. We have
Archaeometry Thank you for your interest in Archaeometry, we look forward to receiving your paper. We are aiming for the printed edition of Archaeometry to publish papers of no more than 15 pages. We have
HOW ARE ELECTRONIC JOURNALS COOKED?
 HOW ARE ELECTRONIC JOURNALS COOKED? Jacek Ciesielski Publishing Director Central European Science Journals, Poland INFORUM 2005: 11th Conference on Professional Information Resources Prague, May 24-26,
HOW ARE ELECTRONIC JOURNALS COOKED? Jacek Ciesielski Publishing Director Central European Science Journals, Poland INFORUM 2005: 11th Conference on Professional Information Resources Prague, May 24-26,
Accepted manuscript deposits via Symplectic Elements
 Accepted manuscript deposits via Symplectic Elements Contents Process... 1 Accessing Symplectic... 2 Depositing an Accepted Author Manuscript or OA published article... 3 1. Let s get started... 3 2. Tell
Accepted manuscript deposits via Symplectic Elements Contents Process... 1 Accessing Symplectic... 2 Depositing an Accepted Author Manuscript or OA published article... 3 1. Let s get started... 3 2. Tell
OCLQS Portal Document Management
 OCLQS Portal Document Management Description: This Job Aid describes the process of performing various actions related to documents in the Ohio Child Licensing and Quality System (OCLQS) Portal. Table
OCLQS Portal Document Management Description: This Job Aid describes the process of performing various actions related to documents in the Ohio Child Licensing and Quality System (OCLQS) Portal. Table
Page 1/6 MEMORANDUM. DATE: 7 July Faculty & Graduate Students. John Allen, Melissa Soenke, & Jeff Greenberg
 MEMORANDUM Page 1/6 DATE: 7 July 2010 TO: FROM: RE: Faculty & Graduate Students John Allen, Melissa Soenke, & Jeff Greenberg INTRODUCTORY PSYCHOLOGY STUDENT SURVEY To all Mass Survey contributors: This
MEMORANDUM Page 1/6 DATE: 7 July 2010 TO: FROM: RE: Faculty & Graduate Students John Allen, Melissa Soenke, & Jeff Greenberg INTRODUCTORY PSYCHOLOGY STUDENT SURVEY To all Mass Survey contributors: This
Editors. Getting Started
 Editors The Editor oversees entire editorial and publishing process. The Editor, working with the Journal Manager, typically establishes the policies and procedures for the journal, which are used in setting
Editors The Editor oversees entire editorial and publishing process. The Editor, working with the Journal Manager, typically establishes the policies and procedures for the journal, which are used in setting
Brill s Editorial Manager (EM) Manual for Authors Contents
 Brill s Editorial Manager (EM) Manual for Authors Contents 1. Introduction... 2 2. Getting Started: Creating an Account... 2 2.1 Creating an Account Using Your ORCID Record... 3 3. Logging into EM... 4
Brill s Editorial Manager (EM) Manual for Authors Contents 1. Introduction... 2 2. Getting Started: Creating an Account... 2 2.1 Creating an Account Using Your ORCID Record... 3 3. Logging into EM... 4
Instruction for Web Submission
 e-jssnt Instruction for Web Submission using J-STAGE3 / ScholarOne Manuscript system Ver. 2.0 Submit a New Manuscript (1) Preparation of manuscript files Please refer to the Instruction for manuscript
e-jssnt Instruction for Web Submission using J-STAGE3 / ScholarOne Manuscript system Ver. 2.0 Submit a New Manuscript (1) Preparation of manuscript files Please refer to the Instruction for manuscript
Guidelines to Format and Upload Final Version of Accepted Paper Land and Poverty Conference 2018
 Guidelines to Format and Upload Final Version of Accepted Paper Land and Poverty Conference 2018 1. To upload the final version of your accepted paper for the 2018 Land and Poverty Conference, please log
Guidelines to Format and Upload Final Version of Accepted Paper Land and Poverty Conference 2018 1. To upload the final version of your accepted paper for the 2018 Land and Poverty Conference, please log
