LSM 5 MP, LSM 510 and LSM 510 META Laser Scanning Microscopes
|
|
|
- Alexia Kennedy
- 6 years ago
- Views:
Transcription
1 LSM 5 MP, LSM 510 and LSM 510 META Laser Scanning Microscopes Brief Operating Manual Release 4.2 January 2007
2 Contents Page Starting the System...3 Setting the microscope...6 Configuring the beam path and lasers...8 Scanning an image...11 Storing an image...15 Switching off the system...15 Introduction For your safety! Observe the following instructions: The LSM 510 and LSM 510 META laser scanning microscope, including its original accessories and compatible accessories from other manufacturers, may only be used for the purposes and microscopy techniques described in this manual (intended use). In the Operating Manual, read the chapter Safety Instructions carefully before starting operation. Follow the safety instructions described in the operating manual of the microscope and HBO 100 mercury lamp. 2 01/07
3 Starting the System Switching on the LSM system When set to ON the REMOTE CONTROL switch labeled System/PC provides power to the microscope and the computer. This allows to use the microscope and the computer without running the LSM Software (Fig. 1). To switch on the system completely put the Components switch also to ON. Now the complete system is ready to be initialized with the LSM Software. Switching on the HBO 100 mercury lamp Switch on the HBO 100 mercury lamp via the switch of the power supply, see operating manual of the mercury lamp or microscope. Fig. 1 REMOTE CONTROL switch Switching on the Enterprise UV-Ar Laser If the UV laser is required, switch it on via the toggle switch (Fig. 2/1) of the power supply. It will be ready for operation after a few seconds. Fig. 2 Power supply of UV-Ar laser 01/07 3
4 Starting the LSM 5 software program LSM 510 Double click the LSM 510 icon on the desktop of WINDOWS to start the LSM 5 software program. The LSM 510 Switchboard window appears on the screen (Fig. 3). Fig. 3 LSM 510 Switchboard menu Click on the Scan New Images button in the LSM 510 Switchboard window. Clicking on this button activates the complete LSM hardware (on-line mode). Click on the Start Expert Mode button in the LSM 510 Switchboard window. The LSM Expert Mode Main menu appears on the screen. Use of this mode requires to be thoroughly familiar with the exact microscope procedures and interrelations. Main menu (pull-down) Main menu toolbar Subordinate toolbar Fig. 4 Main menu for Expert Mode 4 01/07
5 Creating a database for acquired images Click on the File button in the Main menu toolbar. The File subordinate toolbar appears in the Main menu. Fig. 5 Main menu with File subordinate toolbar Click on the New button in the File subordinate toolbar. The Create New Database window appears. Select drive C: or D: from pull down menu. Create a new directory if needed. Fig. 6 Create New Database window Fig. 7 Create New Database window Turning on the lasers Click on the Acquire button in the Main menu to open the Acquire subordinate toolbar. Fig. 8 Acquire subordinate toolbar 01/07 5
6 Click on the Laser button to open the Laser Control window. Select the appropriate Laser Unit by clicking on the name of it. Click the Standby button to switch required laser(s) to Standby. When status is Ready click the On button. Set Output [%] so that the Tube Current is about 4 A (50% of the output power). Fig. 9 Laser Control window Setting the microscope Changing between direct observation or laser scanning The VIS, TV and LSM buttons switch the beam path and indicate which beam path has been set in the binocular tube of the microscope: Click on the VIS button to set the microscope for direct observation via the eyepieces of the binocular tube, lasers are off. Click on the TV button to set the microscope camera observation (if connected) via camera adapter of the binocular tube. Click on the LSM button to set the microscope screen observation via laser excitation using the LSM 510 and software evaluation. Setting the microscope and storing the settings Click on the VIS button for direct observation. Click on the Micro button in the Acquire subordinate toolbar to open the Microscope Control window of the used microscope. The Microscope Control window appears (Fig. 10). 6 01/07
7 Selecting an objective Open the graphical pop-up menu by clicking on the Objective button (Fig. 10). Click on the objective you want to select. The selected objective will automatically move into the beam path. Focussing the microscope for transmitted light Open the graphical pop-up menu by clicking on the Transmitted Light button (Fig. 11). Click on the On button. Set the intensity of the Halogen illuminator using the slider. Click on Close to close the pop-up menu. Place specimen on microscope stage. The cover slip must be facing the objective. Use the focusing drive of the microscope to focus the required object plane. Select specimen detail by moving the stage in X and Y using the XY stage fine motion control. Setting the microscope for reflected light Click on the Reflected Light button to open the shutter of the HBO 100 mercury lamp. Click on the Reflector button and select the desired filter set by clicking on it. Fig. 10 Microscope Control window, e.g.: Axio Observer Storing the microscope settings Microscope settings can be stored and up to 8 buttons assigned for fast retrieval and adjustment using the Microscope Settings panel. The Store button permits existing microscope configurations to be stored under any name. The Apply button permits existing stored microscope configurations to be loaded. The Delete button permits existing microscope configurations to be deleted. The Assign button permits the assignment of a microscope configuration to a button. Note: Depending on the microscope configuration, settings must be done manually if necessary. Fig. 11 Microscope Control window with Transmitted Light pop-up menu 01/07 7
8 Configuring the beam path and lasers Click on the LSM button in the Acquire subordinate toolbar for laser scanning. Choosing the configuration Single Track Use for single, double and triple labeling; simultaneous scanning only Advantage: faster image acquisition Disadvantage: potential cross talk between channels Multi Track Use for double and triple labeling; sequential scanning, line by line or frame by frame Advantage: when one track is active, only one detector and one laser is switched on. This reduces cross talk. Disadvantage: slower image acquisition Fig. 12 Configuration Control window for Single Track Click on the Config button in the Acquire subordinate toolbar to open the Configuration Control window (Fig. 12). Setting for single track configuration in Channel Mode Select Channel Mode (Fig. 12). Click on the Single Track button in the Configuration Control window (Fig. 12). Click on the Descanned button if available. The Beam Path and Channel Assignment panel of the Configuration Control window displays the selected track configuration which is used for the scan procedure. You can change the settings of this panel using the following function elements: Activation / deactivation of the excitation wavelengths (check box) and setting of excitation intensities (slider). Open the Laser Control window via the Laser button. Selection of the main dichroic beam splitter (HFT) or secondary dichroic beam splitter (NFT) position through selection from the relevant list box. Selection of an emission filter through selection from the relevant list box. Activation / deactivation (via check box) of the selected channel (Ch 1-4, monitor diode ChM, META detectors ChS1-8, transmission ChD) for the scanning procedure by assigning an existing color icon or defining a new one. 8 01/07
9 Select the appropriate filters and activate the channels. Click the Excitation button to select the laser lines and set the attenuation values (transmission in %) in the displayed window. For the configuration of the beam path, please refer to the application-specific configurations depending on the used dyes and markers and the existing instrument configuration. Clicking on the Spectr button opens the Detection Spectra & Laser Lines window (Fig. 13) to display the activated laser lines for excitation (colored vertical lines) and channels (colored horizontal bars). Fig. 13 Detection Spectra & Laser Lines window Clicking on the Config button opens the Track Configurations window (Fig. 14) to load, store or delete track configurations. For storing a new track configuration enter a desired name in the first line of the Configurations list box and click an Store. For loading an existing configuration select it in the list box and click on Apply. For deleting an existing configuration select it in the list box and click on Delete. Fig. 14 Track Configurations window Setting for multi track configuration in Channel Mode The Multi Track function permits several tracks to be defined as one configuration for the scan procedure, to be stored under any name, reloaded or deleted. The maximum of four tracks with up to 8 channels can be defined simultaneously and then scanned one after the other. Each track is a separate unit and can be configured independently of the other tracks with regard to detection channels, laser power, emission filters and dichroic beam splitters. Select Channel Mode (Fig. 15). Fig. 15 Configuration Control window for Multi Track 01/07 9