Instructions/template for preparing your NOLTA manuscript (Ver. 3.02, Aug. 28, 2015)
 NOLTA, IEICE Paper Instructions/template for preparing your NOLTA manuscript (Ver. 3.02, Aug. 28, 2015) Name Author1 1a), Name Author2 2, and Name Author3 1 1 Institute of Industrial Science, University
NOLTA, IEICE Paper Instructions/template for preparing your NOLTA manuscript (Ver. 3.02, Aug. 28, 2015) Name Author1 1a), Name Author2 2, and Name Author3 1 1 Institute of Industrial Science, University
Institute of Economic Sciences, Belgrade, Serbia Manual
 Institute of Economic Sciences, Belgrade, Serbia Manual User Registration and Submission of Articles to the Journal of Women's Entrepreneurship and Education Editorial Board of the Journal of Women s Entrepreneurship
Institute of Economic Sciences, Belgrade, Serbia Manual User Registration and Submission of Articles to the Journal of Women's Entrepreneurship and Education Editorial Board of the Journal of Women s Entrepreneurship
BSCP128 Production, Submission, Audit and Approval of Line Loss Factors Version 3.0. Balancing and Settlement Code. BSC Procedure DRAFT
 Balancing and Settlement Code BSC Procedure Production, Submission, Audit and Approval of Line Loss Factors BSCP128 Version 3.0- Effective Date: 30 June 2011 Balancing and Settlement Code Page 1 of 32
Balancing and Settlement Code BSC Procedure Production, Submission, Audit and Approval of Line Loss Factors BSCP128 Version 3.0- Effective Date: 30 June 2011 Balancing and Settlement Code Page 1 of 32
BSCP128 Production, Submission, Audit and Approval of Line Loss Factors Version 3.0. Balancing and Settlement Code. BSC Procedure DRAFT
 Balancing and Settlement Code BSC Procedure Production, Submission, Audit and Approval of Line Loss Factors BSCP128 Version 3.0 Effective Date: 30 June 2011 Balancing and Settlement Code Page 1 of 33 30
Balancing and Settlement Code BSC Procedure Production, Submission, Audit and Approval of Line Loss Factors BSCP128 Version 3.0 Effective Date: 30 June 2011 Balancing and Settlement Code Page 1 of 33 30
The Swirl logo is a trade mark of the Cabinet Office ITIL is a registered trade mark of the Cabinet Office
 PROFESSIONAL QUALIFICATION SCHEME ITIL PRACTICES FOR SERVICE MANAGEMENT THE ITIL MASTER QUALIFICATION ONLINE SYSTEM USER GUIDE FOR CANDIDATES The Swirl logo is a trade mark of the Cabinet Office ITIL is
PROFESSIONAL QUALIFICATION SCHEME ITIL PRACTICES FOR SERVICE MANAGEMENT THE ITIL MASTER QUALIFICATION ONLINE SYSTEM USER GUIDE FOR CANDIDATES The Swirl logo is a trade mark of the Cabinet Office ITIL is
OJS Quick Guide for ASSOciAte editors 1
 OJS Quick Guide for Associate Editors 1 Content Quick Guide for Asociate EditorS SECTION 1 Login 1 page... 3 SECTION 2 Review 2... 5 1SECTION 1 Login Go to website Login with your username and password
OJS Quick Guide for Associate Editors 1 Content Quick Guide for Asociate EditorS SECTION 1 Login 1 page... 3 SECTION 2 Review 2... 5 1SECTION 1 Login Go to website Login with your username and password
GUIDE FOR AUTHORS PREPARING A MANUSCRIPT: INSTRUCTIONS FOR
 GUIDE FOR AUTHORS PREPARING A MANUSCRIPT: INSTRUCTIONS FOR AUTHORS Below you will find a fairly detailed description of how a paper to be submitted to Health Professions Education should look like. Attached
GUIDE FOR AUTHORS PREPARING A MANUSCRIPT: INSTRUCTIONS FOR AUTHORS Below you will find a fairly detailed description of how a paper to be submitted to Health Professions Education should look like. Attached
16th ISQOLS Annual Conference: Promotion of Quality of Life in the Changing World June 14 16, 2018 in Hong Kong. EasyChair Instruction for Authors
 16th ISQOLS Annual Conference: Promotion of Quality of Life in the Changing World June 14 16, 2018 in Hong Kong EasyChair Instruction for Authors Contents 1 Log in or create an account as an Author...