10 Click on the Multi Track button in the Configuration Control window (Fig. 15). Click on the Descanned button if available. The following functions are available in the List of Tracks panel (Fig. 15). Modes Switch tracks after each Line button Tracks are switched during scanning line by line. The following settings can be changed between tracks: Laser line and intensity and channels incl. settings for gain and offset. Switch tracks after each Frame button Frame Fast button Tracks are switched during scanning frame by frame. The following settings can be changed between tracks: Laser line and intensity, all filters and beam splitters, the channels incl. settings for gain and offset and the pinhole position and diameter. The scanning procedure can be made faster. Only the laser line and intensity and the Amplifier Offset are switched, but no other hardware components. The tracks are all matched to the first track with regard to emission filters, dichroic beam splitters, setting of Detector Gain, pinhole position and diameter. When Line button is selected, the same rules apply as for Frame Fast. Settings Add Track button Remove button Store/Apply Single Track button An additional track is added to the configuration list. A maximum of four tracks can be used. One track with a default configuration is added, i.e.: Ch 1 channel is activated, all laser lines are switched off, emission filters and dichroic beam splitters are set in accordance with the configuration last used. The single track previously marked in the List of Tracks panel in the Name column is deleted. Opens the Track Configurations window. A selected track defined in a Channel Mode Configuration can also be stored as a single track for single tracking applications. Also, it's possible to load a single track into a multi tracking configuration. A click on this arrow button will move the selected track (highlighted in blue) one position upwards in the list box. A click on this arrow button will move the selected track (highlighted in blue) one position downwards in the list box. Configure each desired track for Multi Track function as described for Single Track. For storing/applying or deleting a Channel Mode Configuration use the Config button /07
11 Scanning an image Click on the Scan button in the Acquire subordinate toolbar to open the Scan Control window (Fig. 16). Setting the parameters for scanning Select Mode in the Scan Control window. Select the Frame Size as predefined number of pixels or enter your own values (e.g. 300 x 600) in the Objective Lens, Image Size & Line Step Factor panel. Click on the Optimal button for calculation of appropriate number of pixels depending on N.A. and λ. The number of pixels influences the image resolution! Note: When using an Axioskop 2 FS MOT, indicate the objective that is in use in the Scan Control window. This ensures correct calculation of pinhole size, frame size, speed, and Z stack parameters. Fig. 16 Scan Control window, Mode settings Adjusting the scan speed Use the slider in the Speed panel (Fig. 16) to adjust the scan speed. A higher speed with averaging results in the best signal to noise ratio. Scan speed 8 usually produces good results. Us speed 6 or 7 for superior images. Choosing the dynamic range Select the dynamic range 8 or 12 Bit (per pixel) in the Pixel Depth, Scan Direction & Scan Average panel (Fig. 16). 8 Bit corresponds to 256 gray levels; 12 Bit to 4096 levels. Publication quality images should be acquired using 12 Bit data depth. 12 Bit is also recommended when doing quantitative measurements or when imaging low fluorescence intensities. Setting the scan averaging Averaging improves the image by increasing the signal : noise ratio. It can be achieved line by line or frame by frame. Frame averaging helps reduce photobleaching, but should not be applied for moving samples. Select the Line or Frame mode for averaging. Select the desired scan average method Mean or Sum in the Method selection box. If you are using the Mean method, the image information is generated by adding up all scans pixel by pixel and then calculating the mean value. 01/07 11
12 In the Sum method, the pixel values of all scans are only added up, without a mean value being calculated. Select the Number for averaging. Continuous averaging is possible in the Frame mode. In this case a Finish button for ending continuous averaging is displayed instead of the Cont. button. Adjusting the pinhole Select Channels in the Scan Control window. Set the Pinhole size to 1 (Airy unit) for best compromise between depth discrimination and efficiency. Pinhole adjustment changes the Optical Slice. When collecting multi channel images, adjust the pinholes such that each channel has the same Optical Size. This is important for colocalization studies. Image acquisition Fig. 17 Scan Control window, Channel settings Once you have set up your parameter as defined in the above section, you can acquire a frame image of your specimen. Use one of the Find, Fast XY, Single or Cont. buttons to start the scanning procedure and acquire an image. Scanned images are shown in separate windows. Click on the Stop button to stop the current scan procedure if wanted. Select Find to automatically pre-adjust of detector sensitivity. Select Fast for continuous and fast scanning useful to find and change the focus. Select Single to record a single image. Fig. 18 Image window Select Cont. to continuously scan with the preset scan speed. Select Stop to stop the current scan procedure /07
13 Image optimization Choosing a lookup table Select Palette in the Image window of the scanned image (Fig. 19). The Color Palette window appears. In the Color Palette List panel, click on the Range Indicator item (Fig. 20). The scanned image appears in a false-color presentation (Fig. 19). If the image is too bright, it appears red on the screen. Red = saturation (maximum grey value). If the image is not bright enough, it appears blue on the screen. Blue = zero (minimum grey value). Adjusting the laser intensity Set the Pinhole to 1 Airy Unit (Fig. 21). Set the Detector Gain high. When the image is saturated, decrease laser power in the Excitation panel (Fig. 21) using the slider. Fig. 19 Image window Adjusting gain and offset Increase the Amplifier Offset until all blue pixels disappear, and then make it slightly positive (Fig. 21). Reduce the Detector Gain until the red pixels only just disappear. Fig. 20 Color Palette window Fig. 21 Scan Control window, Channel settings 01/07 13
14 Fig. 22 Scan Control window, Z Stack settings Scanning a Z stack Select Z Stack in the Scan Control window. Select Frame Select Mark First/Last on the Z Settings panel. Click on the XY cont button. A continuous XY-scan of the set focus position will be performed. Use the focusing drive of the microscope to focus on the upper position of the specimen area where the Z Stack is to start. Click on the Mark First button to set the upper position of the Z Stack. Then focus on the lower specimen area where the recording of the Z Stack is to end. Click on the Mark Last button to set this lower position. Click on X:Y:Z=1:1:1 button to set the Z- interval in such a way that the voxel has identical dimensions in the X-, Y- and Z- directions (cube). Click on the Start button to start the recording of the Z Stack /07
15 Storing an image Click on the Save or Save As button in the Image window or in the File subordinate toolbar of the Main menu. The Save Image and Parameter As window appears. Fig. 23 Save Image and Parameter As window Enter file name, description and notes in the appropriate text boxes. Click on the OK button. Switching off the system Click on the File button in the Main menu and then click on the Exit button to leave LSM 5 software program (Fig. 5). If any lasers are still running you should shut them off now in the pop up window indicating the lasers still in use. Shut down the computer. Wait until the fan of the Argon laser has switched off. On the REMOTE CONTROL switch turn off the Components switch and the System/PC switch (Fig. 1). Switch off the HBO 100 mercury lamp. Switch the UV-Ar laser of via the toggle switch of the power supply (Fig. 2). 01/07 15
LSM510 Confocal Microscope Standard Operation Protocol Basic Operation
 LSM510 Confocal Microscope Standard Operation Protocol Basic Operation Please make sure that the COMPRESSED AIR has been TURNED ON prior to the use of the equipment. Kindly inform the administrator if
LSM510 Confocal Microscope Standard Operation Protocol Basic Operation Please make sure that the COMPRESSED AIR has been TURNED ON prior to the use of the equipment. Kindly inform the administrator if
Basic Users Guide for the LSM 510 Confocal Microscope
 Basic Users Guide for the LSM 510 Confocal Microscope Ian Jones and Adam Westmacott Department of Biology & Biochemistry, University of Bath, Bath, UK Updated 01 October 2003 This guide describes the basic
Basic Users Guide for the LSM 510 Confocal Microscope Ian Jones and Adam Westmacott Department of Biology & Biochemistry, University of Bath, Bath, UK Updated 01 October 2003 This guide describes the basic
ConfoCor 2 Fluorescence Correlation Microscope
 ConfoCor 2 Fluorescence Correlation Microscope Operating Manual Release 3.2 INTRODUCTION Carl Zeiss ConfoCor 2 Knowledge of this manual is required for the operation of the instrument. Would you therefore
ConfoCor 2 Fluorescence Correlation Microscope Operating Manual Release 3.2 INTRODUCTION Carl Zeiss ConfoCor 2 Knowledge of this manual is required for the operation of the instrument. Would you therefore
Samples Carolina sample slides (pollen, algae, ). Clean off oil with lens paper then OpticPad around lens (metal not glass) when done.