16th ISQOLS Annual Conference: Promotion of Quality of Life in the Changing World June 14 16, 2018 in Hong Kong EasyChair Instruction for Authors Contents 1 Log in or create an account as an Author...
Author Guidelines for Online Submission
 CSIR-NATIONAL INSTITUTE OF SCIENCE COMMUNICATION AND INFORMATION RESOURCES (CSIR-NISCAIR) Dr K S Krishnan Marg, New delhi-110012 14, Satsang Vihar Marg, New Delhi - 110067 Author Guidelines for Online
CSIR-NATIONAL INSTITUTE OF SCIENCE COMMUNICATION AND INFORMATION RESOURCES (CSIR-NISCAIR) Dr K S Krishnan Marg, New delhi-110012 14, Satsang Vihar Marg, New Delhi - 110067 Author Guidelines for Online
The Journal of The Textile Institute
 The Journal of The Textile Institute And Tutorial for Authors Table of Contents Registering 3 Logging In 4 Changing your password 5 Submitting a paper 6-9 Reviewing & approving your paper 10 Tracking the
The Journal of The Textile Institute And Tutorial for Authors Table of Contents Registering 3 Logging In 4 Changing your password 5 Submitting a paper 6-9 Reviewing & approving your paper 10 Tracking the
Hispania A journal devoted to the teaching of Spanish and Portuguese published by the American Association of Teachers of Spanish and Portuguese
 Frequently Asked Questions about Submitting to and Reviewing for Hispania, page 1 Frequently Asked Questions about Submitting to and Reviewing for Hispania Hispania A journal devoted to the teaching of
Frequently Asked Questions about Submitting to and Reviewing for Hispania, page 1 Frequently Asked Questions about Submitting to and Reviewing for Hispania Hispania A journal devoted to the teaching of
CTO Provincial Study Closure Form
 CTO Orange text indicates an upload or action feature Red/italics/bold indicates question/feature dependencies Questions with an asterisk (*) are mandatory and must be completed prior to signatures/submission
CTO Orange text indicates an upload or action feature Red/italics/bold indicates question/feature dependencies Questions with an asterisk (*) are mandatory and must be completed prior to signatures/submission
Questions and Answers. Converting Existing Protocols into CHeRP IRB
 Questions and Answers Converting Existing Protocols into CHeRP IRB Questions: (hold ctrl and click on the question to jump directly to that answer) 1. Do I need to convert my existing protocol applications
Questions and Answers Converting Existing Protocols into CHeRP IRB Questions: (hold ctrl and click on the question to jump directly to that answer) 1. Do I need to convert my existing protocol applications
Academic Editor Tutorial
 Academic Editor Tutorial Contents I. Assignments a. Receiving an invitation b. Responding to an invitation c. Primary review i. Cascading peer review II. Inviting additional reviewers a. Reviewer selection
Academic Editor Tutorial Contents I. Assignments a. Receiving an invitation b. Responding to an invitation c. Primary review i. Cascading peer review II. Inviting additional reviewers a. Reviewer selection
Plagiarism Page 1/13
 Plagiarism... 2 Page 1/13 Plagiarism 1. Policy statement 2. Special circumstances for Cochrane Systematic Reviews 3. Avoiding plagiarism 3.1. Use of text templates 3.2. Cochrane Overviews of reviews (Cochrane
Plagiarism... 2 Page 1/13 Plagiarism 1. Policy statement 2. Special circumstances for Cochrane Systematic Reviews 3. Avoiding plagiarism 3.1. Use of text templates 3.2. Cochrane Overviews of reviews (Cochrane
o Changes that require author intervention, e.g., address/ requirements and/or missing sections, should be conveyed through remarks.