 Bi/BE 227 Winter 2018 Assignment #1 Widefield and confocal laser scanning microscopy Schedule: Jan 3: Lecture Jan 3-12: Students get trained on how to use scopes, start on assignment Jan 3-17: Carrying
Bi/BE 227 Winter 2018 Assignment #1 Widefield and confocal laser scanning microscopy Schedule: Jan 3: Lecture Jan 3-12: Students get trained on how to use scopes, start on assignment Jan 3-17: Carrying
1. Getting Started: Brief Step-By-Step Guide (PDF File)
 Page 1 of 5 1. Getting Started: Brief Step-By-Step Guide (PDF File) Leica SP2 confocal microscope is controlled via software LCS (Leica Confocal Software). The latest version is V2.5. Bellowing is a brief
Page 1 of 5 1. Getting Started: Brief Step-By-Step Guide (PDF File) Leica SP2 confocal microscope is controlled via software LCS (Leica Confocal Software). The latest version is V2.5. Bellowing is a brief
Zeiss LSM 510 Meta inverted confocal manual
 Power Up Zeiss LSM 510 Meta inverted confocal manual *adapted from the Duke University Light Microscopy Core Facility manual 1. Turn on the mercury lamp (if required) and sign in the log book, recording
Power Up Zeiss LSM 510 Meta inverted confocal manual *adapted from the Duke University Light Microscopy Core Facility manual 1. Turn on the mercury lamp (if required) and sign in the log book, recording
DSU Start-Up instructions
 DSU Start-Up instructions Always: - start with the 10x objective - properly center the stage around the current objective before changing to another objective - when done, leave the 10x objective in standby
DSU Start-Up instructions Always: - start with the 10x objective - properly center the stage around the current objective before changing to another objective - when done, leave the 10x objective in standby
User s Guide to the LMD Laser Micro-dissection. System
 User s Guide to the LMD-6000 Laser Micro-dissection System Page 1 Glen MacDonald October 31, 2018 Start-up Procedure for Leica LMD-6000. 1. Turn on mercury lamp by pressing the rocker switch; a. beige
User s Guide to the LMD-6000 Laser Micro-dissection System Page 1 Glen MacDonald October 31, 2018 Start-up Procedure for Leica LMD-6000. 1. Turn on mercury lamp by pressing the rocker switch; a. beige
Multi-Photon Training
 Multi-Photon Training Overview This training will take approximately 4 hours. The first 2 hours will be spent learning one-on-one with your trainer, while the following 2 hours will be your opportunity
Multi-Photon Training Overview This training will take approximately 4 hours. The first 2 hours will be spent learning one-on-one with your trainer, while the following 2 hours will be your opportunity
Indiana Center for Biological Microscopy. BioRad MRC 1024 MP Confocal & Multi-Photon Microscope
 Indiana Center for Biological Microscopy BioRad MRC 1024 MP Confocal & Multi-Photon Microscope Microscope and the Attached Accessories A: B: C: D: E: F: G: H: Mercury Lamp Transmission Light Kr/Ar Laser
Indiana Center for Biological Microscopy BioRad MRC 1024 MP Confocal & Multi-Photon Microscope Microscope and the Attached Accessories A: B: C: D: E: F: G: H: Mercury Lamp Transmission Light Kr/Ar Laser
OIC. User Guide to Confocal Microscope: Zeiss LSM Pascal
 User Guide to Confocal Microscope: Zeiss LSM Pascal This guide does not cover all the details in documentation that comes with the Zeiss micro-scope. Please do find time to go through the original documentation
User Guide to Confocal Microscope: Zeiss LSM Pascal This guide does not cover all the details in documentation that comes with the Zeiss micro-scope. Please do find time to go through the original documentation
Introductory Guide to Light Microscopy - Biomedical Confocal Microscopy
 Introductory Guide to Light Microscopy - Biomedical Confocal Microscopy 7 May 2007 Michael Hooker Microscopy Facility Michael Chua microscopy@unc.edu 843-3268 6007 Thurston Bowles Wendy Salmon wendy_salmon@med.unc.edu
Introductory Guide to Light Microscopy - Biomedical Confocal Microscopy 7 May 2007 Michael Hooker Microscopy Facility Michael Chua microscopy@unc.edu 843-3268 6007 Thurston Bowles Wendy Salmon wendy_salmon@med.unc.edu
Standard Operating Procedure for the Horiba FluroMax-4
 Standard Operating Procedure for the Horiba FluroMax-4 Adapted from Horiba Operations Manual Created by Michael Delcau, Modified by Brian Lamp The Fluoromax is capable of making a variety of measurements.
Standard Operating Procedure for the Horiba FluroMax-4 Adapted from Horiba Operations Manual Created by Michael Delcau, Modified by Brian Lamp The Fluoromax is capable of making a variety of measurements.
LSM 510 Frequently Asked Questions
 LSM 510 Frequently Asked Questions you to the top of this Click on the question below to view the answer - CTRL HOME will bring page! Q: I cannot get the motorized functions of the microscope to operate
LSM 510 Frequently Asked Questions you to the top of this Click on the question below to view the answer - CTRL HOME will bring page! Q: I cannot get the motorized functions of the microscope to operate
Leica TCS SP8 X Confocal Microscope and Leica Application Suite X software DRAFT VERSION Room B123b
 Leica TCS SP8 X Confocal Microscope and Leica Application Suite X software DRAFT VERSION Room B123b User Guide Biomedicum Imaging Unit (BIU) University of Helsinki www.biu.helsinki.fi 11.11.2016 1 GENERAL...1
Leica TCS SP8 X Confocal Microscope and Leica Application Suite X software DRAFT VERSION Room B123b User Guide Biomedicum Imaging Unit (BIU) University of Helsinki www.biu.helsinki.fi 11.11.2016 1 GENERAL...1
The Paper Project Guide to Confocal Imaging at Home & in the Classroom
 The Paper Project Guide to Confocal Imaging at Home & in the Classroom http://lifesciences.asu.edu/paperproject The Paper Project Guide to Confocal Imaging at Home & in the Classroom CJ Kazilek & Dennis
The Paper Project Guide to Confocal Imaging at Home & in the Classroom http://lifesciences.asu.edu/paperproject The Paper Project Guide to Confocal Imaging at Home & in the Classroom CJ Kazilek & Dennis
PCIC Plant Cell Imaging Center (BTI, room 104B) CONFOCAL LEICA SP5 user manual
 PCIC Plant Cell Imaging Center (BTI, room 104B) CONFOCAL LEICA SP5 user manual -Turn on the mercury lamp if you need to see the fluorescence through the eye pieces. Check that its shutter is on the open
PCIC Plant Cell Imaging Center (BTI, room 104B) CONFOCAL LEICA SP5 user manual -Turn on the mercury lamp if you need to see the fluorescence through the eye pieces. Check that its shutter is on the open
Quick Guide ZEN 2. First steps with ZEN
 Quick Guide ZEN 2 First steps with ZEN Carl Zeiss Microscopy GmbH Carl-Zeiss-Promenade 10 07745 Jena, Germany microscopy@zeiss.com www.zeiss.com/microscopy Carl Zeiss Microscopy GmbH Königsallee 9-21 37081
Quick Guide ZEN 2 First steps with ZEN Carl Zeiss Microscopy GmbH Carl-Zeiss-Promenade 10 07745 Jena, Germany microscopy@zeiss.com www.zeiss.com/microscopy Carl Zeiss Microscopy GmbH Königsallee 9-21 37081
Cell Imaging Unit UIC Bárbara Fekete ZEISS AXIOIMAGER MICROSCOPE MANUAL
 Cell Imaging Unit UIC Bárbara Fekete ZEISS AXIOIMAGER MICROSCOPE MANUAL 1 TABLE OF CONTENTS ANATOMY OF THE ZEISS AXIOIMAGE MICROSCOPE 3 INITIALISATION PROCEDURE 4 LEFT SIDE OF MICROSCOPE (DETAILED VIEW)
Cell Imaging Unit UIC Bárbara Fekete ZEISS AXIOIMAGER MICROSCOPE MANUAL 1 TABLE OF CONTENTS ANATOMY OF THE ZEISS AXIOIMAGE MICROSCOPE 3 INITIALISATION PROCEDURE 4 LEFT SIDE OF MICROSCOPE (DETAILED VIEW)
Renishaw invia Raman Microscope (April 2006)
 Renishaw invia Raman Microscope (April 2006) I. Starting the System 1. The main system unit is ON all the time. 2. Switch on the Leica microscope and light source for reflective bright field (BF) imaging.