 This document covers all Formatting-related points to be adhered to by all Enago Professional Editors when an author opts for Formatting. This is conveyed through the assignment email as Formatting: Yes,
This document covers all Formatting-related points to be adhered to by all Enago Professional Editors when an author opts for Formatting. This is conveyed through the assignment email as Formatting: Yes,
How to complete and submit the beneficiary report
 How to complete and submit the beneficiary report This page describes the steps to be taken to complete and submit the beneficiary report. The steps described follow the example of using a project for
How to complete and submit the beneficiary report This page describes the steps to be taken to complete and submit the beneficiary report. The steps described follow the example of using a project for
USI Electronic Medical Records Athletic Trainer System. New Student-Athletes
 USI Electronic Medical Records Athletic Trainer System New Student-Athletes Please view the website https://www.atsusers.com and complete the following items. Preferred deadline is July 17 th, 2017. This
USI Electronic Medical Records Athletic Trainer System New Student-Athletes Please view the website https://www.atsusers.com and complete the following items. Preferred deadline is July 17 th, 2017. This
Electronic Meeting Registration System. Step-by-Step User s Guide
 Electronic Meeting Registration System Step-by-Step User s Guide May 2014 1 Table of Contents Introduction... 3 STEP 1: TO CREATE AN ACCOUNT FOR THE REGISTRATION FOCAL POINT... 4 STEP 2: TO REGISTER DELEGATES...
Electronic Meeting Registration System Step-by-Step User s Guide May 2014 1 Table of Contents Introduction... 3 STEP 1: TO CREATE AN ACCOUNT FOR THE REGISTRATION FOCAL POINT... 4 STEP 2: TO REGISTER DELEGATES...
STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018. I. Protocol Application & IRB Certification Tutorial...2
 STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018 I. Protocol Application & IRB Certification Tutorial...2 II. Logging In (Password)... 2 III. Creating a New Protocol Record / Menu Bar...
STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR Rev. 10/2018 I. Protocol Application & IRB Certification Tutorial...2 II. Logging In (Password)... 2 III. Creating a New Protocol Record / Menu Bar...
IRBManager Quick Start Guide AMENDMENT SUBMISSION - CHANGE IN PERSONNEL
 Page 1 of 12 IRBManager Quick Start Guide AMENDMENT SUBMISSION - CHANGE IN PERSONNEL NOTE: This Quick Start Guide provides instructions for removing and/or adding personnel, other than the Principal Investigator,
Page 1 of 12 IRBManager Quick Start Guide AMENDMENT SUBMISSION - CHANGE IN PERSONNEL NOTE: This Quick Start Guide provides instructions for removing and/or adding personnel, other than the Principal Investigator,
IRMA Human Ethics Researcher User Guide
 IRMA Human Ethics Researcher User Guide IRMA Researcher User Guide 1. Overview 1.01 What is IRMA? 1.02 What are the Benefits? 1.03 ISLHD Research and IRMA 2. Key Terms in IRMA 2.01 Coversheets 2.02 Templates
IRMA Human Ethics Researcher User Guide IRMA Researcher User Guide 1. Overview 1.01 What is IRMA? 1.02 What are the Benefits? 1.03 ISLHD Research and IRMA 2. Key Terms in IRMA 2.01 Coversheets 2.02 Templates
IRPS 2018 Manuscript & Platform Presentation Submission Process
 IRPS 2018 Manuscript & Platform Presentation Submission Process Susumu Shuto 1 Jason Ryan 2 1 IRPS 2018 Publications Chair, Toshiba 2 IRPS 2018 Audio Visual / Presentations Chair, NIST Submission Web-Sites
IRPS 2018 Manuscript & Platform Presentation Submission Process Susumu Shuto 1 Jason Ryan 2 1 IRPS 2018 Publications Chair, Toshiba 2 IRPS 2018 Audio Visual / Presentations Chair, NIST Submission Web-Sites
P a g e 1. Dear AJCH Authors,
 P a g e 1 Stephen R. Lankton, LCSW, DAHB P.O. Box 34177 Phoenix, AZ 85067 Phone: 602-532-0800 Fax: 602-532-0801 E-mail: steve@lankton.com Dear AJCH Authors, I would like to formally introduce and invite
P a g e 1 Stephen R. Lankton, LCSW, DAHB P.O. Box 34177 Phoenix, AZ 85067 Phone: 602-532-0800 Fax: 602-532-0801 E-mail: steve@lankton.com Dear AJCH Authors, I would like to formally introduce and invite
STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR / STUDENT
 Rev: 06/2017 STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR / STUDENT In This Document Protocol Application (Exempt and Expedited/Full Board Review)...2 NIH Certificate (Required)... 2 Logging In...
Rev: 06/2017 STREAMLYNE INITIAL GUIDE FOR PRINCIPAL INVESTIGATOR / STUDENT In This Document Protocol Application (Exempt and Expedited/Full Board Review)...2 NIH Certificate (Required)... 2 Logging In...
PLAViMoP Database User Manual. Version
 PLAViMoP Database User Manual Version 1 2018 CONTENTS 1 Navigation... 3 1.1 Header navigation... 3 1.2 Footer navigation... 3 1.3 User menu... 3 2 Access roles... 4 3 PLAViMoP Database... 5 3.1 Motion
PLAViMoP Database User Manual Version 1 2018 CONTENTS 1 Navigation... 3 1.1 Header navigation... 3 1.2 Footer navigation... 3 1.3 User menu... 3 2 Access roles... 4 3 PLAViMoP Database... 5 3.1 Motion
CANDIDATE BRIEF. Web Content Editor, Marketing. Salary: Grade 6 ( 26,495 31,604 p.a.) Reference: CSMAR1014
 CANDIDATE BRIEF Web Content Editor, Marketing Salary: Grade 6 ( 26,495 31,604 p.a.) Reference: CSMAR1014 Fixed-term for 12 Months, available from 01 September 2017 to 30 September 2018 (To provide project
CANDIDATE BRIEF Web Content Editor, Marketing Salary: Grade 6 ( 26,495 31,604 p.a.) Reference: CSMAR1014 Fixed-term for 12 Months, available from 01 September 2017 to 30 September 2018 (To provide project
ScholarOne Manuscripts Author Guide
 ScholarOne Manuscripts Author Guide Getting Started Logging In If you have not accessed the website before you will need to create your own account. Please click on the Create Account link found on the
ScholarOne Manuscripts Author Guide Getting Started Logging In If you have not accessed the website before you will need to create your own account. Please click on the Create Account link found on the
PSD2 Assessment of Operational and Security Risks 1 ONR User Guidelines Document
 PSD2 Assessment of Operational and Security Risks 1 ONR User Guidelines Document Published Date September 2018 Version V 1.0 1 Reporting of operational and security risks relating to payment services in
PSD2 Assessment of Operational and Security Risks 1 ONR User Guidelines Document Published Date September 2018 Version V 1.0 1 Reporting of operational and security risks relating to payment services in
pg. 1 Author Tutorial
 pg. 1 Author Tutorial Contents 1. Registration... 4 2. Signing In... 5 3. Search Bar... 6 4. Notifications... 6 5. Message Box... 6 6. Chat Box... 7 7. Logout... 7 8. The User Dashboard... 8 9. New Manuscript
pg. 1 Author Tutorial Contents 1. Registration... 4 2. Signing In... 5 3. Search Bar... 6 4. Notifications... 6 5. Message Box... 6 6. Chat Box... 7 7. Logout... 7 8. The User Dashboard... 8 9. New Manuscript
HRA Open User Guide for Awardees
 HRA Open User Guide for Awardees HRA Open is an web-based service sponsored by the Health Research Alliance (HRA) that promotes open access by linking your award information with PubMed Central so you
HRA Open User Guide for Awardees HRA Open is an web-based service sponsored by the Health Research Alliance (HRA) that promotes open access by linking your award information with PubMed Central so you
Researcher User Manual
 Researcher User Manual Post-Review Application Management Cloning Applications, Creating and Managing Events Audience: Principal Investigators & Project Team Members Updated: December 1, 2016 Checkpoint
Researcher User Manual Post-Review Application Management Cloning Applications, Creating and Managing Events Audience: Principal Investigators & Project Team Members Updated: December 1, 2016 Checkpoint
CCGT Grant Application System User Guide for Applicants
 CCGT Grant Application System User Guide for Applicants About this document This document describes how to register as a user of the CCGT Grant Application System, and how to use the system to apply for
CCGT Grant Application System User Guide for Applicants About this document This document describes how to register as a user of the CCGT Grant Application System, and how to use the system to apply for