Renishaw invia Raman Microscope (April 2006) I. Starting the System 1. The main system unit is ON all the time. 2. Switch on the Leica microscope and light source for reflective bright field (BF) imaging.
Operating Procedure for Horiba Raman Microscope
 Operating Procedure for Horiba Raman Microscope SAFETY Be aware of Laser radiation at all times! Do not remove the covers of the instrument. Components are supplied with 110V electric source. Do not touch
Operating Procedure for Horiba Raman Microscope SAFETY Be aware of Laser radiation at all times! Do not remove the covers of the instrument. Components are supplied with 110V electric source. Do not touch
Presentation and analysis of multidimensional data sets
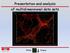 Presentation and analysis of multidimensional data sets Overview 1. 3D data visualisation Multidimensional images Data pre-processing Visualisation methods Multidimensional images wavelength time 3D image
Presentation and analysis of multidimensional data sets Overview 1. 3D data visualisation Multidimensional images Data pre-processing Visualisation methods Multidimensional images wavelength time 3D image
Confocal Raman Imaging with WITec Sensitivity - Resolution - Speed. Always - Provable - Routinely
 Confocal Raman Imaging with WITec Sensitivity - Resolution - Speed Always - Provable - Routinely WITec GmbH, Ulm, Germany, info@witec.de, www.witec.de A modular microscope series An Example: FLIM optical
Confocal Raman Imaging with WITec Sensitivity - Resolution - Speed Always - Provable - Routinely WITec GmbH, Ulm, Germany, info@witec.de, www.witec.de A modular microscope series An Example: FLIM optical
Confocal Microscope - Radiance. Basic Instructions. (Stefanie Reichelt) The Radiance. Confocal Scanning Microscope. designed and developed by
 Confocal Microscope - Radiance Basic Instructions (Stefanie Reichelt) The Radiance Confocal Scanning Microscope designed and developed by Brad Amos (MRC-LMB) Commercialised by Bio-Rad Inc. (1998 2004)
Confocal Microscope - Radiance Basic Instructions (Stefanie Reichelt) The Radiance Confocal Scanning Microscope designed and developed by Brad Amos (MRC-LMB) Commercialised by Bio-Rad Inc. (1998 2004)
Leica TCS STED. The Fast Track to Superresolution Technical Documentation
 Leica TCS STED The Fast Track to Superresolution Technical Documentation 8 9 6 7 Inverted research microscope Leica DMI6000 CS Scan head Laser and power supply Computer table Air damped optical table 6
Leica TCS STED The Fast Track to Superresolution Technical Documentation 8 9 6 7 Inverted research microscope Leica DMI6000 CS Scan head Laser and power supply Computer table Air damped optical table 6
LEXT 3D Measuring LASER Microscope
 LEXT 3D Measuring LASER Microscope Warning: This instrument may only be operated by those who have been trained by AAF staff and have read and signed the AAF laboratory policies. A) STARTUP 1. Computer
LEXT 3D Measuring LASER Microscope Warning: This instrument may only be operated by those who have been trained by AAF staff and have read and signed the AAF laboratory policies. A) STARTUP 1. Computer
Mightex Simply Brighter. Polygon400 Multiwavelength Dynamic Spatial Illuminators (Part Numbers: DSI-x-xxxx-xxxx-xxx-xxx) PRODUCT DESCRIPTION FEATURES
 FEATURES Programmable image patterns Up to 4,000 frames per second 416K pixel DLP panel Wide range of available wavelengths Support LEDs or arc lamps Diffraction-limited projection High-throughput Etenduepreserving
FEATURES Programmable image patterns Up to 4,000 frames per second 416K pixel DLP panel Wide range of available wavelengths Support LEDs or arc lamps Diffraction-limited projection High-throughput Etenduepreserving
Nikon A1-B Confocal Operating Manual. Start-up. Microscope
 Nikon A1-B Confocal Operating Manual Start-up 1. Turn on Excite 120 LED power supply. a. No need for cool down as for mercury bulb b. Open shutter by pushing down contol knob. c. Adjust intensity by turning
Nikon A1-B Confocal Operating Manual Start-up 1. Turn on Excite 120 LED power supply. a. No need for cool down as for mercury bulb b. Open shutter by pushing down contol knob. c. Adjust intensity by turning
What should I know about the 3D Restoration module? Improvision Technical Note No. 142 Last updated: 28 June, 2000
 What should I know about the 3D Restoration module? Improvision Technical Note No. 142 Last updated: 28 June, 2000 Topic This technical note discusses some aspects of iterative deconvolution and how to
What should I know about the 3D Restoration module? Improvision Technical Note No. 142 Last updated: 28 June, 2000 Topic This technical note discusses some aspects of iterative deconvolution and how to
Olympus BX51 on Aura. Introduction to the NRI-MCDB Microscopy Facility BX51 Upright Microscope
 Olympus BX51 on Aura Introduction to the NRI-MCDB Microscopy Facility BX51 Upright Microscope Contents Start-up Preparing for Imaging Part I General Part II Transmitted Brightfield Part III Transmitted
Olympus BX51 on Aura Introduction to the NRI-MCDB Microscopy Facility BX51 Upright Microscope Contents Start-up Preparing for Imaging Part I General Part II Transmitted Brightfield Part III Transmitted
Leica TCS SPE. Spectacular Imaging! Technical Documentation
 Leica TCS SPE Spectacular Imaging! Technical Documentation Leica TCS SPE Spectacular Imaging Easy to Achieve A Reliable System Affordable Excellence The high resolution spectral confocal Leica TCS SPE
Leica TCS SPE Spectacular Imaging! Technical Documentation Leica TCS SPE Spectacular Imaging Easy to Achieve A Reliable System Affordable Excellence The high resolution spectral confocal Leica TCS SPE
Quick Guide. ZEN (blue edition) First steps
 Quick Guide ZEN (blue edition) First steps Carl Zeiss Microscopy GmbH Carl-Zeiss-Promenade 10 07745 Jena, Germany microscopy@zeiss.com www.zeiss.com/microscopy Carl Zeiss Microscopy GmbH Königsallee 9-21
Quick Guide ZEN (blue edition) First steps Carl Zeiss Microscopy GmbH Carl-Zeiss-Promenade 10 07745 Jena, Germany microscopy@zeiss.com www.zeiss.com/microscopy Carl Zeiss Microscopy GmbH Königsallee 9-21
Start-up system. Nikon Wide Field Microscope Ee1078. Complete manual
 Start-up system Switch on the power socket located on the floor Switch devices on in the order of: o Mercury lamp control-unit switch on and ignite o Halogen lamp o Incubator control-box o Microscope stand
Start-up system Switch on the power socket located on the floor Switch devices on in the order of: o Mercury lamp control-unit switch on and ignite o Halogen lamp o Incubator control-box o Microscope stand
TissueFAXS SL Confocal high throughput configuration (actual appearance of the product may differ)
 TISSUEFAXS CONFOCAL TissueFAXS SL Confocal high throughput configuration (actual appearance of the product may differ) TissueFAXS Confocal provides a unique combination of digital slide scanning and laser
TISSUEFAXS CONFOCAL TissueFAXS SL Confocal high throughput configuration (actual appearance of the product may differ) TissueFAXS Confocal provides a unique combination of digital slide scanning and laser
Multi Time Series Rev
 Multi Time Series Rev. 4.0.12 The Multi Time Series program is designed to provide flexible programming of automated time dependent experiments. The basic programming unit is a single Time Series Block
Multi Time Series Rev. 4.0.12 The Multi Time Series program is designed to provide flexible programming of automated time dependent experiments. The basic programming unit is a single Time Series Block
Independent Resolution Test of
 Independent Resolution Test of as conducted and published by Dr. Adam Puche, PhD University of Maryland June 2005 as presented by (formerly Thales Optem Inc.) www.qioptiqimaging.com Independent Resolution