IRMA Researcher User Guide v2 DRAFT. IRMA Researcher User Guide
 IRMA Researcher User Guide v2 IRMA Researcher User Guide IRMA Researcher User Guide 1. Overview 1.01 What is IRMA? 1.02 What are the Benefits? 1.03 ISLHD Research and IRMA 2. Key Terms in IRMA 2.01 Coversheets
IRMA Researcher User Guide v2 IRMA Researcher User Guide IRMA Researcher User Guide 1. Overview 1.01 What is IRMA? 1.02 What are the Benefits? 1.03 ISLHD Research and IRMA 2. Key Terms in IRMA 2.01 Coversheets
Ethical Review Manager. Applicant User Guide
 Ethical Review Manager Applicant User Guide Last Updated: June 2017 Introduction The Ethical Review Manager (ERM) System has been designed to enable applications for ethical approval to conduct research
Ethical Review Manager Applicant User Guide Last Updated: June 2017 Introduction The Ethical Review Manager (ERM) System has been designed to enable applications for ethical approval to conduct research
ClinicalTrials.gov PRS How to Register and Maintain a Record
 ClinicalTrials.gov PRS How to Register and Maintain a Record IRB Compliance Program PRS Administrator, Brian Brotzman Human Subjects Office/Institutional Review Board Overview Purpose Rules and Regulations
ClinicalTrials.gov PRS How to Register and Maintain a Record IRB Compliance Program PRS Administrator, Brian Brotzman Human Subjects Office/Institutional Review Board Overview Purpose Rules and Regulations
Guide to Allen verifig
 Guide to Allen verifig Version 03.05.12 How To Upload Graphic Files to VeriFig To upload graphic files for preflight in VeriFig, go to http://verifig.allenpress.com. Allen verifig is a web-based application
Guide to Allen verifig Version 03.05.12 How To Upload Graphic Files to VeriFig To upload graphic files for preflight in VeriFig, go to http://verifig.allenpress.com. Allen verifig is a web-based application
NHS Health Check Internal/External Quality control Crib Sheet. Table of Contents. Alere Afinion HbA1c Control (Internal)
 NHS Health Check Internal/External Quality control Crib Sheet Table of Contents Alere Afinion HbA1c Control (Internal) Page 2 WEQAS HbA1c Quality Control Assessment (External) Page 3 WEQAS Afinion Lipid
NHS Health Check Internal/External Quality control Crib Sheet Table of Contents Alere Afinion HbA1c Control (Internal) Page 2 WEQAS HbA1c Quality Control Assessment (External) Page 3 WEQAS Afinion Lipid
This tutorial will guide a curator/user to create the files to upload phenotype experiment annotation and data onto T3.
 This tutorial will guide a curator/user to create the files to upload phenotype experiment annotation and data onto T3. Updated December 2012. Any feedback is welcome! Sections 1. Download the Template
This tutorial will guide a curator/user to create the files to upload phenotype experiment annotation and data onto T3. Updated December 2012. Any feedback is welcome! Sections 1. Download the Template
Last revised: September 30, e-protocol User Guide 1
 e-protocol User Guide Last revised: September 30, 2015 e-protocol User Guide 1 e-protocol is an electronic system for submitting and monitoring the status of Institutional Review Board (IRB) submissions.
e-protocol User Guide Last revised: September 30, 2015 e-protocol User Guide 1 e-protocol is an electronic system for submitting and monitoring the status of Institutional Review Board (IRB) submissions.
ecampus Submission Process
 ecampus Submission Process Progress Report Submission, and Installment Submission & Feedback 1 All Progress Reports and Installment Submissions are found on the Assignments Page. 2 Individual assignments
ecampus Submission Process Progress Report Submission, and Installment Submission & Feedback 1 All Progress Reports and Installment Submissions are found on the Assignments Page. 2 Individual assignments