Independent Resolution Test of as conducted and published by Dr. Adam Puche, PhD University of Maryland June 2005 as presented by (formerly Thales Optem Inc.) www.qioptiqimaging.com Independent Resolution
NDD FLIM Systems for Leica SP2 MP and SP5 MP Multiphoton Microscopes
 NDD FLIM Systems for Leica SP2 MP and SP5 MP Multiphoton Microscopes bh FLIM systems for the confocal and the multiphoton versions of the Leica SP2 and SP5 microscopes are available since 2002 [4]. These
NDD FLIM Systems for Leica SP2 MP and SP5 MP Multiphoton Microscopes bh FLIM systems for the confocal and the multiphoton versions of the Leica SP2 and SP5 microscopes are available since 2002 [4]. These
Wide Guy: Inverted Widefield Microscope
 Wide Guy: Inverted Widefield Microscope Kyle Marchuk Adam Fries Jordan Briscoe August 2017 Contents 1 Introduction 2 2 Initial Setup 3 2.1 Hardware Startup........................................... 3
Wide Guy: Inverted Widefield Microscope Kyle Marchuk Adam Fries Jordan Briscoe August 2017 Contents 1 Introduction 2 2 Initial Setup 3 2.1 Hardware Startup........................................... 3
Olympus IX-70 Imaging Protocol
 Olympus IX-70 Imaging Protocol 1) System Startup Please note our sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused
Olympus IX-70 Imaging Protocol 1) System Startup Please note our sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused
M50 and M50RT Series. Brightfield / Darkfield Metallurgical Microscopes
 M50 and M50RT Series Brightfield / Darkfield Metallurgical Microscopes M50 / M50RT Brightfield / Darkfiefld Metallurgical Microscope Features Semi APO BF MET Infinity Corrected Objectives Part # Objective
M50 and M50RT Series Brightfield / Darkfield Metallurgical Microscopes M50 / M50RT Brightfield / Darkfiefld Metallurgical Microscope Features Semi APO BF MET Infinity Corrected Objectives Part # Objective
Last class. This class. Single molecule imaging Deconvolution. FLIM Confocal
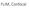 FLIM, Confocal Last class Single molecule imaging Deconvolution This class FLIM Confocal FLIM Fluorescence Lifetime IMaging FLIM Fluorescence lifetime imaging Easier and more accurate quantitation Much
FLIM, Confocal Last class Single molecule imaging Deconvolution This class FLIM Confocal FLIM Fluorescence Lifetime IMaging FLIM Fluorescence lifetime imaging Easier and more accurate quantitation Much
SERIES Z10 Z8 Z6 SOLUTIONS FOR SCIENCE AND INDUSTRY
 SERIES Z10 Z8 Z6 SOLUTIONS FOR SCIENCE AND INDUSTRY Z SERIES... SOLUTIONS FOR EVERY APPLICATION Z SERIES Z10 Z8 Z6 Z MODELS STAND AND DELIVER Ergonomic, slim design for true comfort viewing UNITRON s newly
SERIES Z10 Z8 Z6 SOLUTIONS FOR SCIENCE AND INDUSTRY Z SERIES... SOLUTIONS FOR EVERY APPLICATION Z SERIES Z10 Z8 Z6 Z MODELS STAND AND DELIVER Ergonomic, slim design for true comfort viewing UNITRON s newly
Confocal 6 Guide. Switching On Components of the system should be switched on in the following order: 1. Lasers Argon laser (488nm and 514nm)
 Confocal 6 Guide The system is kept at 37⁰C. If you require the system at a lower temperature book at least one hour before the beginning of your session to allow the system to cool down. It is your responsibility
Confocal 6 Guide The system is kept at 37⁰C. If you require the system at a lower temperature book at least one hour before the beginning of your session to allow the system to cool down. It is your responsibility
COLOCALISATION. Alexia Ferrand. Imaging Core Facility Biozentrum Basel
 COLOCALISATION Alexia Ferrand Imaging Core Facility Biozentrum Basel OUTLINE Introduction How to best prepare your samples for colocalisation How to acquire the images for colocalisation How to analyse
COLOCALISATION Alexia Ferrand Imaging Core Facility Biozentrum Basel OUTLINE Introduction How to best prepare your samples for colocalisation How to acquire the images for colocalisation How to analyse
Navigator. The Navigator
 TM002-02-A Introduction to WiRE and System start-up Navigator The Navigator Enables control of what data is open and where it is viewed Windows Measurement - Viewers and data housed together (different
TM002-02-A Introduction to WiRE and System start-up Navigator The Navigator Enables control of what data is open and where it is viewed Windows Measurement - Viewers and data housed together (different
Olympus IX-70 Imaging Protocol
 Olympus IX-70 Imaging Protocol 1) System Startup F Please note our sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for
Olympus IX-70 Imaging Protocol 1) System Startup F Please note our sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for
Non-Descanned FLIM Systems for Olympus FV-1000 and FV-300 Multiphoton Microscopes
 Non-Descanned FLIM Systems for Olympus FV-1000 and FV-300 Multiphoton Microscopes Abstract. Recently multiphoton versions of the Olympus FV 1000 and FV 300 laser scanning microscopes have become available.
Non-Descanned FLIM Systems for Olympus FV-1000 and FV-300 Multiphoton Microscopes Abstract. Recently multiphoton versions of the Olympus FV 1000 and FV 300 laser scanning microscopes have become available.
Zeiss Universal SIM. A structured illumination system based on a Zeiss Universal microscope stand. by Nidhogg
 Zeiss Universal SIM A structured illumination system based on a Zeiss Universal microscope stand. by Nidhogg Structured illumination Structured illumination applied to fluorescence microscopy is a super
Zeiss Universal SIM A structured illumination system based on a Zeiss Universal microscope stand. by Nidhogg Structured illumination Structured illumination applied to fluorescence microscopy is a super
Bruker OminiFlex MALDI-TOF Mass Spectrometer Operation Quick Start
 Bruker OminiFlex MALDI-TOF Mass Spectrometer Operation Quick Start Sample preparation for the Bruker OminiFlex MALDI-TOF Mass Spectrometer is of crucial importance. Please take care when preparing your
Bruker OminiFlex MALDI-TOF Mass Spectrometer Operation Quick Start Sample preparation for the Bruker OminiFlex MALDI-TOF Mass Spectrometer is of crucial importance. Please take care when preparing your
Spinning Disk Protocol
 Spinning Disk Protocol 1) System Startup F Please note our sign-up policy You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused time
Spinning Disk Protocol 1) System Startup F Please note our sign-up policy You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge for unused time
Short instructions. Olympus FV1000
 Olympus FV1000-1- Short instructions Short instructions. Olympus FV1000 Version 140416 This short instruction will guide you through the turn on and off sequence of the microscope system. For a detailed
Olympus FV1000-1- Short instructions Short instructions. Olympus FV1000 Version 140416 This short instruction will guide you through the turn on and off sequence of the microscope system. For a detailed
Olympus cellsens Live/TIRF Unit
 Version 4 (July 2017) Startup (Widefield Mode) Switch on power strips #1-4 (behind the monitor/lasers) in the order as they are labeled. Switch on #2 (TIRF lasers) only if you need one or more lasers for
Version 4 (July 2017) Startup (Widefield Mode) Switch on power strips #1-4 (behind the monitor/lasers) in the order as they are labeled. Switch on #2 (TIRF lasers) only if you need one or more lasers for
EPFL SV PTBIOP BIOP COURSE 2015 OPTICAL SLICING METHODS
 BIOP COURSE 2015 OPTICAL SLICING METHODS OPTICAL SLICING METHODS Scanning Methods Wide field Methods Point Scanning Deconvolution Line Scanning Multiple Beam Scanning Single Photon Multiple Photon Total
BIOP COURSE 2015 OPTICAL SLICING METHODS OPTICAL SLICING METHODS Scanning Methods Wide field Methods Point Scanning Deconvolution Line Scanning Multiple Beam Scanning Single Photon Multiple Photon Total
Olympus DeltaVision Microscope Start-Up and Shut-Down Instructions.
 DeltaVision Instructions: 1. Start-up (Olympus DV) 2. Basic operation (Olympus DV) 2.1 set-up 2.2 designing and running an experiment 3. Transferring files via FTP 4. Deconvolving files 5. File conversion
DeltaVision Instructions: 1. Start-up (Olympus DV) 2. Basic operation (Olympus DV) 2.1 set-up 2.2 designing and running an experiment 3. Transferring files via FTP 4. Deconvolving files 5. File conversion
University of Minnesota Nano Fabrication Center Standard Operating Procedure
 Equipment Name: University of Minnesota Nano Fabrication Center Coral Name: hs-scope Revision Number: 1.5 Model: HS200A Revisionist: M. Fisher Location: Bay 1 Date: 9/12/2013 1 Description The Hyphenated
Equipment Name: University of Minnesota Nano Fabrication Center Coral Name: hs-scope Revision Number: 1.5 Model: HS200A Revisionist: M. Fisher Location: Bay 1 Date: 9/12/2013 1 Description The Hyphenated
3-D. Here red spheres show the location of gold nanoparticles inside/around a cell nucleus.
 3-D The CytoViva 3-D System allows the user can locate objects of interest in a 3-D space. It does this by acquiring multiple Z planes and performing our custom software routines to locate and observe
3-D The CytoViva 3-D System allows the user can locate objects of interest in a 3-D space. It does this by acquiring multiple Z planes and performing our custom software routines to locate and observe
STEP-BY-STEP INSTRUCTIONS FOR BUILDING A FLUORESCENCE MICROSCOPE. TECHSPEC Optical Cage System
 STEP-BY-STEP INSTRUCTIONS FOR BUILDING A FLUORESCENCE MICROSCOPE TECHSPEC Optical Cage System INTRODUCTION 2 What is a Digital Fluorescence Microscope? Unlike traditional microscopes, which utilize an
STEP-BY-STEP INSTRUCTIONS FOR BUILDING A FLUORESCENCE MICROSCOPE TECHSPEC Optical Cage System INTRODUCTION 2 What is a Digital Fluorescence Microscope? Unlike traditional microscopes, which utilize an
Confocal Microscope Imaging of Single-Emitter Fluorescence and Hanbury Brown & Twiss Setup for Photon Antibunching. Edward Pei
 Confocal Microscope Imaging of Single-Emitter Fluorescence and Hanbury Brown & Twiss Setup for Photon Antibunching Edward Pei Abstract The purpose of these labs was to study single photon sources and measure
Confocal Microscope Imaging of Single-Emitter Fluorescence and Hanbury Brown & Twiss Setup for Photon Antibunching Edward Pei Abstract The purpose of these labs was to study single photon sources and measure
Non-Contact Depth Measuring Microscope System DH2/IMH
 Non-Contact Depth Measuring Microscope System DH2/IMH Specially designed focus indicator (Target Mark) facilitates focusing operation greatly. Highly accurate and repeatable measurement is possible. No
Non-Contact Depth Measuring Microscope System DH2/IMH Specially designed focus indicator (Target Mark) facilitates focusing operation greatly. Highly accurate and repeatable measurement is possible. No
COLOCALISATION. Alexia Loynton-Ferrand. Imaging Core Facility Biozentrum Basel
 COLOCALISATION Alexia Loynton-Ferrand Imaging Core Facility Biozentrum Basel OUTLINE Introduction How to best prepare your samples for colocalisation How to acquire the images for colocalisation How to
COLOCALISATION Alexia Loynton-Ferrand Imaging Core Facility Biozentrum Basel OUTLINE Introduction How to best prepare your samples for colocalisation How to acquire the images for colocalisation How to
BioImageXD Getting started
 BioImageXD Getting started Updated 2012-07-01 by Pasi Kankaanpää Introduction Welcome to BioImageXD, free open source software for biomedical image post-processing, visualization and analysis. This guide
BioImageXD Getting started Updated 2012-07-01 by Pasi Kankaanpää Introduction Welcome to BioImageXD, free open source software for biomedical image post-processing, visualization and analysis. This guide
User Operation Manual
 User Operation Manual Manual Version: 1.3 For Single Cell Bioanalyzer (SCB-402) Doc# 102-SCB-402-1.3 Table of Contents 1. General Information... 1 1.1 How to Use This Manual... 1 1.2 Operation Safety...
User Operation Manual Manual Version: 1.3 For Single Cell Bioanalyzer (SCB-402) Doc# 102-SCB-402-1.3 Table of Contents 1. General Information... 1 1.1 How to Use This Manual... 1 1.2 Operation Safety...
Lightsheet Z.1. Light Sheet Fluorescence Microscopy by Carl Zeiss. Fabrice Schmitt, Sales Manager Carl ZEISS France
 Lightsheet Z.1 Light Sheet Fluorescence Microscopy by Carl Zeiss Fabrice Schmitt, Sales Manager Carl ZEISS France 12.12.2012 Light Sheet Fluorescence Microscopy (LSFM) Principle The Principle of Light
Lightsheet Z.1 Light Sheet Fluorescence Microscopy by Carl Zeiss Fabrice Schmitt, Sales Manager Carl ZEISS France 12.12.2012 Light Sheet Fluorescence Microscopy (LSFM) Principle The Principle of Light
Jasco FP-6500 Spectrofluorimeter Updated November 14, 2017
 Jasco FP-6500 Spectrofluorimeter Updated November 14, 2017 Instrument instructions can be found at: http://academic.bowdoin.edu/chemistry/resources/instructions.shtml If you have any problems with the
Jasco FP-6500 Spectrofluorimeter Updated November 14, 2017 Instrument instructions can be found at: http://academic.bowdoin.edu/chemistry/resources/instructions.shtml If you have any problems with the
UHP-M Ultra-High Power UV-Visible LED Light Source Fluorescence Microscopy Ver. 04
 Introduction UHP-M Ultra-High Power UV-Visible LED Light Source Fluorescence Microscopy Ver. 04 Prizmatix UHP-M Dual-LED light engine is designed to substitute the Metal-Halide and Mercury lamps in routine
Introduction UHP-M Ultra-High Power UV-Visible LED Light Source Fluorescence Microscopy Ver. 04 Prizmatix UHP-M Dual-LED light engine is designed to substitute the Metal-Halide and Mercury lamps in routine
Prizmatix. Optical Specifications. Mic-LED-635 spectrum. BLCC-02 Benchtop LED Current Controller Specifications
 Mic-LED-635 High Power Collimated Red LED Light Source for Fluorescence Microscopy Featuring Prizmatix Modular Design for Multi-Wavelength and Fiberoptic Setup Ver. 01 Introduction The compact Mic-LED-635,
Mic-LED-635 High Power Collimated Red LED Light Source for Fluorescence Microscopy Featuring Prizmatix Modular Design for Multi-Wavelength and Fiberoptic Setup Ver. 01 Introduction The compact Mic-LED-635,
Prism Starter Guide 1.0 Hoskins Lab Last Modified 03/14/2017 Chris DeCiantis
 Start Up: Upon entering the laser room turn on the wall mounted Laser Power Button by pulling it away from the wall. Turn on Shutter controllers (toggle switch on back of unit). There should be a U in
Start Up: Upon entering the laser room turn on the wall mounted Laser Power Button by pulling it away from the wall. Turn on Shutter controllers (toggle switch on back of unit). There should be a U in
QUANTAX EDS SYSTEM SOP
 QUANTAX EDS SYSTEM SOP December 2017 Energy-Dispersive X-Ray Spectroscopy (EDS, EDX, EDXS or XEDS), is an analytical technique used for the elemental analysis or chemical characterization of a sample.
QUANTAX EDS SYSTEM SOP December 2017 Energy-Dispersive X-Ray Spectroscopy (EDS, EDX, EDXS or XEDS), is an analytical technique used for the elemental analysis or chemical characterization of a sample.
INFINITY-CORRECTED TUBE LENSES
 INFINITY-CORRECTED TUBE LENSES For use with Infinity-Corrected Objectives Available in Focal Lengths Used by Thorlabs, Nikon, Leica, Olympus, and Zeiss Designs for Widefield and Laser Scanning Applications
INFINITY-CORRECTED TUBE LENSES For use with Infinity-Corrected Objectives Available in Focal Lengths Used by Thorlabs, Nikon, Leica, Olympus, and Zeiss Designs for Widefield and Laser Scanning Applications
Virtual Frap User Guide
 Virtual Frap User Guide http://wiki.vcell.uchc.edu/twiki/bin/view/vcell/vfrap Center for Cell Analysis and Modeling University of Connecticut Health Center 2010-1 - 1 Introduction Flourescence Photobleaching
Virtual Frap User Guide http://wiki.vcell.uchc.edu/twiki/bin/view/vcell/vfrap Center for Cell Analysis and Modeling University of Connecticut Health Center 2010-1 - 1 Introduction Flourescence Photobleaching
Confocal vs. Deconvolution
 Confocal vs. Deconvolution Cesare Covino ALEMBIC Advanced Light and Electron Bio-Imaging Center Istituto Scientifico San Raffaele (Milano) www.hsr.it/research/alembic Fluorescence high contrast high sensibility
Confocal vs. Deconvolution Cesare Covino ALEMBIC Advanced Light and Electron Bio-Imaging Center Istituto Scientifico San Raffaele (Milano) www.hsr.it/research/alembic Fluorescence high contrast high sensibility
CoSMoS Starter Guide 1.4 Hoskins Lab Last Modified 03/03/2017 Chris DeCiantis
 Contents Start Up:... 2 Turn on Shutter controllers... 2 Turn on Lasers... 2 Turn on other equipment:... 2 Load Slide onto Sample Holder:... 2 Turn on Computer/ Start Labview/Glimpse:... 3 Shutters Activate.vi
Contents Start Up:... 2 Turn on Shutter controllers... 2 Turn on Lasers... 2 Turn on other equipment:... 2 Load Slide onto Sample Holder:... 2 Turn on Computer/ Start Labview/Glimpse:... 3 Shutters Activate.vi
ICBM Spinning Disc Microscope System User Notes
 6/17/09SCAndoriQversion1.9.1DRAFT fastmodeonly ICBM SpinningDiscMicroscopeSystemUserNotes (PerkinElmer)R2 E2 Whenyousignuponthecalendarpleaseindicate: 1)Whichobjectiveyouwilluse. 100xOilisthedefaultobjectiveonthesystem.
6/17/09SCAndoriQversion1.9.1DRAFT fastmodeonly ICBM SpinningDiscMicroscopeSystemUserNotes (PerkinElmer)R2 E2 Whenyousignuponthecalendarpleaseindicate: 1)Whichobjectiveyouwilluse. 100xOilisthedefaultobjectiveonthesystem.
Zeiss AxioImager.Z2 Fluorescence Protocol
 Zeiss AxioImager.Z2 Fluorescence Protocol 1) System Startup Please note put sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge
Zeiss AxioImager.Z2 Fluorescence Protocol 1) System Startup Please note put sign-up policy. You must inform the facility at least 24 hours beforehand if you can t come; otherwise, you will receive a charge
BIOLOGICAL MICROSCOPE
 BIOLOGICAL MICROSCOPE Instruction Manual LBM-102 B "Please read this manual carefully before using the instrument" Labnics Equipment Thank You for Buying a LABNICS BIOLOGICAL MICROSCOPE! For your personal
BIOLOGICAL MICROSCOPE Instruction Manual LBM-102 B "Please read this manual carefully before using the instrument" Labnics Equipment Thank You for Buying a LABNICS BIOLOGICAL MICROSCOPE! For your personal
Widefield High Content Screening System User Guide
 ImageXpress Micro Widefield High Content Screening System User Guide 5015321 C April 2015 This document is provided to customers who have purchased Molecular Devices equipment, software, reagents, and
ImageXpress Micro Widefield High Content Screening System User Guide 5015321 C April 2015 This document is provided to customers who have purchased Molecular Devices equipment, software, reagents, and
Model FP-6500 Spectrofluorometer Instruction Manual. FP-6500 for Windows
 Model FP-6500 Spectrofluorometer Instruction Manual FP-6500 for Windows P/N: 0302-9999 April 2000 Contents Safety Considerations...i Regulatory Statements... iii Preface... iv Installation Conditions...v
Model FP-6500 Spectrofluorometer Instruction Manual FP-6500 for Windows P/N: 0302-9999 April 2000 Contents Safety Considerations...i Regulatory Statements... iii Preface... iv Installation Conditions...v
Solar Module Quantum Efficiency Characterization System (LabVIEW Sheds Light on Solar Cell Quality Improvements)
 Solar Module Quantum Efficiency Characterization System (LabVIEW Sheds Light on Solar Cell Quality Improvements) by Richard M. Brueggman President & CEO and Gregory C. Cala, Ph.D. Chief Technology Officer
Solar Module Quantum Efficiency Characterization System (LabVIEW Sheds Light on Solar Cell Quality Improvements) by Richard M. Brueggman President & CEO and Gregory C. Cala, Ph.D. Chief Technology Officer
Fluorimeter User s Booklet
 Fluorimeter User s Booklet 1) Verify that the instrument power and the computer are both off. Then turn on the lamp. The red indicator light will glow when the lamp is on. Turn on lamp switch here. This
Fluorimeter User s Booklet 1) Verify that the instrument power and the computer are both off. Then turn on the lamp. The red indicator light will glow when the lamp is on. Turn on lamp switch here. This
A TOPCON EXAM SOLUTION. Topcon Slit Lamp Series.» Quality» Precision» Durability
 A TOPCON EXAM SOLUTION Topcon Slit Lamp Series» Quality» Precision» Durability Quality. Precision. Durability. The Topcon family of slit lamps include a wide variety of models including traditional clinical
A TOPCON EXAM SOLUTION Topcon Slit Lamp Series» Quality» Precision» Durability Quality. Precision. Durability. The Topcon family of slit lamps include a wide variety of models including traditional clinical
BA310 Epi-LED FL. New innovative Fluorescence microscope
 BA310 Epi-LED FL New innovative Fluorescence microscope BA BA310 Epi-LED FL New innovative Fluorescence microscope BA310 Epi-LED FL is designed specifically for the daily routine work in the demanding
BA310 Epi-LED FL New innovative Fluorescence microscope BA BA310 Epi-LED FL New innovative Fluorescence microscope BA310 Epi-LED FL is designed specifically for the daily routine work in the demanding
The Microscopic Investigation of Microorganisms in Sewage Purification Plants
 The Microscopic Investigation of Microorganisms in Sewage Purification Plants Introduction Biological sewage purification relies on the biodegradation of organic material in waste water by bacteria, fungi
The Microscopic Investigation of Microorganisms in Sewage Purification Plants Introduction Biological sewage purification relies on the biodegradation of organic material in waste water by bacteria, fungi
CoSMoS Starter Guide 1.6 Hoskins Lab Last Modified 03/30/2017 Chris DeCiantis
 Contents Start Up:... 2 Turn on Shutter controllers... 2 Turn on Lasers... 2 Turn on other equipment:... 2 Load Slide onto Sample Holder:... 2 Turn on Computer/ Start Labview/Glimpse:... 3 Shutters Activate.vi
Contents Start Up:... 2 Turn on Shutter controllers... 2 Turn on Lasers... 2 Turn on other equipment:... 2 Load Slide onto Sample Holder:... 2 Turn on Computer/ Start Labview/Glimpse:... 3 Shutters Activate.vi
Application for Inverted Research Microscope ECLIPSE Ti2 Series Ti2Control Ver Instruction Manual (for Android)
 M705E 17.3.Nx.4 Application for Inverted Research Microscope ECLIPSE Ti2 Series Ti2Control Ver.1.0.2 Instruction Manual (for Android) Introduction Introduction Thank you for purchasing a Nikon product.
M705E 17.3.Nx.4 Application for Inverted Research Microscope ECLIPSE Ti2 Series Ti2Control Ver.1.0.2 Instruction Manual (for Android) Introduction Introduction Thank you for purchasing a Nikon product.
OMM200 and OMM200T. Metallurgical Microscope
 OMM200 and OMM200T Metallurgical Microscope Instruction Manual (Please read the manual carefully before using the microscope) Contents Safety ----------------------------------------------------------------------------
OMM200 and OMM200T Metallurgical Microscope Instruction Manual (Please read the manual carefully before using the microscope) Contents Safety ----------------------------------------------------------------------------
Manual ANGSTROM grid confocal microscope
 Manual ANGSTROM grid confocal microscope Switching on the system 1) Start by switching on the different components of the microscope system. When using live cell imaging also start the temperature and
Manual ANGSTROM grid confocal microscope Switching on the system 1) Start by switching on the different components of the microscope system. When using live cell imaging also start the temperature and
Optical Ptychography Imaging
 Optical Ptychography Imaging Summer Project Annafee Azad Supervisors: Dr Fucai Zhang Prof Ian Robinson Summer 2014 23 October 2014 Optical Ptychography Imaging P a g e 2 Abstract This report details a
Optical Ptychography Imaging Summer Project Annafee Azad Supervisors: Dr Fucai Zhang Prof Ian Robinson Summer 2014 23 October 2014 Optical Ptychography Imaging P a g e 2 Abstract This report details a
ECLIPSE LV Series Support Tools
 M374E 07.1.NF.2 (3/3) ECLIPSE LV Series Support Tools (Setup software for ECLIPSE LV series microscopes) Software Manual Introduction Thank you for purchasing the Nikon products. This manual describes
M374E 07.1.NF.2 (3/3) ECLIPSE LV Series Support Tools (Setup software for ECLIPSE LV series microscopes) Software Manual Introduction Thank you for purchasing the Nikon products. This manual describes
Colocalization in Confocal Microscopy. Quantitative Colocalization Analysis
 Quantitative Colocalization Analysis 1 Quantitative Colocalization Analysis Colocalization of fluorescently labeled molecules (immunostainings, fluorescent proteins, etc.) is often used as the first indicator
Quantitative Colocalization Analysis 1 Quantitative Colocalization Analysis Colocalization of fluorescently labeled molecules (immunostainings, fluorescent proteins, etc.) is often used as the first indicator
Leica TCS SPE. Spectacular Imaging! Technical Documentation
 Leica TCS SPE Spectacular Imaging! Technical Documentation Leica TCS SPE Spectacular Imaging Easy to Achieve Built-in Reliability Affordable Excellence The new high resolution spectral confocal Leica TCS
Leica TCS SPE Spectacular Imaging! Technical Documentation Leica TCS SPE Spectacular Imaging Easy to Achieve Built-in Reliability Affordable Excellence The new high resolution spectral confocal Leica TCS
Zeiss Efficient Navigation (ZEN) Blue Edition Standard Operation Protocol
 Faculty Core Facility ZEN BLUE 2.3 SOP A-1 Zeiss Efficient Navigation (ZEN) Blue Edition Standard Operation Protocol Faculty Core Facility ZEN BLUE 2.3 SOP A-2 A. Content Overview. 3 Start up. 4 Display
Faculty Core Facility ZEN BLUE 2.3 SOP A-1 Zeiss Efficient Navigation (ZEN) Blue Edition Standard Operation Protocol Faculty Core Facility ZEN BLUE 2.3 SOP A-2 A. Content Overview. 3 Start up. 4 Display
Fluorescence Microscope with Spinning Disk Confocal and Total Internal Reflection Fluorescence Modules
 Open Tender Notification for the procurement of Fluorescence Microscope with Spinning Disk Confocal and Total Internal Reflection Fluorescence Modules at the Indian Institute of Science, Bangalore (Last
Open Tender Notification for the procurement of Fluorescence Microscope with Spinning Disk Confocal and Total Internal Reflection Fluorescence Modules at the Indian Institute of Science, Bangalore (Last
PALM RoboSoftware. Quick Software Guide. MicroBeam Version (EN)
 PALM RoboSoftware Quick Software Guide MicroBeam Version 2.2-0103 (EN) This document is delivered only to persons who are trained and authorized by P.A.L.M. Microlaser Technologies AG. No part of this
PALM RoboSoftware Quick Software Guide MicroBeam Version 2.2-0103 (EN) This document is delivered only to persons who are trained and authorized by P.A.L.M. Microlaser Technologies AG. No part of this
Light Sources opmaak.indd :32:50
 Light Sources Introduction Illumination light sources are needed for transmission, absorption and reflection spectroscopic setups. For the convenient coupling of the light into our range of fiber optic
Light Sources Introduction Illumination light sources are needed for transmission, absorption and reflection spectroscopic setups. For the convenient coupling of the light into our range of fiber optic
Look Ahead to Get Ahead
 Microscopy from Carl Zeiss Archive 3D Deconvolution Z-stack 3D Deconvolution Look Ahead to Get Ahead Mark & Find Multichannel Timelapse Greater brilliance and resolution More convenient and precise than
Microscopy from Carl Zeiss Archive 3D Deconvolution Z-stack 3D Deconvolution Look Ahead to Get Ahead Mark & Find Multichannel Timelapse Greater brilliance and resolution More convenient and precise than
Range Imaging Through Triangulation. Range Imaging Through Triangulation. Range Imaging Through Triangulation. Range Imaging Through Triangulation
 Obviously, this is a very slow process and not suitable for dynamic scenes. To speed things up, we can use a laser that projects a vertical line of light onto the scene. This laser rotates around its vertical
Obviously, this is a very slow process and not suitable for dynamic scenes. To speed things up, we can use a laser that projects a vertical line of light onto the scene. This laser rotates around its vertical
Chemical Characterization of Diverse Pharmaceutical Samples by Confocal Raman Microscopy
 Whitepaper Chemical Characterization of Diverse Pharmaceutical Samples by Confocal Raman Microscopy WITec GmbH, Lise-Meitner-Str. 6, 89081 Ulm, Germany, www.witec.de Introduction The development and production
Whitepaper Chemical Characterization of Diverse Pharmaceutical Samples by Confocal Raman Microscopy WITec GmbH, Lise-Meitner-Str. 6, 89081 Ulm, Germany, www.witec.de Introduction The development and production
TN425: A study of fluorescence standards confirms that OptiGrid confocal images are suitable for quantitative microscopy
 TN425: A study of fluorescence standards confirms that OptiGrid confocal images are suitable for quantitative microscopy Introduction The OptiGrid converts the illumination system of a conventional wide
TN425: A study of fluorescence standards confirms that OptiGrid confocal images are suitable for quantitative microscopy Introduction The OptiGrid converts the illumination system of a conventional wide
Supplementary Figure 1 Guide stars of progressively longer wavelengths can be used for direct wavefront sensing at increasingly large depth in the
 Supplementary Figure 1 Guide stars of progressively longer wavelengths can be used for direct wavefront sensing at increasingly large depth in the cortex of the living mouse. Typical SH images of guide
Supplementary Figure 1 Guide stars of progressively longer wavelengths can be used for direct wavefront sensing at increasingly large depth in the cortex of the living mouse. Typical SH images of guide
Setting up Micro-Magellan for Device control
 Setting up Micro-Magellan for Device control From the plug-in menu, select Micro-Magellan and the unpopulated window opens Select Configure device control The window below will open, select devices to
Setting up Micro-Magellan for Device control From the plug-in menu, select Micro-Magellan and the unpopulated window opens Select Configure device control The window below will open, select devices to
